ether - HCC Southeast Commons
... are prepared by the sulfuric acid-catalyzed dehydration procedure? What product(s) would you expect if ethanol and 1-propanol were allowed to react together? In what ratio would the products be formed if the two alcohols were of equal reactivity? ...
... are prepared by the sulfuric acid-catalyzed dehydration procedure? What product(s) would you expect if ethanol and 1-propanol were allowed to react together? In what ratio would the products be formed if the two alcohols were of equal reactivity? ...
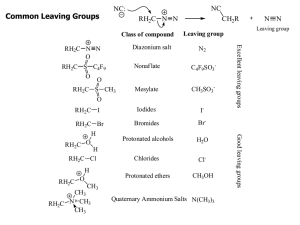
Common Leaving Groups
... Whenever substitution reactions are possible, we must also consider whether or not elimination reactions might occur under the same reaction conditions. In elimination reactions, a “neutral” molecule is „eliminated‟ from the substrate to form a π bond. The π bond is formed between the two carbon ato ...
... Whenever substitution reactions are possible, we must also consider whether or not elimination reactions might occur under the same reaction conditions. In elimination reactions, a “neutral” molecule is „eliminated‟ from the substrate to form a π bond. The π bond is formed between the two carbon ato ...
Alcohols
... Phenols and thiols are more acidic than alcohols • Both are soluble in dilute aqueous NaOH • Can often be separated from a mixture by basic extraction into aqueous solution, followed by reacidification Phenols are more acidic than alcohols because the phenoxide anion is resonance-stabilized • Deloca ...
... Phenols and thiols are more acidic than alcohols • Both are soluble in dilute aqueous NaOH • Can often be separated from a mixture by basic extraction into aqueous solution, followed by reacidification Phenols are more acidic than alcohols because the phenoxide anion is resonance-stabilized • Deloca ...
ALKANE ALKYL HALIDE Halogenation of Alkanes
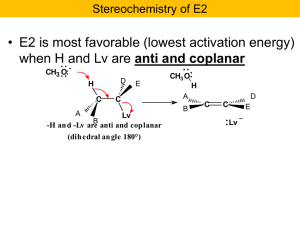
... E2 mechanism (write on back of card) only works with 2o and 3o alkyl halides with 2o alkyl halides, use (CH3)3COK to avoid SN2 reactions must have a strong base (usually alkoxide ) constitutional isomers may form if more than one ! hydrogen is available anti elimination: when both carbons are stereo ...
... E2 mechanism (write on back of card) only works with 2o and 3o alkyl halides with 2o alkyl halides, use (CH3)3COK to avoid SN2 reactions must have a strong base (usually alkoxide ) constitutional isomers may form if more than one ! hydrogen is available anti elimination: when both carbons are stereo ...
Alcohols and Thiols
... The alcoholic proton is also somewhat acidic. The pKa for alcohols runs between 15 and 18 - certainly more acidic than your average alkyne! The general trend in acidity is 1°>2°>3°, with t-butanol being about the least acidic alcohol you’ll find...this is why t-butoxide is used as a base in many of ...
... The alcoholic proton is also somewhat acidic. The pKa for alcohols runs between 15 and 18 - certainly more acidic than your average alkyne! The general trend in acidity is 1°>2°>3°, with t-butanol being about the least acidic alcohol you’ll find...this is why t-butoxide is used as a base in many of ...
- Wiley Online Library
... comparable to those obtained with 1 (front vs. middle row). The requirement for an aqueous solvent (Table 1) is therefore likely associated with chloride dissociation from 1. Still, water cannot be fully excluded from the solvent even with the chloride-free complex 8; in pure THF 8 afforded 3 a in o ...
... comparable to those obtained with 1 (front vs. middle row). The requirement for an aqueous solvent (Table 1) is therefore likely associated with chloride dissociation from 1. Still, water cannot be fully excluded from the solvent even with the chloride-free complex 8; in pure THF 8 afforded 3 a in o ...
19.7 Reversible Addition Reactions of Aldehydes and Ketones
... • Reactions by these and related reagents are referred to as hydride reductions • These reactions are further examples of nucleophilic addition ...
... • Reactions by these and related reagents are referred to as hydride reductions • These reactions are further examples of nucleophilic addition ...
Organic Chemistry Fifth Edition
... group. More substituted carbon (more positive charge although more sterically hindered) is attacked by a weak nucleophile. Very similar to opening of cyclic bromonium ion. Review that subject. Due to resonance, some positive charge is located on this carbon. ...
... group. More substituted carbon (more positive charge although more sterically hindered) is attacked by a weak nucleophile. Very similar to opening of cyclic bromonium ion. Review that subject. Due to resonance, some positive charge is located on this carbon. ...
Aldehydes And Ketones
... the flow of pairs of e• Draw the arrow from higher e- density to lower e- density i.e. from the nucleophile to the electrophile • Removing e- density from an atom will create a formal + charge • Adding e- density to an atom will create a formal - charge • Proton transfer is fast (kinetics) and usual ...
... the flow of pairs of e• Draw the arrow from higher e- density to lower e- density i.e. from the nucleophile to the electrophile • Removing e- density from an atom will create a formal + charge • Adding e- density to an atom will create a formal - charge • Proton transfer is fast (kinetics) and usual ...
Chapter 8 I. Nucleophilic Substitution
... L. Hase's group at Texas Tech University, provide direct evidence for this mechanism in the gas phase. However, they also detected an additional, unexpected mechanism. In this new pathway, called the roundabout mechanism, chloride bumps into the methyl group and spins the entire methyl iodide molecu ...
... L. Hase's group at Texas Tech University, provide direct evidence for this mechanism in the gas phase. However, they also detected an additional, unexpected mechanism. In this new pathway, called the roundabout mechanism, chloride bumps into the methyl group and spins the entire methyl iodide molecu ...
Mock Exam One
... a.) The bond angle around the oxygen of an alcohol is < 109.5°. b.) Alcohols contain a polar covalent bond that an alkane does not have. c.) The strongest intermolecular force present in an alcohol is hydrogen bonding while the strongest intermolecular force present in alkanes is London dispersion. ...
... a.) The bond angle around the oxygen of an alcohol is < 109.5°. b.) Alcohols contain a polar covalent bond that an alkane does not have. c.) The strongest intermolecular force present in an alcohol is hydrogen bonding while the strongest intermolecular force present in alkanes is London dispersion. ...
Aldol reaction
The aldol reaction is a means of forming carbon–carbon bonds in organic chemistry.Discovered independently by Charles-Adolphe Wurtz and Alexander Borodin in 1872, the reaction combines two carbonyl compounds (the original experiments used aldehydes) to form a new β-hydroxy carbonyl compound. These products are known as aldols, from the aldehyde + alcohol, a structural motif seen in many of the products. Aldol structural units are found in many important molecules, whether naturally occurring or synthetic.For example, the aldol reaction has been used in the large-scale production of the commodity chemical pentaerythritoland the synthesis of the heart disease drug Lipitor (atorvastatin, calcium salt).The aldol reaction unites two relatively simple molecules into a more complex one. Increased complexity arises because up to two new stereogenic centers (on the α- and β-carbon of the aldol adduct, marked with asterisks in the scheme below) are formed. Modern methodology is capable of not only allowing aldol reactions to proceed in high yield but also controlling both the relative and absolute stereochemical configuration of these stereocenters. This ability to selectively synthesize a particular stereoisomer is significant because different stereoisomers can have very different chemical and biological properties.For example, stereogenic aldol units are especially common in polyketides, a class of molecules found in biological organisms. In nature, polyketides are synthesized by enzymes that effect iterative Claisen condensations. The 1,3-dicarbonyl products of these reactions can then be variously derivatized to produce a wide variety of interesting structures. Often, such derivitization involves the reduction of one of the carbonyl groups, producing the aldol subunit. Some of these structures have potent biological properties: the immunosuppressant FK506, the anti-tumor agent discodermolide, or the antifungal agent amphotericin B, for example. Although the synthesis of many such compounds was once considered nearly impossible, aldol methodology has allowed their efficient synthesis in many cases.A typical modern aldol addition reaction, shown above, might involve the nucleophilic addition of a ketone enolate to an aldehyde. Once formed, the aldol product can sometimes lose a molecule of water to form an α,β-unsaturated carbonyl compound. This is called aldol condensation. A variety of nucleophiles may be employed in the aldol reaction, including the enols, enolates, and enol ethers of ketones, aldehydes, and many other carbonyl compounds. The electrophilic partner is usually an aldehyde or ketone (many variations, such as the Mannich reaction, exist). When the nucleophile and electrophile are different, the reaction is called a crossed aldol reaction; on the converse, when the nucleophile and electrophile are the same, the reaction is called an aldol dimerization.