Ir-catalysed formation of C− F bonds. From allylic alcohols to α
... mixture of 5 : 1. Less water failed to dissolve SelectF, and more water increased by-product (3a) formation. The catalyst loading could be lowered to 1 mol% (Table 2, entry 1). At acidic pH, higher amounts of unwanted ketone 3a are obtained and the reaction is complete in a shorter time (15 min) (Ta ...
... mixture of 5 : 1. Less water failed to dissolve SelectF, and more water increased by-product (3a) formation. The catalyst loading could be lowered to 1 mol% (Table 2, entry 1). At acidic pH, higher amounts of unwanted ketone 3a are obtained and the reaction is complete in a shorter time (15 min) (Ta ...
New Phenylglycine-Derived Primary Amine Organocatalysts for the
... provided in the Experimental Section) (Figure 4). The results in Table 3 provide interesting insights. First, by comparing catalysts 18, 19, 15 and 20 (in the standard system of THF with 1 equiv. CH3CO2H), we found the enantioselectivity to increase from 33 % ee for the simple α-methylbenzylamine (1 ...
... provided in the Experimental Section) (Figure 4). The results in Table 3 provide interesting insights. First, by comparing catalysts 18, 19, 15 and 20 (in the standard system of THF with 1 equiv. CH3CO2H), we found the enantioselectivity to increase from 33 % ee for the simple α-methylbenzylamine (1 ...
Microsoft Word
... widely used as a chiral auxiliary in numerous asymmetric transformations. The efficacy of the sulfoxide group in the stereoselective auxiliary-induced reactions is mainly due to the steric and stereoelectronic differences between the substituents of the chiral sulfur atom: a lone pair of electrons, ...
... widely used as a chiral auxiliary in numerous asymmetric transformations. The efficacy of the sulfoxide group in the stereoselective auxiliary-induced reactions is mainly due to the steric and stereoelectronic differences between the substituents of the chiral sulfur atom: a lone pair of electrons, ...
Unit-8-Alcohols-Aldehydes-Ketones
... Unit 8 - Organic Molecules III Alcohols, Thiols, Ethers, Aldehydes and Ketones In this unit we continue surveying some of the families of organic molecules that play important roles in biochemistry; looking both at their physical and chemical properties. The Group VIA elements, oxygen and sulfur, ty ...
... Unit 8 - Organic Molecules III Alcohols, Thiols, Ethers, Aldehydes and Ketones In this unit we continue surveying some of the families of organic molecules that play important roles in biochemistry; looking both at their physical and chemical properties. The Group VIA elements, oxygen and sulfur, ty ...
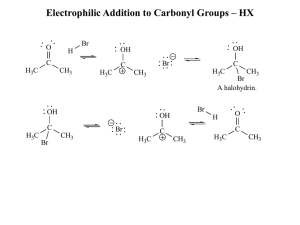
Electrophilic Addition to Carbonyl Groups – HX
... 2) Do the desired transformation (on another part of the molecule) that would have been incompatible with the unprotected functional group. 3) Remove the “protecting group” to recover the initial functional group (in this case, addition of aqueous acid reverts the equilibrium to reactants, regenerat ...
... 2) Do the desired transformation (on another part of the molecule) that would have been incompatible with the unprotected functional group. 3) Remove the “protecting group” to recover the initial functional group (in this case, addition of aqueous acid reverts the equilibrium to reactants, regenerat ...
Chem 314 Preorganic Evaluation
... possible E2 products a completely substituted Cβ makes E2 impossible from that position, but if other Cβ's are present with a hydrogen present, then E2 can occur from those atoms vinyl & phenyl are fairly unreactive, but with really stong bases (R2N ) E2 can form an alkyne, or if the alkyne pi bond ...
... possible E2 products a completely substituted Cβ makes E2 impossible from that position, but if other Cβ's are present with a hydrogen present, then E2 can occur from those atoms vinyl & phenyl are fairly unreactive, but with really stong bases (R2N ) E2 can form an alkyne, or if the alkyne pi bond ...
Disproportionation of Monolithium Acetylide into
... most widely used ethynylation and alkynylation reaction.1 Monolithium acetylide (1) disproportionates readily above -25 °C into the more stable dilithium carbide (2) and acetylene.2 Liquid ammonia which is utilized in processes claimed to have industrial economics1,3 serves not only as a solvent but ...
... most widely used ethynylation and alkynylation reaction.1 Monolithium acetylide (1) disproportionates readily above -25 °C into the more stable dilithium carbide (2) and acetylene.2 Liquid ammonia which is utilized in processes claimed to have industrial economics1,3 serves not only as a solvent but ...
OC 2/e Ch 15
... like alcohols, add to the C=O of aldehydes and ketones to give tetrahedral carbonyl addition products The sulfur atom of a thiol is a better nucleophile than the oxygen atom of an alcohol A common sulfur nucleophile used for this purpose is 1,3-propanedithiol • the product is a 1,3-dithiane O ...
... like alcohols, add to the C=O of aldehydes and ketones to give tetrahedral carbonyl addition products The sulfur atom of a thiol is a better nucleophile than the oxygen atom of an alcohol A common sulfur nucleophile used for this purpose is 1,3-propanedithiol • the product is a 1,3-dithiane O ...
Enantioselective Organocatalytic Aminomethylation of Aldehydes: A
... catalyst F and recrystallizing the HCl salt of the γ-amino alcohol gave a 72% yield of material with >98% ee. The benzyl groups were removed and replaced by Boc in an efficient one-pot operation. Jones oxidation12 then provided desired β2-amino acid product after simple extraction, with >50% overall ...
... catalyst F and recrystallizing the HCl salt of the γ-amino alcohol gave a 72% yield of material with >98% ee. The benzyl groups were removed and replaced by Boc in an efficient one-pot operation. Jones oxidation12 then provided desired β2-amino acid product after simple extraction, with >50% overall ...
Highly Enantioselective Cyclocarbonylation of Allylic
... The observed stereospecificity probably results from cis addition of the palladium hydride to the allylic CdC bond as proposed by Alper. A notable advance emerging from these studies is the asymmetric cyclocarbonylation of the six-membered ring allylicalcohol 3e using a Pd-BICP catalyst. At 80 °C, c ...
... The observed stereospecificity probably results from cis addition of the palladium hydride to the allylic CdC bond as proposed by Alper. A notable advance emerging from these studies is the asymmetric cyclocarbonylation of the six-membered ring allylicalcohol 3e using a Pd-BICP catalyst. At 80 °C, c ...
Aldehydes and Ketones - Belle Vernon Area School District
... Reduction of aldes and kets to alcohols O R-C-H + H2 –Pt/Pd R-C-OH RCHO + H2 –Pt/Pd RCH2OH O O R-C-R + H2 –Pt/Pd R-C-R RCOR + H2 –Pt/Pd RCHOHR ...
... Reduction of aldes and kets to alcohols O R-C-H + H2 –Pt/Pd R-C-OH RCHO + H2 –Pt/Pd RCH2OH O O R-C-R + H2 –Pt/Pd R-C-R RCOR + H2 –Pt/Pd RCHOHR ...
Aldehydes Ketones Carboxylic Acid
... nucleophilic addition reactions is due the positive charge on carbonyl carbon. Greater positive charge means greater reactivity. Electron releasing power of two alkyl groups in ketones is more than one in aldehyde. Therefore positive charge is reduced in ketones as compared to aldehydes. Thus ketone ...
... nucleophilic addition reactions is due the positive charge on carbonyl carbon. Greater positive charge means greater reactivity. Electron releasing power of two alkyl groups in ketones is more than one in aldehyde. Therefore positive charge is reduced in ketones as compared to aldehydes. Thus ketone ...
Aldol reaction
The aldol reaction is a means of forming carbon–carbon bonds in organic chemistry.Discovered independently by Charles-Adolphe Wurtz and Alexander Borodin in 1872, the reaction combines two carbonyl compounds (the original experiments used aldehydes) to form a new β-hydroxy carbonyl compound. These products are known as aldols, from the aldehyde + alcohol, a structural motif seen in many of the products. Aldol structural units are found in many important molecules, whether naturally occurring or synthetic.For example, the aldol reaction has been used in the large-scale production of the commodity chemical pentaerythritoland the synthesis of the heart disease drug Lipitor (atorvastatin, calcium salt).The aldol reaction unites two relatively simple molecules into a more complex one. Increased complexity arises because up to two new stereogenic centers (on the α- and β-carbon of the aldol adduct, marked with asterisks in the scheme below) are formed. Modern methodology is capable of not only allowing aldol reactions to proceed in high yield but also controlling both the relative and absolute stereochemical configuration of these stereocenters. This ability to selectively synthesize a particular stereoisomer is significant because different stereoisomers can have very different chemical and biological properties.For example, stereogenic aldol units are especially common in polyketides, a class of molecules found in biological organisms. In nature, polyketides are synthesized by enzymes that effect iterative Claisen condensations. The 1,3-dicarbonyl products of these reactions can then be variously derivatized to produce a wide variety of interesting structures. Often, such derivitization involves the reduction of one of the carbonyl groups, producing the aldol subunit. Some of these structures have potent biological properties: the immunosuppressant FK506, the anti-tumor agent discodermolide, or the antifungal agent amphotericin B, for example. Although the synthesis of many such compounds was once considered nearly impossible, aldol methodology has allowed their efficient synthesis in many cases.A typical modern aldol addition reaction, shown above, might involve the nucleophilic addition of a ketone enolate to an aldehyde. Once formed, the aldol product can sometimes lose a molecule of water to form an α,β-unsaturated carbonyl compound. This is called aldol condensation. A variety of nucleophiles may be employed in the aldol reaction, including the enols, enolates, and enol ethers of ketones, aldehydes, and many other carbonyl compounds. The electrophilic partner is usually an aldehyde or ketone (many variations, such as the Mannich reaction, exist). When the nucleophile and electrophile are different, the reaction is called a crossed aldol reaction; on the converse, when the nucleophile and electrophile are the same, the reaction is called an aldol dimerization.