AP Biology Unit 1- The Chemistry of Life
... Amino acids are organic molecules that serve as building blocks of proteins. They contain carbon, hydrogen, oxygen, and nitrogen atoms. There are 20 different amino acids commonly found in proteins. You don’t have to memorize the 20 amino acids. But you do have to remember that every amino acid has ...
... Amino acids are organic molecules that serve as building blocks of proteins. They contain carbon, hydrogen, oxygen, and nitrogen atoms. There are 20 different amino acids commonly found in proteins. You don’t have to memorize the 20 amino acids. But you do have to remember that every amino acid has ...
Document
... There are through-bond interactions and through-space interactions. The latter usually being a consequence of the so-called nuclear Overhauser effect (NOE). Experiments of the nuclear-Overhauser variety may establish distances between atoms. • These distances are subjected to a technique called Dist ...
... There are through-bond interactions and through-space interactions. The latter usually being a consequence of the so-called nuclear Overhauser effect (NOE). Experiments of the nuclear-Overhauser variety may establish distances between atoms. • These distances are subjected to a technique called Dist ...
2015 Blue Waters book
... Recent developments on hybrid experimental methods based on the revolutionary advance of electron microscopy have led to previously unimaginable information on cell-level structures that computational modeling requires for solidly based descriptions. Very fortunately, computational modeling can play ...
... Recent developments on hybrid experimental methods based on the revolutionary advance of electron microscopy have led to previously unimaginable information on cell-level structures that computational modeling requires for solidly based descriptions. Very fortunately, computational modeling can play ...
Protein and Enzyme Check for Understanding
... Protein and Enzyme Check for Understanding: 1. What is the monomer of a protein? 2. What is the name of the bond between the amino acids in a protein? 3. Label the following parts: ...
... Protein and Enzyme Check for Understanding: 1. What is the monomer of a protein? 2. What is the name of the bond between the amino acids in a protein? 3. Label the following parts: ...
macromolecule notes
... 1.) ________________________ compounds- Contain ______________ and __________________ atoms 2.) ________________________ compounds- Can have one or the other, but do not contain both carbon and hydrogen atoms A. Most of your body’s molecules are _______________________ compounds. a. ________________ ...
... 1.) ________________________ compounds- Contain ______________ and __________________ atoms 2.) ________________________ compounds- Can have one or the other, but do not contain both carbon and hydrogen atoms A. Most of your body’s molecules are _______________________ compounds. a. ________________ ...
Anti-Heat Shock Protein 27 (HSP27) Developed in Rabbit, IgG
... expression, correlates with the oncogenic status of the cell and plays a role in their tumorigenicity. HSP27 is expressed in the cytoplasm and colocalizes to the nucleus upon stress stimuli.2,4 HSP27, like other heat shock proteins, accumulates in cells exposed to a short period of hyperthermia, and ...
... expression, correlates with the oncogenic status of the cell and plays a role in their tumorigenicity. HSP27 is expressed in the cytoplasm and colocalizes to the nucleus upon stress stimuli.2,4 HSP27, like other heat shock proteins, accumulates in cells exposed to a short period of hyperthermia, and ...
Branched Chain Amino Acid
... What Are They? • Amino acids are the building blocks of protein and there are three specific essential amino acids that make up as much as 33 percent of skeletal muscle. • These amino acids are known as the branched chain amino acids (BCAAs) and they include, valine, leucine and isoleucine. • Val ...
... What Are They? • Amino acids are the building blocks of protein and there are three specific essential amino acids that make up as much as 33 percent of skeletal muscle. • These amino acids are known as the branched chain amino acids (BCAAs) and they include, valine, leucine and isoleucine. • Val ...
Document
... In 1912, M. von Laue discovered that crystals, solid structures forme In the early ‘50s, pioneering scientists such as D. Hodgkin were able ...
... In 1912, M. von Laue discovered that crystals, solid structures forme In the early ‘50s, pioneering scientists such as D. Hodgkin were able ...
Lecture 1: Protein sorting (endoplasmic reticulum and Golgi
... Step 2: The SRP escorts the complex to the ER membrane, where it binds to the SRP receptor. Step 3: The SRP is released, the ribosome binds to a membrane translocation complex of Sec61 proteins, and the signal sequence is inserted into a membrane channel. Step 4: Translation resumes, and the growing ...
... Step 2: The SRP escorts the complex to the ER membrane, where it binds to the SRP receptor. Step 3: The SRP is released, the ribosome binds to a membrane translocation complex of Sec61 proteins, and the signal sequence is inserted into a membrane channel. Step 4: Translation resumes, and the growing ...
the essence of life
... of 4 electrons, leaving 4 free spaces • Organic molecules are thus generally based on a unit shape of a triangular based pyramid • Organic molecules are generally defined by the elements other than carbon in them, and by the types of bonds they form with carbon ...
... of 4 electrons, leaving 4 free spaces • Organic molecules are thus generally based on a unit shape of a triangular based pyramid • Organic molecules are generally defined by the elements other than carbon in them, and by the types of bonds they form with carbon ...
DISULFIDE GROUPS Disulfide bonds in proteins are
... Disulfide bonds in proteins are subject to cleavage by mild reducing agents (Section 8-1), by a few oxidizing agents (Section 8-6), or by nucleophilic displacement (Section 8-2). Of the procedures for cleavage, those involving reduction are probably the mildest and most specific. Many smaller protei ...
... Disulfide bonds in proteins are subject to cleavage by mild reducing agents (Section 8-1), by a few oxidizing agents (Section 8-6), or by nucleophilic displacement (Section 8-2). Of the procedures for cleavage, those involving reduction are probably the mildest and most specific. Many smaller protei ...
Protein Synthesis
... ribosomes by the tRNA molecules to be assembled into a protein (translation) • The order of the amino acids is determined by the codon on the mRNA ...
... ribosomes by the tRNA molecules to be assembled into a protein (translation) • The order of the amino acids is determined by the codon on the mRNA ...
lec07_2013 - Andrew.cmu.edu
... therefore unfavorable. The peptide bond is said to be a "partial double bond". The N-H group within the peptide bond can only act as a hydrogen bond donor. The partial negative charge on the nitrogen is delocalized. Cis and trans Peptide Bonds: Two possible orientations of the peptide bond that main ...
... therefore unfavorable. The peptide bond is said to be a "partial double bond". The N-H group within the peptide bond can only act as a hydrogen bond donor. The partial negative charge on the nitrogen is delocalized. Cis and trans Peptide Bonds: Two possible orientations of the peptide bond that main ...
Ch. 2 The Chemistry of Life
... Proteins - molecules that contain nitrogen, carbon, hydrogen, & oxygen Proteins are made up of chains of amino acids ...
... Proteins - molecules that contain nitrogen, carbon, hydrogen, & oxygen Proteins are made up of chains of amino acids ...
Slide 1 - Ommbid.com
... Relationship of integral and peripheral membrane proteins to the membrane phospholipid bilayer. Integral membrane proteins (a) have portions of their mass embedded in the membrane that interact directly with the hydrophobic tails of the phospholipids. Other portions of these proteins are exposed on ...
... Relationship of integral and peripheral membrane proteins to the membrane phospholipid bilayer. Integral membrane proteins (a) have portions of their mass embedded in the membrane that interact directly with the hydrophobic tails of the phospholipids. Other portions of these proteins are exposed on ...
File
... The Biuret reagent, regularly colored blue, is used to identify proteins. When the copper ions (Cu2+) in the reagent interact with peptide bonds, a violet color is produced. Ror the interaction between Cu2+ and the peptide bonds to result in a color change, a minimum of 4-6 peptide bonds is required ...
... The Biuret reagent, regularly colored blue, is used to identify proteins. When the copper ions (Cu2+) in the reagent interact with peptide bonds, a violet color is produced. Ror the interaction between Cu2+ and the peptide bonds to result in a color change, a minimum of 4-6 peptide bonds is required ...
exam bullet points
... Because wavelength of electron beam smaller Differences between prokaryotic and eukaryotic ribosomes ...
... Because wavelength of electron beam smaller Differences between prokaryotic and eukaryotic ribosomes ...
Ms Gentry`s Proteins powerpoint File
... •Explain the term secondary structure with reference to hydrogen bonding. •Explain the term tertiary structure with reference to hydrophobic and hydrophilic interactions, disulfide bonds and ionic interactions. •Explain the term quaternary structure with reference to the structure of haemoglobin. •D ...
... •Explain the term secondary structure with reference to hydrogen bonding. •Explain the term tertiary structure with reference to hydrophobic and hydrophilic interactions, disulfide bonds and ionic interactions. •Explain the term quaternary structure with reference to the structure of haemoglobin. •D ...
The role of the C-terminal tail of the ribosomal protein S13 in protein
... acid bound tRNA and the mRNA, and the peptidyl transferase center where the peptide bond formation occurs. There are also several factors to assist the function of the ribosome including initiation factors to aid in starting the translation, elongation factors during translation and release factors ...
... acid bound tRNA and the mRNA, and the peptidyl transferase center where the peptide bond formation occurs. There are also several factors to assist the function of the ribosome including initiation factors to aid in starting the translation, elongation factors during translation and release factors ...
A7: Decoding genome encoded host-pathogen
... adaptation. Our analysis will unravel their functional relations and organizational principles. Previous work: We have identified GGDEF, EAL, HD and PilZ domain containing 16179 proteins in 779 completely sequenced bacterial genomes. We found that these domains cooccur with 124 other domains, which ...
... adaptation. Our analysis will unravel their functional relations and organizational principles. Previous work: We have identified GGDEF, EAL, HD and PilZ domain containing 16179 proteins in 779 completely sequenced bacterial genomes. We found that these domains cooccur with 124 other domains, which ...
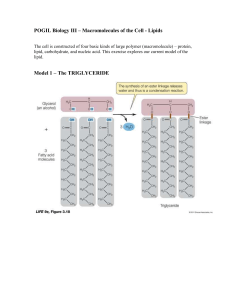
pogil 3
... Using the diagrams above, your textbook, and your note set, answer the following questions. 1. In model 1, what does the C stand for? The O? The H? What does the straight line between atoms represent? ...
... Using the diagrams above, your textbook, and your note set, answer the following questions. 1. In model 1, what does the C stand for? The O? The H? What does the straight line between atoms represent? ...
Cyclol

The cyclol hypothesis is the first structural model of a folded, globular protein. It was developed by Dorothy Wrinch in the late 1930s, and was based on three assumptions. Firstly, the hypothesis assumes that two peptide groups can be crosslinked by a cyclol reaction (Figure 1); these crosslinks are covalent analogs of non-covalent hydrogen bonds between peptide groups. These reactions have been observed in the ergopeptides and other compounds. Secondly, it assumes that, under some conditions, amino acids will naturally make the maximum possible number of cyclol crosslinks, resulting in cyclol molecules (Figure 2) and cyclol fabrics (Figure 3). These cyclol molecules and fabrics have never been observed. Finally, the hypothesis assumes that globular proteins have a tertiary structure corresponding to Platonic solids and semiregular polyhedra formed of cyclol fabrics with no free edges. Such ""closed cyclol"" molecules have not been observed either.Although later data demonstrated that this original model for the structure of globular proteins needed to be amended, several elements of the cyclol model were verified, such as the cyclol reaction itself and the hypothesis that hydrophobic interactions are chiefly responsible for protein folding. The cyclol hypothesis stimulated many scientists to research questions in protein structure and chemistry, and was a precursor of the more accurate models hypothesized for the DNA double helix and protein secondary structure. The proposal and testing of the cyclol model also provides an excellent illustration of empirical falsifiability acting as part of the scientific method.