Unit 10 - Renton School District
... Identification of typical functional groups in molecules, e.g. phenyl, hydroxyl, carbonyl, carboxyl, carboxamide, aldehyde, ester, ether, amine, nitrile, alkyl, alkenyl, and alkynyl. Construction of 3D models (real or virtual) or organic molecules. Application of IUPAC rules in the nomenclature of s ...
... Identification of typical functional groups in molecules, e.g. phenyl, hydroxyl, carbonyl, carboxyl, carboxamide, aldehyde, ester, ether, amine, nitrile, alkyl, alkenyl, and alkynyl. Construction of 3D models (real or virtual) or organic molecules. Application of IUPAC rules in the nomenclature of s ...
Solutions
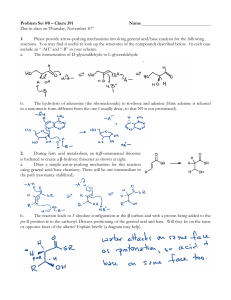
... in a tautomeric form different from the one I usually draw, so that N9 is not protonated). ...
... in a tautomeric form different from the one I usually draw, so that N9 is not protonated). ...
Aldehydes and Ketones
... Primary amine adds to C=O Proton is lost from N and adds to O to yield a neutral amino alcohol (carbinolamine) Protonation of OH converts into water as the leaving group Result is iminium ion, which loses proton Acid is required for loss of OH – too much acid blocks RNH2 ...
... Primary amine adds to C=O Proton is lost from N and adds to O to yield a neutral amino alcohol (carbinolamine) Protonation of OH converts into water as the leaving group Result is iminium ion, which loses proton Acid is required for loss of OH – too much acid blocks RNH2 ...
Carboxylic Acid Nomenclature
... 18.4: Acidity of Carboxylic Acids. The pKa of carboxylic acids typically ~ 5. They are significantly more acidic than water or alcohols. Bronsted Acidity (Ch. 1.14): Carboxylic acids transfer a proton to water to give H3O+ and carboxylate anions, RCO2 ...
... 18.4: Acidity of Carboxylic Acids. The pKa of carboxylic acids typically ~ 5. They are significantly more acidic than water or alcohols. Bronsted Acidity (Ch. 1.14): Carboxylic acids transfer a proton to water to give H3O+ and carboxylate anions, RCO2 ...
Aldehydes and Ketones
... Primary amine adds to C=O Proton is lost from N and adds to O to yield a neutral amino alcohol (carbinolamine) Protonation of OH converts into water as the leaving group Result is iminium ion, which loses proton Acid is required for loss of OH – too much acid blocks RNH2 ...
... Primary amine adds to C=O Proton is lost from N and adds to O to yield a neutral amino alcohol (carbinolamine) Protonation of OH converts into water as the leaving group Result is iminium ion, which loses proton Acid is required for loss of OH – too much acid blocks RNH2 ...
Lecture Resource ()
... The lone-pair electrons on nitrogen allows an amine to turn “inside out” rapidly at room temperature ...
... The lone-pair electrons on nitrogen allows an amine to turn “inside out” rapidly at room temperature ...
Sample Paper - Army Public School Jammu Cantt
... -amino acids have relatively higher melting points than the corresponding halo acids. Explain. ...
... -amino acids have relatively higher melting points than the corresponding halo acids. Explain. ...
PPT: Intro to Organic Chemistry
... C, H, and O: products are…CO2 and H2O Write the equation for the complete combustion of 2-methyl-2-pentene. C6H12 + 9 O2 6 CO2 + 6 H2O O Write the equation for the complete combustion of ethylbutanoate. O C6H12O2 + 8 O2 6 CO2 + 6 H2O ...
... C, H, and O: products are…CO2 and H2O Write the equation for the complete combustion of 2-methyl-2-pentene. C6H12 + 9 O2 6 CO2 + 6 H2O O Write the equation for the complete combustion of ethylbutanoate. O C6H12O2 + 8 O2 6 CO2 + 6 H2O ...
Reactive Materials - NC State University
... The reactivity of inorganic compounds may be frequently correlated with their “family” in the periodic table. Within a given family, similar types of behavior are observed with changes in the magnitude of reactivity varying consistently with atomic weight. The reactivity of chemical compounds shows ...
... The reactivity of inorganic compounds may be frequently correlated with their “family” in the periodic table. Within a given family, similar types of behavior are observed with changes in the magnitude of reactivity varying consistently with atomic weight. The reactivity of chemical compounds shows ...
Review Problems
... 3-ethyl-2-methylpentane 2,2-dichloropropane propylpental ether 2,2,3,3-tetramethylpentanal ...
... 3-ethyl-2-methylpentane 2,2-dichloropropane propylpental ether 2,2,3,3-tetramethylpentanal ...
Lab Report: Qualitative Organic Analysis
... 2. Write the products of the reaction that would occur when one of your unknowns reacts with the following acids and bases. Refer to the solubility classification flowchart and what you know about the reactions of acids and bases for help. Draw arrows indicating bond breakage and ...
... 2. Write the products of the reaction that would occur when one of your unknowns reacts with the following acids and bases. Refer to the solubility classification flowchart and what you know about the reactions of acids and bases for help. Draw arrows indicating bond breakage and ...
Summary Notes All that you need given 1 week left
... Chemical properties of alkenes -Reaction with bromine -Reaction with hydrogen -Reaction with itself (addition polymerisation) Alcohol is formed from the reaction between an alkene and steam. Catalyst : Phosphoric (V) acid Chemical properties of alcohol -Burn in air to form carbon dioxide and water - ...
... Chemical properties of alkenes -Reaction with bromine -Reaction with hydrogen -Reaction with itself (addition polymerisation) Alcohol is formed from the reaction between an alkene and steam. Catalyst : Phosphoric (V) acid Chemical properties of alcohol -Burn in air to form carbon dioxide and water - ...
Ethers, Sulfides, Epoxides
... Generally, the hemiacetals and acetals are only a minor component of an equilibrium mixture. In order to favor formation of acetals the carbonyl compound and alcohol is reacted with acid in the absence of water. Dry HCl) The acetals or hemiacetals maybe converted back to the carbonyl compound by tre ...
... Generally, the hemiacetals and acetals are only a minor component of an equilibrium mixture. In order to favor formation of acetals the carbonyl compound and alcohol is reacted with acid in the absence of water. Dry HCl) The acetals or hemiacetals maybe converted back to the carbonyl compound by tre ...
Dehydration notes-1
... Carbocation intermediate formation. First two steps of the mechanism at the same as for SN1. Carbocation will rearrange for increased stability, if possible. 5. Protons can be removed from any adjacent position leading to multiple products. 6. Major product is the most stable alkene from the most st ...
... Carbocation intermediate formation. First two steps of the mechanism at the same as for SN1. Carbocation will rearrange for increased stability, if possible. 5. Protons can be removed from any adjacent position leading to multiple products. 6. Major product is the most stable alkene from the most st ...
Carbonyl The carbonyl function, C=O, exists in a number of organic
... Reactions of RXC=O groups; Acid, ester, acid halide, anhydride, amide The characteristic reaction of this class of carbonyl compounds is the first step of addition of the nucleophile followed by elimination of the X function to give the final product. In acid catalysis the carbonyl is first protonat ...
... Reactions of RXC=O groups; Acid, ester, acid halide, anhydride, amide The characteristic reaction of this class of carbonyl compounds is the first step of addition of the nucleophile followed by elimination of the X function to give the final product. In acid catalysis the carbonyl is first protonat ...
C3 3.1-3.4 part 1 Alcohols, carboxlic acids and esters progress ticket
... Use the correct answer from the box to complete the word equation for the reaction. carbon ...
... Use the correct answer from the box to complete the word equation for the reaction. carbon ...
Erik`s Chemistry: Organic Chemistry Notes - ECHS Chemistry
... (6C's), heptane (7C's), octane (8C's), nonane(9C's), decane(10C's) Isomerization- same molecular formula, but different structure. Also creates different properties for the molecule. The number of possible isomers increases rapidly as the length of the chain increases. ...
... (6C's), heptane (7C's), octane (8C's), nonane(9C's), decane(10C's) Isomerization- same molecular formula, but different structure. Also creates different properties for the molecule. The number of possible isomers increases rapidly as the length of the chain increases. ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... b) How will you prepare isobutyl alcohol using a Grignard reagent? 15. An amide(A) having molecular formula C3H7ON on hydrolysis gives an acid C3H6O2(B) which upon chlorination in the presence of red phosphorus produces a chloroacid(C).The latter on boiling with aqueous NaOH and subsequent acidifica ...
... b) How will you prepare isobutyl alcohol using a Grignard reagent? 15. An amide(A) having molecular formula C3H7ON on hydrolysis gives an acid C3H6O2(B) which upon chlorination in the presence of red phosphorus produces a chloroacid(C).The latter on boiling with aqueous NaOH and subsequent acidifica ...
halogenoalkanes (haloalkanes)
... This form of nucleophilic substitution discussed so far is known as SN2; it is a bimolecular process. An alternative method involves the initial breaking of the C-X bond to form a carbocation, or carbonium ion, (a unimolecular process - SN1 mechanism), which is then attacked by the nucleophile. SN1 ...
... This form of nucleophilic substitution discussed so far is known as SN2; it is a bimolecular process. An alternative method involves the initial breaking of the C-X bond to form a carbocation, or carbonium ion, (a unimolecular process - SN1 mechanism), which is then attacked by the nucleophile. SN1 ...
Science 30 Chemistry
... The first global agreement to restrict CFCs came with the signing of the MONTREAL PROTOCOL in 1987 ultimately aiming to reduce them by half by the year 2000. The main CFCs will not be produced by any of the signing countries after the end of 1995, except for a limited amount for essential uses, suc ...
... The first global agreement to restrict CFCs came with the signing of the MONTREAL PROTOCOL in 1987 ultimately aiming to reduce them by half by the year 2000. The main CFCs will not be produced by any of the signing countries after the end of 1995, except for a limited amount for essential uses, suc ...
Review for SNC 2P Chemistry Unit(SPRING 2014)
... (a) An atom with more electrons than protons will be a positive ion. (b) A molecular compound is held together with ionic bonds. (c) The chloride ion is an example of a polyatomic ion. (d) The chemical test for hydrogen gas of to use a glowing splint. (e) A word equation contains words to represent ...
... (a) An atom with more electrons than protons will be a positive ion. (b) A molecular compound is held together with ionic bonds. (c) The chloride ion is an example of a polyatomic ion. (d) The chemical test for hydrogen gas of to use a glowing splint. (e) A word equation contains words to represent ...
Chem 226 — Problem Set #10
... Which is more likely, route Ê or route Ë ? Route Ê produces O-2, which is not a stable anion. Even if it were stable enough to form, it would have to be a very powerful nucleophile. Owing to this, route Ê would be reversible and the tetrahedral dianion on the left would be reformed. Route Ë , on the ...
... Which is more likely, route Ê or route Ë ? Route Ê produces O-2, which is not a stable anion. Even if it were stable enough to form, it would have to be a very powerful nucleophile. Owing to this, route Ê would be reversible and the tetrahedral dianion on the left would be reformed. Route Ë , on the ...
Honors Chemistry II Review 1. Express the following in scientific
... 15. A binary compound of zinc and sulfur contains 67.1% zinc by mass. What is the ratio of zinc and sulfur atoms in the compound? 16. Naturally occurring boron consists of two isotopes, 10B (19.9%), with an atomic mass of 10.0129, and 11B (80.1%) with an atomic mass of 11.00931. What is the atomic w ...
... 15. A binary compound of zinc and sulfur contains 67.1% zinc by mass. What is the ratio of zinc and sulfur atoms in the compound? 16. Naturally occurring boron consists of two isotopes, 10B (19.9%), with an atomic mass of 10.0129, and 11B (80.1%) with an atomic mass of 11.00931. What is the atomic w ...
Nomenclature of Carboxylic Acids 1. Parent alkane + the suffix
... Carboxylic acids can be reduced to primary alcohols with LiAlH4 or with BH3 followed by work-up with aqueous acid. In the reduction with LiAlH4, an intermediate aldehyde is formed, which is rapidly reduced to give the primary alcohol. Reduction by H2 / Pd and by NaBH4 are uneffective! Selective redu ...
... Carboxylic acids can be reduced to primary alcohols with LiAlH4 or with BH3 followed by work-up with aqueous acid. In the reduction with LiAlH4, an intermediate aldehyde is formed, which is rapidly reduced to give the primary alcohol. Reduction by H2 / Pd and by NaBH4 are uneffective! Selective redu ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.