Constitutional isomers and stereoisomers
... i) State the structural requirement for a molecule to be able to exist as enantiomers. ii) Describe a property of enantiomers that would enable them to be distinguished from each other. iii) Draw the structural formulae of the enantiomers of alcohol A. ...
... i) State the structural requirement for a molecule to be able to exist as enantiomers. ii) Describe a property of enantiomers that would enable them to be distinguished from each other. iii) Draw the structural formulae of the enantiomers of alcohol A. ...
oxidation and reduction
... hydrolysed before it is re-oxidised, or does it get oxidised first? The details are still being sorted out. The key observation was: some amines accelerate the reaction (by complexing to Os), though some amines slow it down. If you have an amine which is chiral and accelerates the reaction, this wil ...
... hydrolysed before it is re-oxidised, or does it get oxidised first? The details are still being sorted out. The key observation was: some amines accelerate the reaction (by complexing to Os), though some amines slow it down. If you have an amine which is chiral and accelerates the reaction, this wil ...
Ch12-Alcohols-Grignard
... Retrosynthetic Analysis Retrosynthetic analysis is the process by which we plan a synthesis by reasoning backward from the desired product (the "target molecule"). ...
... Retrosynthetic Analysis Retrosynthetic analysis is the process by which we plan a synthesis by reasoning backward from the desired product (the "target molecule"). ...
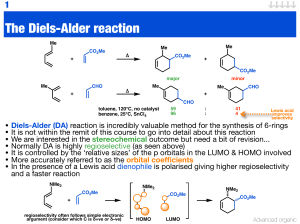
The Diels-Alder reaction
... chiral auxiliary often excellent ee’s; built in extra steps to introduce resolving agent and remove auxiliary ...
... chiral auxiliary often excellent ee’s; built in extra steps to introduce resolving agent and remove auxiliary ...
Synthesis of the hexoses
... so the success of a reaction above the dotted line does not necessarily ...
... so the success of a reaction above the dotted line does not necessarily ...
COMMON SYNTHETIC SEQUENCES FOR OCHEM I
... and reductions actually take place in biological systems. There are some differences in the way these reactions happen in biological systems as opposed to the organic chemistry lab. For example most biological reactions take place in water as the medium, not in organic solvents like methylene chlori ...
... and reductions actually take place in biological systems. There are some differences in the way these reactions happen in biological systems as opposed to the organic chemistry lab. For example most biological reactions take place in water as the medium, not in organic solvents like methylene chlori ...
Alcohol - SanfordChemistry
... Alcohols are one form of organic compounds which contain Oxygen, another being Ethers. Ethers are very similar in composition to Alcohol. Alcohol: R – OH Ethers: R-O-R Two other classes including Carboxylic acids and Esters (also having similar molecular structures) both have two oxygens, one of th ...
... Alcohols are one form of organic compounds which contain Oxygen, another being Ethers. Ethers are very similar in composition to Alcohol. Alcohol: R – OH Ethers: R-O-R Two other classes including Carboxylic acids and Esters (also having similar molecular structures) both have two oxygens, one of th ...
102 Lecture Ch15
... - the right hand is the mirror image of the left hand - no matter how you turn them, they can’t be superimposed • Many organic compounds are also chiral - most biomolecules (amino acids, sugars, etc.) are chiral and usually only one of the stereoisomers is used • In order for a carbon in an organic ...
... - the right hand is the mirror image of the left hand - no matter how you turn them, they can’t be superimposed • Many organic compounds are also chiral - most biomolecules (amino acids, sugars, etc.) are chiral and usually only one of the stereoisomers is used • In order for a carbon in an organic ...
Naming organic compounds
... Indicate the position of the branches with a number, numbering from the end nearest the functional group. If there is more than one branch, the branches are identified in alphabetical order ignoring any di, tri etc. Be aware! Each branch needs to be numbered individually, even if they are attached t ...
... Indicate the position of the branches with a number, numbering from the end nearest the functional group. If there is more than one branch, the branches are identified in alphabetical order ignoring any di, tri etc. Be aware! Each branch needs to be numbered individually, even if they are attached t ...
Laboratory 22: Properties of Alcohols Introduction Discussion
... The most common alcohols are colorless liquids at room temperature. Their melting and boiling points are considerably higher than those of alkanes, alkenes, and alkynes of similar size due to the ability to hydrogen bond (See Hein 13.8). The ability to form hydrogen bonds also makes low molar mass a ...
... The most common alcohols are colorless liquids at room temperature. Their melting and boiling points are considerably higher than those of alkanes, alkenes, and alkynes of similar size due to the ability to hydrogen bond (See Hein 13.8). The ability to form hydrogen bonds also makes low molar mass a ...
Title Carbonyl reduction with CaH2 and R3SiCl catalyzed by ZnCl2
... 1. N. Greeves, In Comprehensive Organic Synthesis; B. M. Trost and I. Fleming, Eds.; Pergamon Press: Oxford, 1991, vol. 9, p. 1. J. Seyden-Penne, Reductions by the Alumino- and Borohydrides in Organic Synthesis; VHC: 1991. 2. Ohkuma, T.; Hashiguchi, S.; Noyori, R. J. Org. Chem. 1994, ...
... 1. N. Greeves, In Comprehensive Organic Synthesis; B. M. Trost and I. Fleming, Eds.; Pergamon Press: Oxford, 1991, vol. 9, p. 1. J. Seyden-Penne, Reductions by the Alumino- and Borohydrides in Organic Synthesis; VHC: 1991. 2. Ohkuma, T.; Hashiguchi, S.; Noyori, R. J. Org. Chem. 1994, ...
Paper
... In the course of determining the group composition of the waxes, the esters obtained can be hydrolyzed in a more ready manner than n ative esters to serve as a source of the replenishment of the saponifiable fraction. Chain length distribution inherent in saponifiable acids contained in the liquid ( ...
... In the course of determining the group composition of the waxes, the esters obtained can be hydrolyzed in a more ready manner than n ative esters to serve as a source of the replenishment of the saponifiable fraction. Chain length distribution inherent in saponifiable acids contained in the liquid ( ...
Properties of Alcohol
... Test for methanol: Mix in a test tube about 1 part of methanol, 4 parts of dil. NaOH solution, and 1 ml of iodine solution then warm. ...
... Test for methanol: Mix in a test tube about 1 part of methanol, 4 parts of dil. NaOH solution, and 1 ml of iodine solution then warm. ...
Alcohol oxidation
... ozone. This indicates complete consumption of the alkene. Alternatively, various other chemicals can be used as indicators of this endpoint by detecting the presence of ozone. If ozonolysis is performed by bubbling a stream of ozone-enriched oxygen through the reaction mixture, the gas that bubbles ...
... ozone. This indicates complete consumption of the alkene. Alternatively, various other chemicals can be used as indicators of this endpoint by detecting the presence of ozone. If ozonolysis is performed by bubbling a stream of ozone-enriched oxygen through the reaction mixture, the gas that bubbles ...
Chem 341 Review for Finals Key Reactions Mechanisms
... Chem 341 Review for Finals Reaction Types • RMgBr (Grignard Reagents) –Preparation from alkyl halides –Reactions with aldehydes, ketones, and esters • Nucleophilic Addition to Ketones – Irreversible additions: LiAlH4 RMgBr – Reversible additions: alcohols => hemiacetal & acetal – Addition-Eliminati ...
... Chem 341 Review for Finals Reaction Types • RMgBr (Grignard Reagents) –Preparation from alkyl halides –Reactions with aldehydes, ketones, and esters • Nucleophilic Addition to Ketones – Irreversible additions: LiAlH4 RMgBr – Reversible additions: alcohols => hemiacetal & acetal – Addition-Eliminati ...
Alcohols and Carbonyls - Deans Community High School
... C2H4 + H2O CH3CH2OH Cracking fractions from crude oil is a cheaper way to produce ethanol. (more expensive than making petrol) Reaction Conditions: 300oC , High Pressure 60 Atm. , phosphoric acid catalyst. ...
... C2H4 + H2O CH3CH2OH Cracking fractions from crude oil is a cheaper way to produce ethanol. (more expensive than making petrol) Reaction Conditions: 300oC , High Pressure 60 Atm. , phosphoric acid catalyst. ...
Additional file 1
... anhydrous CH2Cl2 (50 mL) at 0°C. After the addition was complete, the mixture was stirred at room temperature for 2 h, then diluted with anhydrous ether and filtered through celite. The filtrate was concentrated under vacuum and the residue was purified by chromatography to yield ketone 3. 1H NMR (C ...
... anhydrous CH2Cl2 (50 mL) at 0°C. After the addition was complete, the mixture was stirred at room temperature for 2 h, then diluted with anhydrous ether and filtered through celite. The filtrate was concentrated under vacuum and the residue was purified by chromatography to yield ketone 3. 1H NMR (C ...
Substrate scope of the re-engineered enzyme, FucO D93
... of most enzymes to work well in water solutions, without strong acids or bases, makes them powerful tools when looking for environmentally friendly alternatives for mass production of chemicals. [9] The catalytical part of a protein is called the active site. This is often a pocket or fold in the en ...
... of most enzymes to work well in water solutions, without strong acids or bases, makes them powerful tools when looking for environmentally friendly alternatives for mass production of chemicals. [9] The catalytical part of a protein is called the active site. This is often a pocket or fold in the en ...
Synthesis of (−)-Epibatidine - David A. Evans
... Although the reduction proceeded without affecting the chloropyridine ring,21 a 75:25 mixture of inseparable alcohols was obtained. The structural assignment of the major diastereomer was complicated as a result of slowly interconverting conformations as observed by 1H NMR spectroscopy, even at elev ...
... Although the reduction proceeded without affecting the chloropyridine ring,21 a 75:25 mixture of inseparable alcohols was obtained. The structural assignment of the major diastereomer was complicated as a result of slowly interconverting conformations as observed by 1H NMR spectroscopy, even at elev ...
14. The Direct and Enantioselective Organocatalytic -Oxidation of Aldehydes
... oxidation approaches to this asymmetric motif have relied exclusively upon the use of preformed enolates or enolate equivalents.1 As part of a program aimed at developing broadly useful organic catalysts for asymmetric synthesis,2 we recently reported the first direct proline-catalyzed cross-aldol r ...
... oxidation approaches to this asymmetric motif have relied exclusively upon the use of preformed enolates or enolate equivalents.1 As part of a program aimed at developing broadly useful organic catalysts for asymmetric synthesis,2 we recently reported the first direct proline-catalyzed cross-aldol r ...
Toluenediamine
... clay impregnated with anhydrous cupric nitrate. The yield after 4 h at 25C was 95%, and the product mixture contained 86% 2,4-DNT and 13% 2,6-DNT (ratio 6.6:1). Later investigations of another group revealed that the same results could also be obtained with a different solvent (dichloromethane inst ...
... clay impregnated with anhydrous cupric nitrate. The yield after 4 h at 25C was 95%, and the product mixture contained 86% 2,4-DNT and 13% 2,6-DNT (ratio 6.6:1). Later investigations of another group revealed that the same results could also be obtained with a different solvent (dichloromethane inst ...
EXPERIMENT 3: The Grignard Reaction: Synthesis of
... The reactions involved in the synthesis of complex organic molecules can commonly be categorized into either functional group interconversions or skeleton building reactions. The latter category, primarily those involving carbon-carbon bond formations, is most important in anabolic organic synthesis ...
... The reactions involved in the synthesis of complex organic molecules can commonly be categorized into either functional group interconversions or skeleton building reactions. The latter category, primarily those involving carbon-carbon bond formations, is most important in anabolic organic synthesis ...
-23- ORGANIC CHEMISTRY A. STRUCTURE AND ISOMERISM 1
... (b) ester formation (substitution) - alcohol/H+ (c) amide formation (substitution) - amine or ammonia Esters hydrolysis (substitution) - H2O and H+ or HOAmides hydrolysis (substitution) - H2O and H+ or HOAmines (a) reaction with acid (acid-base) - e.g. HCl, CH3COOH, etc. (b) amide formation (substit ...
... (b) ester formation (substitution) - alcohol/H+ (c) amide formation (substitution) - amine or ammonia Esters hydrolysis (substitution) - H2O and H+ or HOAmides hydrolysis (substitution) - H2O and H+ or HOAmines (a) reaction with acid (acid-base) - e.g. HCl, CH3COOH, etc. (b) amide formation (substit ...
Full answers
... Co2+ is a d7 system, so must have at least 1 unpaired electron. Consequently it must be paramagnetic. • Briefly explain how a catalyst works. A catalyst provides an alternative reaction pathway that has a lower activation energy. This allows the reaction to proceed at lower temperatures or under mil ...
... Co2+ is a d7 system, so must have at least 1 unpaired electron. Consequently it must be paramagnetic. • Briefly explain how a catalyst works. A catalyst provides an alternative reaction pathway that has a lower activation energy. This allows the reaction to proceed at lower temperatures or under mil ...
Alcohols, Phenols, and Thiols
... Saytzeff’s Rule According to Saytzeff’s rule, the dehydration of a secondary alcohol favors the product in which hydrogen is removed from the carbon atom in the chain with the smaller number of H atoms ...
... Saytzeff’s Rule According to Saytzeff’s rule, the dehydration of a secondary alcohol favors the product in which hydrogen is removed from the carbon atom in the chain with the smaller number of H atoms ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.