Dehydration of n-propanol and methanol to produce
... ion-exchange resin Amberlyst 36 was used as the catalyst [12]. MPE has been produced as fraction of biocrude produced using Hydrothermal Upgrading (HTU) process by thermochemical conversion of biomass. This process requires high temperature and pressure, around 350 °C and 180 bar respectively [13]. ...
... ion-exchange resin Amberlyst 36 was used as the catalyst [12]. MPE has been produced as fraction of biocrude produced using Hydrothermal Upgrading (HTU) process by thermochemical conversion of biomass. This process requires high temperature and pressure, around 350 °C and 180 bar respectively [13]. ...
Project Overview
... from a compound containing an –OH group, an –NH– group, an –SH group, a –CO2H group, or an –SO3H group ...
... from a compound containing an –OH group, an –NH– group, an –SH group, a –CO2H group, or an –SO3H group ...
A Straightforward Route to Enantiopure Pyrrolizidines and
... especially ethylene glycol. The principal shortcoming of all homogeneous CO hydrogenation reactions is their low catalytic activity, which results in the need to use high catalyst loadings and drastic reaction conditions (usually at temperatures above 200°C and pressures above 30MPa) [34]. Rh- and R ...
... especially ethylene glycol. The principal shortcoming of all homogeneous CO hydrogenation reactions is their low catalytic activity, which results in the need to use high catalyst loadings and drastic reaction conditions (usually at temperatures above 200°C and pressures above 30MPa) [34]. Rh- and R ...
Epoxidation and oxidation reactions using 1,4
... being readily soluble in water, requires much simpler steps in the workup procedure when compared to [VO (acac)2]. Hence the use of BTEAH which is a phase transfer catalyst seems to suit the reaction, particularly with solid-phase reagents. The polymeric byproduct was identified to be polymeric t-bu ...
... being readily soluble in water, requires much simpler steps in the workup procedure when compared to [VO (acac)2]. Hence the use of BTEAH which is a phase transfer catalyst seems to suit the reaction, particularly with solid-phase reagents. The polymeric byproduct was identified to be polymeric t-bu ...
A Model for Catalytically Active Zinc(I1) Ion in Liver
... that the most acidic and coordinatively least saturated Zn" in LIcatalytically generates zinc(11)-alkoxide complex to facilitate the hydride transfer to the hydride acceptor on the Zn" coordination sphere. The present study provides the first chemical model illustrating the significance of the Zn" a ...
... that the most acidic and coordinatively least saturated Zn" in LIcatalytically generates zinc(11)-alkoxide complex to facilitate the hydride transfer to the hydride acceptor on the Zn" coordination sphere. The present study provides the first chemical model illustrating the significance of the Zn" a ...
Supporting Information for Angew. Chem. Int. Ed. Z52444 © Wiley
... Unless stated otherwise, reactions were performed in oven-dried glassware, under an atmosphere of oxygen, using freshly distilled solvents. Although we have never experienced an accident, all reactions must be performed with appropriate caution in a fume hood due to the flammable nature of mixtures ...
... Unless stated otherwise, reactions were performed in oven-dried glassware, under an atmosphere of oxygen, using freshly distilled solvents. Although we have never experienced an accident, all reactions must be performed with appropriate caution in a fume hood due to the flammable nature of mixtures ...
Integration of chemical catalysis with extractive fermentation to
... are self-condensation of the alcohol through the corresponding aldehyde (Guerbet reaction)13 and oligomerization of acetone into diacetone alcohol, mesityl oxide, cyclohexenones and other products14. In particular, self-condensation of acetone affects the overall efficiency of the desired process by ...
... are self-condensation of the alcohol through the corresponding aldehyde (Guerbet reaction)13 and oligomerization of acetone into diacetone alcohol, mesityl oxide, cyclohexenones and other products14. In particular, self-condensation of acetone affects the overall efficiency of the desired process by ...
Practice Problem - HCC Southeast Commons
... into two pieces that are exact mirror images – If an object has a plane of symmetry it is necessarily the same as its mirror image – An achiral molecule is a molecule with a plane of symmetry and is the same as its mirror image ...
... into two pieces that are exact mirror images – If an object has a plane of symmetry it is necessarily the same as its mirror image – An achiral molecule is a molecule with a plane of symmetry and is the same as its mirror image ...
14_chapter 8
... exclusively obtained in 79% yield with no formation of benzoic acid. The benzylic bromides were oxidized with NaIO4, LiBr/H+ and benzoic acid was obtained as product in high yield (71-89%). In this case the reaction could not be stopped at the aldehyde stage itself. The use of stoichiometric amount ...
... exclusively obtained in 79% yield with no formation of benzoic acid. The benzylic bromides were oxidized with NaIO4, LiBr/H+ and benzoic acid was obtained as product in high yield (71-89%). In this case the reaction could not be stopped at the aldehyde stage itself. The use of stoichiometric amount ...
Fluorine notes- EFFECT OF POLYFLUOROALKYL GROUPS IN
... Reaction of polyfluoroalkylchlorosulphites with cinnamic alcohol had been carried in two stages. During first stage reagents (cinnamic alcohol, potassium carbonate and polyfluoroalkylchlorosulphite) were mixed in chloroform at –10÷–5 °C, while there was the appearance of color–the solution became or ...
... Reaction of polyfluoroalkylchlorosulphites with cinnamic alcohol had been carried in two stages. During first stage reagents (cinnamic alcohol, potassium carbonate and polyfluoroalkylchlorosulphite) were mixed in chloroform at –10÷–5 °C, while there was the appearance of color–the solution became or ...
Alcohols, Aldehydes, and Ketones
... and propanol. If the –OH group can be attached at more than one position, then its position is denoted by a number, as in ...
... and propanol. If the –OH group can be attached at more than one position, then its position is denoted by a number, as in ...
Chapter 8 - profpaz.com
... Stoichiometry is the quantitative relationship between the reactants and products in a balanced chemical equation. Stoichiometry allows chemists to predict how much of a reactant is necessary to form a given amount of product or how much of a reactant is required to completely react with another rea ...
... Stoichiometry is the quantitative relationship between the reactants and products in a balanced chemical equation. Stoichiometry allows chemists to predict how much of a reactant is necessary to form a given amount of product or how much of a reactant is required to completely react with another rea ...
Alcohols and Carbonyls
... Primary alcohols can be oxidised by a number of oxidising agents, in two stages, 1st Stage - Hydrogen is lost; 2nd Stage - oxygen is gained. When applied to carbon compounds, oxidation results in an increase in the oxygen to hydrogen ration. ...
... Primary alcohols can be oxidised by a number of oxidising agents, in two stages, 1st Stage - Hydrogen is lost; 2nd Stage - oxygen is gained. When applied to carbon compounds, oxidation results in an increase in the oxygen to hydrogen ration. ...
Organic Chemistry I Laboratory
... tendency to lose the hydrogens they have, but because 2-butene is more stable than 1-butene. Although generalizations like Zaitzev’s rule can help us predict the products of many organic reactions, organic chemistry remains an empirical science –we cannot be certain that a rule that is valid for one ...
... tendency to lose the hydrogens they have, but because 2-butene is more stable than 1-butene. Although generalizations like Zaitzev’s rule can help us predict the products of many organic reactions, organic chemistry remains an empirical science –we cannot be certain that a rule that is valid for one ...
New process of low-temperature methanol synthesis from CO/CO2
... defined to be the sum of the yields of both liquid products. Table 1 lists the reaction results from using 13 kinds of alcohols as solvent separately under the same reaction conditions. For comparison, the results in the cases of cyclohexane as solvent or ethanol as solvent but no catalyst are also ...
... defined to be the sum of the yields of both liquid products. Table 1 lists the reaction results from using 13 kinds of alcohols as solvent separately under the same reaction conditions. For comparison, the results in the cases of cyclohexane as solvent or ethanol as solvent but no catalyst are also ...
Copper(II) bromide as efficient catalyst for silyl
... achieved in good to high yields with CuBr2 as catalyst at room temperature. With very mild conditions, with a wide tolerance to other protecting groups, this new interconversion of silyl to diarylmethyl protecting groups will find applications in organic synthesis, especially in the total synthesis o ...
... achieved in good to high yields with CuBr2 as catalyst at room temperature. With very mild conditions, with a wide tolerance to other protecting groups, this new interconversion of silyl to diarylmethyl protecting groups will find applications in organic synthesis, especially in the total synthesis o ...
Chemistry Name Mr. Reger Review Guide – Ch. 9
... b) How much of the excess reagent is left after the reaction is complete? c) How many moles of NaCl are produced? d) What mass of NaCl is produced? e) What is the % yield if actual amount obtained from the reaction is 6.8g NaCl? ...
... b) How much of the excess reagent is left after the reaction is complete? c) How many moles of NaCl are produced? d) What mass of NaCl is produced? e) What is the % yield if actual amount obtained from the reaction is 6.8g NaCl? ...
69. A general approach to the enantioselective -oxidation of aldehydes via synergistic catalysis
... process (entries 1–2, 5, 7, and 13–14). Notably, significant variation in steric environment on the aldehyde component is readily accommodated, with the more demanding frameworks furnishing the highest levels of asymmetric induction (entries 3– 4, 6, and 13–14). Finally, subjection of (S)- and (R)-c ...
... process (entries 1–2, 5, 7, and 13–14). Notably, significant variation in steric environment on the aldehyde component is readily accommodated, with the more demanding frameworks furnishing the highest levels of asymmetric induction (entries 3– 4, 6, and 13–14). Finally, subjection of (S)- and (R)-c ...
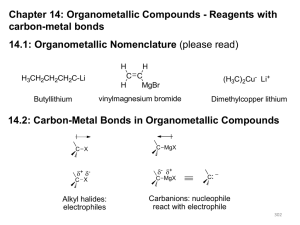
Organometallic Compounds - Reagents
... “it is a problem solving technique for transforming the structure of a synthetic target molecule (TM) to a sequence of progressively simpler structures along the pathway which ultimately leads to simple or commercially available starting materials for a chemical synthesis.” The transformation of a m ...
... “it is a problem solving technique for transforming the structure of a synthetic target molecule (TM) to a sequence of progressively simpler structures along the pathway which ultimately leads to simple or commercially available starting materials for a chemical synthesis.” The transformation of a m ...
Online edition for students of organic chemistry lab
... resulting solution treated with drying agent and heated to evaporate off the methylene chloride. High purity cyclohexanone could then be obtained by distilling the product, a method which would also help to identify the product by giving the boiling point. You will not have time to distill the produ ...
... resulting solution treated with drying agent and heated to evaporate off the methylene chloride. High purity cyclohexanone could then be obtained by distilling the product, a method which would also help to identify the product by giving the boiling point. You will not have time to distill the produ ...
Grignard Reagents
... “it is a problem solving technique for transforming the structure of a synthetic target molecule (TM) to a sequence of progressively simpler structures along the pathway which ultimately leads to simple or commercially available starting materials for a chemical synthesis.” The transformation of a m ...
... “it is a problem solving technique for transforming the structure of a synthetic target molecule (TM) to a sequence of progressively simpler structures along the pathway which ultimately leads to simple or commercially available starting materials for a chemical synthesis.” The transformation of a m ...
CYCLOALKANES, POLYMERS, ALCOHOLS AND ETHERS
... Ethylene oxide when treated with Grignard reagent yields : a) Tertiary alcohol b) Cyclopropyl alcohol c) Primary alcohol d) Secondary alcohol ...
... Ethylene oxide when treated with Grignard reagent yields : a) Tertiary alcohol b) Cyclopropyl alcohol c) Primary alcohol d) Secondary alcohol ...
Copper perchlorate: Efficient acetylation catalyst
... M(ClO4 )2 ·6H2 O where M is Mn, Co, Ni, Cu and Zn as efficient catalysts for acetylation of heteroatoms (Scheme 1) and diacetylation of aldehydes (Scheme 2) under solvent free conditions at room temperature. The catalysts are found to be more efficient compared to the other metal perchlorates and tr ...
... M(ClO4 )2 ·6H2 O where M is Mn, Co, Ni, Cu and Zn as efficient catalysts for acetylation of heteroatoms (Scheme 1) and diacetylation of aldehydes (Scheme 2) under solvent free conditions at room temperature. The catalysts are found to be more efficient compared to the other metal perchlorates and tr ...
Kinetic resolution

In organic chemistry, kinetic resolution is a means of differentiating two enantiomers in a racemic mixture. In kinetic resolution, two enantiomers react with different reaction rates in a chemical reaction with a chiral catalyst or reagent, resulting in an enantioenriched sample of the less reactive enantiomer. As opposed to chiral resolution, kinetic resolution does not rely on different physical properties of diastereomeric products, but rather on the different chemical properties of the racemic starting materials. This enantiomeric excess (ee) of the unreacted starting material continually rises as more product is formed, reaching 100% just before full completion of the reaction. Kinetic resolution relies upon differences in reactivity between enantiomers or enantiomeric complexes. Kinetic resolution is a concept in organic chemistry and can be used for the preparation of chiral molecules in organic synthesis. Kinetic resolution reactions utilizing purely synthetic reagents and catalysts are much less common than the use of enzymatic kinetic resolution in application towards organic synthesis, although a number of useful synthetic techniques have been developed in the past 30 years.