PDF - Nanyang Technological University
... electron-deficient anilines were unsuitable substrates for the oxidative reactions and products 6 c and 6 d were not obtained (Scheme 1). With aliphatic cyclic amines, the size of the ring was found to affect the outcome of the reaction. The reaction of an N-aryl pyrrolidine with 2 a gave the desire ...
... electron-deficient anilines were unsuitable substrates for the oxidative reactions and products 6 c and 6 d were not obtained (Scheme 1). With aliphatic cyclic amines, the size of the ring was found to affect the outcome of the reaction. The reaction of an N-aryl pyrrolidine with 2 a gave the desire ...
Organic Chemistry/Fourth Edition: e-Text
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
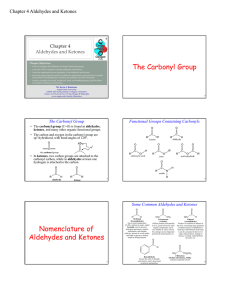
Aldehydes and Ketones The Carbonyl Group
... The Polarity of the Carbonyl Group • Since there is no hydrogen on the carbonyl oxygen, aldehydes and ketones do not form hydrogen bonds with themselves. • Aldehydes and ketones therefore have boiling points that are in between those of alcohols and hydrocarbons of the same molecular weight: – Alcoh ...
... The Polarity of the Carbonyl Group • Since there is no hydrogen on the carbonyl oxygen, aldehydes and ketones do not form hydrogen bonds with themselves. • Aldehydes and ketones therefore have boiling points that are in between those of alcohols and hydrocarbons of the same molecular weight: – Alcoh ...
The Carbonyl Group - Angelo State University
... • Since there is no hydrogen on the carbonyl oxygen, aldehydes and ketones do not form hydrogen bonds with themselves. • Aldehydes and ketones therefore have boiling points that are in between those of alcohols and hydrocarbons of the same molecular weight: – Alcohols form hydrogen bonds, and have h ...
... • Since there is no hydrogen on the carbonyl oxygen, aldehydes and ketones do not form hydrogen bonds with themselves. • Aldehydes and ketones therefore have boiling points that are in between those of alcohols and hydrocarbons of the same molecular weight: – Alcohols form hydrogen bonds, and have h ...
The Carbonyl Group Nomenclature of Aldehydes and Ketones
... 3-(para-Hydroxyphenyl)2-butanone Responsible for the odor of ripe raspberries. ...
... 3-(para-Hydroxyphenyl)2-butanone Responsible for the odor of ripe raspberries. ...
Get PDF - Wiley Online Library
... cyclization of keto-butenolide 3 to establish the central Bring and the adjacent C5 and C6 quaternary stereocenters in a single operation. Cyclization substrate 3 would be prepared through aldol coupling of butenolide 5 with aldehyde 4. Notably, a single C5 stereocenter would template for all remain ...
... cyclization of keto-butenolide 3 to establish the central Bring and the adjacent C5 and C6 quaternary stereocenters in a single operation. Cyclization substrate 3 would be prepared through aldol coupling of butenolide 5 with aldehyde 4. Notably, a single C5 stereocenter would template for all remain ...
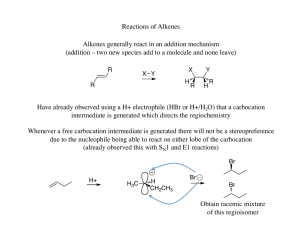
Alkenes 3 - ChemWeb (UCC)
... This reaction illustrated above is called a 1,2- or -elimination to indicate that the groups being eliminated are located on adjacent atoms in the starting material as compared to a 1,1- or -elimination where both are located on the same carbon atom This, in itself, tells you nothing about the act ...
... This reaction illustrated above is called a 1,2- or -elimination to indicate that the groups being eliminated are located on adjacent atoms in the starting material as compared to a 1,1- or -elimination where both are located on the same carbon atom This, in itself, tells you nothing about the act ...
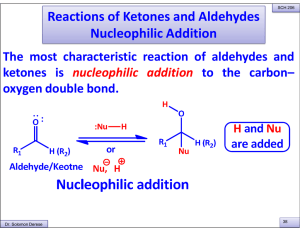
Reactions of Ketones and Aldehydes Nucleophilic Addition
... Five or six-membered cyclic hemiacetals are relatively strain free and thus form spontaneously from the corresponding hydroxy aldehydes in water. ...
... Five or six-membered cyclic hemiacetals are relatively strain free and thus form spontaneously from the corresponding hydroxy aldehydes in water. ...
Aldol reaction
The aldol reaction is a means of forming carbon–carbon bonds in organic chemistry.Discovered independently by Charles-Adolphe Wurtz and Alexander Borodin in 1872, the reaction combines two carbonyl compounds (the original experiments used aldehydes) to form a new β-hydroxy carbonyl compound. These products are known as aldols, from the aldehyde + alcohol, a structural motif seen in many of the products. Aldol structural units are found in many important molecules, whether naturally occurring or synthetic.For example, the aldol reaction has been used in the large-scale production of the commodity chemical pentaerythritoland the synthesis of the heart disease drug Lipitor (atorvastatin, calcium salt).The aldol reaction unites two relatively simple molecules into a more complex one. Increased complexity arises because up to two new stereogenic centers (on the α- and β-carbon of the aldol adduct, marked with asterisks in the scheme below) are formed. Modern methodology is capable of not only allowing aldol reactions to proceed in high yield but also controlling both the relative and absolute stereochemical configuration of these stereocenters. This ability to selectively synthesize a particular stereoisomer is significant because different stereoisomers can have very different chemical and biological properties.For example, stereogenic aldol units are especially common in polyketides, a class of molecules found in biological organisms. In nature, polyketides are synthesized by enzymes that effect iterative Claisen condensations. The 1,3-dicarbonyl products of these reactions can then be variously derivatized to produce a wide variety of interesting structures. Often, such derivitization involves the reduction of one of the carbonyl groups, producing the aldol subunit. Some of these structures have potent biological properties: the immunosuppressant FK506, the anti-tumor agent discodermolide, or the antifungal agent amphotericin B, for example. Although the synthesis of many such compounds was once considered nearly impossible, aldol methodology has allowed their efficient synthesis in many cases.A typical modern aldol addition reaction, shown above, might involve the nucleophilic addition of a ketone enolate to an aldehyde. Once formed, the aldol product can sometimes lose a molecule of water to form an α,β-unsaturated carbonyl compound. This is called aldol condensation. A variety of nucleophiles may be employed in the aldol reaction, including the enols, enolates, and enol ethers of ketones, aldehydes, and many other carbonyl compounds. The electrophilic partner is usually an aldehyde or ketone (many variations, such as the Mannich reaction, exist). When the nucleophile and electrophile are different, the reaction is called a crossed aldol reaction; on the converse, when the nucleophile and electrophile are the same, the reaction is called an aldol dimerization.