Chapter - FIU Faculty Websites
... • Like gem-diol formation, the synthesis of acetals is reversible, and often, the equilibrium favors the reactants. • In acetal synthesis, since water is formed as a by-product, the equilibrium can be driven to the right by removing H2O as it is formed using distillation or other techniques. Please ...
... • Like gem-diol formation, the synthesis of acetals is reversible, and often, the equilibrium favors the reactants. • In acetal synthesis, since water is formed as a by-product, the equilibrium can be driven to the right by removing H2O as it is formed using distillation or other techniques. Please ...
Carbonyl Compounds I. Aldehydes and Ketones
... catalysts; the adducts may be formed reversibly and with relatively unfavorable equilibrium constants. Also, the initial adducts may be unstable and react further by elimination. (We recommend that you work Exercise 16-9 to see examples of these points, or review Section 15-4E.) T o organize this ve ...
... catalysts; the adducts may be formed reversibly and with relatively unfavorable equilibrium constants. Also, the initial adducts may be unstable and react further by elimination. (We recommend that you work Exercise 16-9 to see examples of these points, or review Section 15-4E.) T o organize this ve ...
Chemistry Notes for class 12 Chapter 12 Aldehydes, Ketones and
... (vi) Cross aldol condensation Base catalysed crossed aldol condensation between an aromatic aldehyde and an aliphatic aldehyde or ketone is called Claisen-Schmidt condensation or Claisen reaction. ...
... (vi) Cross aldol condensation Base catalysed crossed aldol condensation between an aromatic aldehyde and an aliphatic aldehyde or ketone is called Claisen-Schmidt condensation or Claisen reaction. ...
Conjugate addition_Clayden
... The reason that α,β-unsaturated carbonyl compounds react differently is conjugation, the phenomenon we discussed in Chapter 7. There we introduced you to the idea that bringing two π systems (two C=C bonds, for example, or a C=C bond and a C=O bond) close together leads to a stabilizing interaction. ...
... The reason that α,β-unsaturated carbonyl compounds react differently is conjugation, the phenomenon we discussed in Chapter 7. There we introduced you to the idea that bringing two π systems (two C=C bonds, for example, or a C=C bond and a C=O bond) close together leads to a stabilizing interaction. ...
Elimination Reactions
... base (H2SO4, H3PO4). To favour SN1 over E1, use a good nucleophile. •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that ...
... base (H2SO4, H3PO4). To favour SN1 over E1, use a good nucleophile. •To favour E2 over SN2, use a strong, bulky non-nucleophilic base. To favour SN2 over E2, use good nucleophiles that are relatively weak bases. •It is important to keep in mind that although you might choose reaction conditions that ...
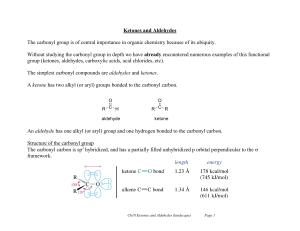
Compounds Containing a C=O (Carbonyl) Group
... reaction at the α-carbon. A C—H bond on the α carbon to a carbonyl group is more acidic than many other C—H bonds, because reaction with base forms a resonance-stabilized enolate anion. ...
... reaction at the α-carbon. A C—H bond on the α carbon to a carbonyl group is more acidic than many other C—H bonds, because reaction with base forms a resonance-stabilized enolate anion. ...
Alcohols I Reading: Wade chapter 10, sections 10-1- 10
... For the chlorinated derivatives of enthanol (ClCH2CH2OH, Cl3CCH2OH), the inductive electron-withdrawing effect of the local halogens enhances the acidity of the parent alcohols; this effect is additive, with trichloroethanol being more acidic than chloroethanol and ethanol. Phenol (an aromatic ring ...
... For the chlorinated derivatives of enthanol (ClCH2CH2OH, Cl3CCH2OH), the inductive electron-withdrawing effect of the local halogens enhances the acidity of the parent alcohols; this effect is additive, with trichloroethanol being more acidic than chloroethanol and ethanol. Phenol (an aromatic ring ...
pdf
... Importance of enan)omerically pure drugs • Different enan)omers can have different effects on the body • Ac)ons of “Inac)ve Isomers” 1. One isomer possesses therapeu)c ac)on while the other contributes to ...
... Importance of enan)omerically pure drugs • Different enan)omers can have different effects on the body • Ac)ons of “Inac)ve Isomers” 1. One isomer possesses therapeu)c ac)on while the other contributes to ...
Organocatalysed asymmetric Mannich reactions
... organocatalytic versions of the well-known Mannich reaction in particular.2 A key element in Mannich reactions is an iminium intermediate 2, which is susceptible to nucleophilic attack by a variety of nucleophiles such as enolised ketones (1) or equivalents thereof, resulting in carbon–carbon bond f ...
... organocatalytic versions of the well-known Mannich reaction in particular.2 A key element in Mannich reactions is an iminium intermediate 2, which is susceptible to nucleophilic attack by a variety of nucleophiles such as enolised ketones (1) or equivalents thereof, resulting in carbon–carbon bond f ...
Aldol reaction
The aldol reaction is a means of forming carbon–carbon bonds in organic chemistry.Discovered independently by Charles-Adolphe Wurtz and Alexander Borodin in 1872, the reaction combines two carbonyl compounds (the original experiments used aldehydes) to form a new β-hydroxy carbonyl compound. These products are known as aldols, from the aldehyde + alcohol, a structural motif seen in many of the products. Aldol structural units are found in many important molecules, whether naturally occurring or synthetic.For example, the aldol reaction has been used in the large-scale production of the commodity chemical pentaerythritoland the synthesis of the heart disease drug Lipitor (atorvastatin, calcium salt).The aldol reaction unites two relatively simple molecules into a more complex one. Increased complexity arises because up to two new stereogenic centers (on the α- and β-carbon of the aldol adduct, marked with asterisks in the scheme below) are formed. Modern methodology is capable of not only allowing aldol reactions to proceed in high yield but also controlling both the relative and absolute stereochemical configuration of these stereocenters. This ability to selectively synthesize a particular stereoisomer is significant because different stereoisomers can have very different chemical and biological properties.For example, stereogenic aldol units are especially common in polyketides, a class of molecules found in biological organisms. In nature, polyketides are synthesized by enzymes that effect iterative Claisen condensations. The 1,3-dicarbonyl products of these reactions can then be variously derivatized to produce a wide variety of interesting structures. Often, such derivitization involves the reduction of one of the carbonyl groups, producing the aldol subunit. Some of these structures have potent biological properties: the immunosuppressant FK506, the anti-tumor agent discodermolide, or the antifungal agent amphotericin B, for example. Although the synthesis of many such compounds was once considered nearly impossible, aldol methodology has allowed their efficient synthesis in many cases.A typical modern aldol addition reaction, shown above, might involve the nucleophilic addition of a ketone enolate to an aldehyde. Once formed, the aldol product can sometimes lose a molecule of water to form an α,β-unsaturated carbonyl compound. This is called aldol condensation. A variety of nucleophiles may be employed in the aldol reaction, including the enols, enolates, and enol ethers of ketones, aldehydes, and many other carbonyl compounds. The electrophilic partner is usually an aldehyde or ketone (many variations, such as the Mannich reaction, exist). When the nucleophile and electrophile are different, the reaction is called a crossed aldol reaction; on the converse, when the nucleophile and electrophile are the same, the reaction is called an aldol dimerization.