Organic Synthesis II
... Mechanisms for many oxidation reactions (even well-known ones) are significantly more complex than drawn throughout this course (and in many cases are not known or understood). Some are based on factual mechanistic data; some should be treated more as a mnemonic than explanation. ...
... Mechanisms for many oxidation reactions (even well-known ones) are significantly more complex than drawn throughout this course (and in many cases are not known or understood). Some are based on factual mechanistic data; some should be treated more as a mnemonic than explanation. ...
Chapter 7- Alcohols
... Usually use SOCl2 or PBr3 with 1o or 2o alcohols- reaction of 3o alcohols with H-X is so fast these reagents are unnecessary ...
... Usually use SOCl2 or PBr3 with 1o or 2o alcohols- reaction of 3o alcohols with H-X is so fast these reagents are unnecessary ...
Microsoft Word
... The homoallylic alcohol 36a was treated with a catalytic amount of osmium tetroxide to give the 1,2-diol, which was oxidatively cleaved with NaIO4 in THF–H2O (5:1) to afford the aldehyde (Scheme 16). The aldehyde was further oxidized to an acid 47 with NaClO2 and NaH2PO4.2H2O in t-BuOH at room tempe ...
... The homoallylic alcohol 36a was treated with a catalytic amount of osmium tetroxide to give the 1,2-diol, which was oxidatively cleaved with NaIO4 in THF–H2O (5:1) to afford the aldehyde (Scheme 16). The aldehyde was further oxidized to an acid 47 with NaClO2 and NaH2PO4.2H2O in t-BuOH at room tempe ...
ethers - WordPress.com
... Nomenclatures, methods nof preparations, properties, reactions and uses ...
... Nomenclatures, methods nof preparations, properties, reactions and uses ...
Expt #7: Synthesis of Esters using Acetic
... There are problems with this reaction however. The esterification reaction is reversible with an equilibrium constant that favors the products only slightly. Excess of either the carboxylic acid or the alcohol must be used to drive the equilibrium to the right (Le Chatelier's Principle) in order to ...
... There are problems with this reaction however. The esterification reaction is reversible with an equilibrium constant that favors the products only slightly. Excess of either the carboxylic acid or the alcohol must be used to drive the equilibrium to the right (Le Chatelier's Principle) in order to ...
Grant MacEwan College - Faculty Web Pages
... Description: This is the second course in organic chemistry. The topics covered include structural and chemical properties of alkenes, alkynes, alcohols, phenols, ethers, aromatic compounds. Aldehyde, ketones, amines, carboxylic acids, and carboxylic acid derivatives. Illustration of these functiona ...
... Description: This is the second course in organic chemistry. The topics covered include structural and chemical properties of alkenes, alkynes, alcohols, phenols, ethers, aromatic compounds. Aldehyde, ketones, amines, carboxylic acids, and carboxylic acid derivatives. Illustration of these functiona ...
DME Rate Equations
... performance was developed and tested with the experimental data. The mean absolute deviation, concerning the data for the steady state conditions, was less than 8 %. Keywords: DME, kinetic parameter, fixed-bed reactor, dynamic modeling ...
... performance was developed and tested with the experimental data. The mean absolute deviation, concerning the data for the steady state conditions, was less than 8 %. Keywords: DME, kinetic parameter, fixed-bed reactor, dynamic modeling ...
Properties of haloalkanes
... between haloalkanes and alcoholic ammonia: R—X + NH3(alc) → R—NH2 + HX amine It must be alcoholic ammonia: if water is present alcohol will be formed instead. ...
... between haloalkanes and alcoholic ammonia: R—X + NH3(alc) → R—NH2 + HX amine It must be alcoholic ammonia: if water is present alcohol will be formed instead. ...
Pyrrolidine-2-carboxylic Acid (l
... This feature focuses on a reagent chosen by a postgraduate, highlighting the uses and preparation of the reagent in current research ...
... This feature focuses on a reagent chosen by a postgraduate, highlighting the uses and preparation of the reagent in current research ...
catalysis lecture
... A catalyst is a substance that increases the rate at which a chemical reaction approaches equilibrium, but is not consumed in the process. Thus a catalyst affects the but does not affect the ...
... A catalyst is a substance that increases the rate at which a chemical reaction approaches equilibrium, but is not consumed in the process. Thus a catalyst affects the but does not affect the ...
Ch14 Lecture
... by humans into glucose. • Cellulose, the major component of wood, cotton, and flax, gives rigidity to plants, however it cannot be metabolized by humans. ...
... by humans into glucose. • Cellulose, the major component of wood, cotton, and flax, gives rigidity to plants, however it cannot be metabolized by humans. ...
Lab 9 - Academic Computer Center
... The second comes from the workup of the reaction, which is normally conducted in aqueous acid. Sodium borohydride, NaBH4, is the mildest of the three hydride reagents and is easy to use in the lab, because it is soluble in water, methanol and ethanol and does not react with these solvents. Therefore ...
... The second comes from the workup of the reaction, which is normally conducted in aqueous acid. Sodium borohydride, NaBH4, is the mildest of the three hydride reagents and is easy to use in the lab, because it is soluble in water, methanol and ethanol and does not react with these solvents. Therefore ...
Synthesis and Structure of Alcohols
... (1) Name the longest carbon chain bearing the –OH group. Drop the last –e from the alkane name and add –ol to obtain the root name. (2) Number the longest chain starting at the end nearest the –OH group, and designate a number for the –OH group. (Hydroxyl has greater priority than carbon-carbon mult ...
... (1) Name the longest carbon chain bearing the –OH group. Drop the last –e from the alkane name and add –ol to obtain the root name. (2) Number the longest chain starting at the end nearest the –OH group, and designate a number for the –OH group. (Hydroxyl has greater priority than carbon-carbon mult ...
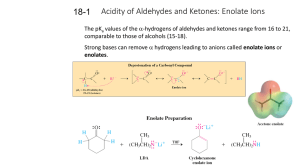
18-1 Enolates (PPT)
... Either equilibration is fast and reversible in solution in the presence of the required catalyst. ...
... Either equilibration is fast and reversible in solution in the presence of the required catalyst. ...
Iron(II) Chloride–1,1′-Binaphthyl-2,2′-diamine
... (L1) (Figure 1). When the reaction was carried out with iron(II) chloride, but without a ligand, only a 27% yield of the product was obtained. ...
... (L1) (Figure 1). When the reaction was carried out with iron(II) chloride, but without a ligand, only a 27% yield of the product was obtained. ...
AS Chemistry: Paper 2 Physical Inorganic Organic 1.0 Basic
... Complete other half of paper 1B Qu 6 - 23 for next lesson 20/4/16 Wednesday Read over 3.4 Alkenes – address problems Write out definitions (in boxes) Produce standard answers for: ...
... Complete other half of paper 1B Qu 6 - 23 for next lesson 20/4/16 Wednesday Read over 3.4 Alkenes – address problems Write out definitions (in boxes) Produce standard answers for: ...
Microsoft Word
... hydrogenation reaction have been obtained. The hydrogenation activity and surface properties of Raney-Ni are found to be strongly influenced by its preparation conditions. The optimum preparation conditions for the Raney-Ni catalyst having the maximum activity for the hydrogenation are as follows: c ...
... hydrogenation reaction have been obtained. The hydrogenation activity and surface properties of Raney-Ni are found to be strongly influenced by its preparation conditions. The optimum preparation conditions for the Raney-Ni catalyst having the maximum activity for the hydrogenation are as follows: c ...
Handout 7
... In conclusion, all steps included in the conversion of an aldehyde or ketone to acetal or ketal via hemiacetal or hemiketal as intermediates, are reversible. Performing the reaction in large excess of an anhydrous alcohol and a small amount of an anhydrous acid will strongly favour the formation of ...
... In conclusion, all steps included in the conversion of an aldehyde or ketone to acetal or ketal via hemiacetal or hemiketal as intermediates, are reversible. Performing the reaction in large excess of an anhydrous alcohol and a small amount of an anhydrous acid will strongly favour the formation of ...
Document
... Chiral phosphine ligands have been developed to synthesize optically active products. Synthesis of L-DOPA (Used in the treatment of Parkinson’s diseases) Synthetic route was developed by Knowles & co-workers at Monsanto ...
... Chiral phosphine ligands have been developed to synthesize optically active products. Synthesis of L-DOPA (Used in the treatment of Parkinson’s diseases) Synthetic route was developed by Knowles & co-workers at Monsanto ...
Organic Reactions Worksheet
... For the questions below, give full structural diagrams and names for all reactants and products. Also indicate any catalysts/reaction conditions for the reaction. Also indicate the TYPE OF REACTION. 1. Oxidation with primary and secondary alcohols via sulfuric acid and potassium dichromate. a. What ...
... For the questions below, give full structural diagrams and names for all reactants and products. Also indicate any catalysts/reaction conditions for the reaction. Also indicate the TYPE OF REACTION. 1. Oxidation with primary and secondary alcohols via sulfuric acid and potassium dichromate. a. What ...
Chapter 3 Properties of organic compounds
... Aldehydes (alkanals – RCHO) and ketones (alkanones – RCOR’) Aldehydes are Ý`Ãi` to carboxylic acids by reaction with Cr2O72–/H+ or MnO4–/H+, as well as mild oxidising agents such as Ag+ and Cu2+, that are too weak to oxidise alcohols. Tollens’ test – reaction with a complex ion Ag(NH3)2+, which o ...
... Aldehydes (alkanals – RCHO) and ketones (alkanones – RCOR’) Aldehydes are Ý`Ãi` to carboxylic acids by reaction with Cr2O72–/H+ or MnO4–/H+, as well as mild oxidising agents such as Ag+ and Cu2+, that are too weak to oxidise alcohols. Tollens’ test – reaction with a complex ion Ag(NH3)2+, which o ...
lecture 1 - alcohols-ethers
... chemical reactions • Therefore, they are ideal to use as solvents ...
... chemical reactions • Therefore, they are ideal to use as solvents ...
ALcohols CPP
... THE CHEMISTRY OF ALCOHOLS INTRODUCTION This Powerpoint show is one of several produced to help students understand selected topics at AS and A2 level Chemistry. It is based on the requirements of the AQA and OCR specifications but is suitable for other examination boards. Individual students may use ...
... THE CHEMISTRY OF ALCOHOLS INTRODUCTION This Powerpoint show is one of several produced to help students understand selected topics at AS and A2 level Chemistry. It is based on the requirements of the AQA and OCR specifications but is suitable for other examination boards. Individual students may use ...
Ruthenium(II) Complexes Bearing a Pyridyl-Supported Pyrazolyl
... palladium(II) analogues,8 while the 13C resonance of its RudC group appeared at 192.5 ppm in solution. The 1H NMR signals of the NH and pyrazolyl CH moieties in 7 were shifted downfield by about 1.04 and 0.21 ppm, respectively, as compared with those of the ligand precursor 6 (δNH, 12.95 ppm; δpyraz ...
... palladium(II) analogues,8 while the 13C resonance of its RudC group appeared at 192.5 ppm in solution. The 1H NMR signals of the NH and pyrazolyl CH moieties in 7 were shifted downfield by about 1.04 and 0.21 ppm, respectively, as compared with those of the ligand precursor 6 (δNH, 12.95 ppm; δpyraz ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.