Chemistry 199 - Department of Chemistry | Oregon State University
... What is meant by a condensation reaction? Give an example. Water is lost during a condensation reaction. Examples include: the formation of an ester from an alcohol and a carboxylic acid, the formation of an amide from an amine and a carboxylic acid. ...
... What is meant by a condensation reaction? Give an example. Water is lost during a condensation reaction. Examples include: the formation of an ester from an alcohol and a carboxylic acid, the formation of an amide from an amine and a carboxylic acid. ...
Acids and Bases
... Most solutions of both acids and bases are clear and colourless. We need an indicator to tell them apart. An indicator is a chemical which changes colour as the concentration of H+ (aq) and OH- (aq) changes. Two common indicators are litmus and phenolpthalein ...
... Most solutions of both acids and bases are clear and colourless. We need an indicator to tell them apart. An indicator is a chemical which changes colour as the concentration of H+ (aq) and OH- (aq) changes. Two common indicators are litmus and phenolpthalein ...
Chapter 16 – Amines and Amides
... in the margin of p. 487 of your textbook.) A result is that they almost always exist as solids when pure (as a result of bonding to each other). They are also generally quite water-soluble. Unlike amines, amides show no measurable basicity. Two factors account for this. Most importantly, the lone pa ...
... in the margin of p. 487 of your textbook.) A result is that they almost always exist as solids when pure (as a result of bonding to each other). They are also generally quite water-soluble. Unlike amines, amides show no measurable basicity. Two factors account for this. Most importantly, the lone pa ...
The Preparation of Fragrant Esters
... CH3-COOH + HO-CH2CH3 CH3-COO-CH2CH3 + H2O The ester product of this reaction (CH3-COO-CH2CH3) is named ethyl acetate, indicating the acid and alcohol from which it is prepared. Esterification is an equilibrium reaction, which means that the reaction does not go to completion on its own. Frequently ...
... CH3-COOH + HO-CH2CH3 CH3-COO-CH2CH3 + H2O The ester product of this reaction (CH3-COO-CH2CH3) is named ethyl acetate, indicating the acid and alcohol from which it is prepared. Esterification is an equilibrium reaction, which means that the reaction does not go to completion on its own. Frequently ...
CfE Higher Chemistry Unit 2: Nature‟s Chemistry Esters, Fats and
... (c) Give one reason why oils can be a useful part of a balanced diet. (1) ...
... (c) Give one reason why oils can be a useful part of a balanced diet. (1) ...
Organic_2_7ed_07th_module_carboxylic_acids 370KB May 03
... If the compound is insoluble in water, go on to the next solubility test. Solubility in 5% NaOH If the compound is soluble in 5% NaOH but insoluble in water, it is acidic, either a phenol (ArOH) or a carboxylic acid (RCO2H). This is reaction solubility, not like-dissolves-like solubility; the alkali ...
... If the compound is insoluble in water, go on to the next solubility test. Solubility in 5% NaOH If the compound is soluble in 5% NaOH but insoluble in water, it is acidic, either a phenol (ArOH) or a carboxylic acid (RCO2H). This is reaction solubility, not like-dissolves-like solubility; the alkali ...
Functional Groups
... Atoms other than hydrogen or carbon covalently bonded to a carbon atom in an organic molecule. Most commonly oxygen, nitrogen, or the halogens. The presence of a functional group drastically changes the chemical properties of a molecule. ...
... Atoms other than hydrogen or carbon covalently bonded to a carbon atom in an organic molecule. Most commonly oxygen, nitrogen, or the halogens. The presence of a functional group drastically changes the chemical properties of a molecule. ...
Chapter 15 Multistep Syntheses
... • Grouping Chemical Reactions • Retrosynthetic Analysis • Reactions Requiring Both Functional-Group Transformation and Skeletal Construction • Extending the Retrosynthetic Approach: Alternative Routes ...
... • Grouping Chemical Reactions • Retrosynthetic Analysis • Reactions Requiring Both Functional-Group Transformation and Skeletal Construction • Extending the Retrosynthetic Approach: Alternative Routes ...
Lab 6
... Acid anhydrides or simply anhydrides are acid derivatives that contain two acyl group joined by an oxygen atom. When carboxylic acids are pyrolyzed (i.e., heated strongly), they produce acid anhydrides by a condensation reaction (overall loss of water from two molecules). Figure 4 shows the equation ...
... Acid anhydrides or simply anhydrides are acid derivatives that contain two acyl group joined by an oxygen atom. When carboxylic acids are pyrolyzed (i.e., heated strongly), they produce acid anhydrides by a condensation reaction (overall loss of water from two molecules). Figure 4 shows the equation ...
Erythro and Threo
... • React the sugar with alcohol in acid. • Since the open chain sugar is in equilibrium with its - and -hemiacetal, both anomers of the acetal are formed. • Aglycone is the term used for the group bonded to the anomeric carbon. ...
... • React the sugar with alcohol in acid. • Since the open chain sugar is in equilibrium with its - and -hemiacetal, both anomers of the acetal are formed. • Aglycone is the term used for the group bonded to the anomeric carbon. ...
ethanoic acid
... fertilisers requires energy from fuels … these are not carbon neutral processes ...
... fertilisers requires energy from fuels … these are not carbon neutral processes ...
review sheet plus practice problems
... Use the Williamson ether synthesis (SN2 reaction of RO-) to prepare an unsymmetrical ether. Given the NMR and molecular formula, what is the structure of this alcohol, ether, or alkyl halide? Preparation of Alkyl Halides: Free-radical halogenation of an alkane with uv light. SOCl2 and PBr3 (R-OH to ...
... Use the Williamson ether synthesis (SN2 reaction of RO-) to prepare an unsymmetrical ether. Given the NMR and molecular formula, what is the structure of this alcohol, ether, or alkyl halide? Preparation of Alkyl Halides: Free-radical halogenation of an alkane with uv light. SOCl2 and PBr3 (R-OH to ...
Organic Chemistry
... Mechanism of Reaction with Base/H2O – Esters • Step 1: Attack of hydroxide ion (a nucleophile) on the carbonyl carbon (an electrophile). (Addition) • Step 2: Collapse of the TCAI. (Elimination) • Step 3: Proton transfer to the alkoxide ion; this step is irreversible and drives saponification to com ...
... Mechanism of Reaction with Base/H2O – Esters • Step 1: Attack of hydroxide ion (a nucleophile) on the carbonyl carbon (an electrophile). (Addition) • Step 2: Collapse of the TCAI. (Elimination) • Step 3: Proton transfer to the alkoxide ion; this step is irreversible and drives saponification to com ...
the original file
... 22. how to find the structure from the NMR and IR and molecular formula (all 3 will be given). 23. how to figure out homotopic, heterotopic, enantiotopic and diastereotopic pairs of protons in a molecule. 24. basic principle of electron impact and electrospray mass spec. 25. mechanism for reaction ...
... 22. how to find the structure from the NMR and IR and molecular formula (all 3 will be given). 23. how to figure out homotopic, heterotopic, enantiotopic and diastereotopic pairs of protons in a molecule. 24. basic principle of electron impact and electrospray mass spec. 25. mechanism for reaction ...
Chapter 18 - Aldehydes and Ketones
... It is also possible to obtain ketones in this reaction simply by performing a second alkylation prior to the hydrolysis of the substituted dithiane. ...
... It is also possible to obtain ketones in this reaction simply by performing a second alkylation prior to the hydrolysis of the substituted dithiane. ...
Chapter 9. CARBOXYLIC ACIDS AND THEIR DERIVATIVES
... monocarboxylic acids (including butyric acid) and the good solubility of the first four solid dicarboxylic acids (including glutaric acid). Acid strength. Most monocarboxylic acids of an aliphatic or aromatic series are acids of moderate strength with pKa values in the range from 4 to 5 (Table 9.1). ...
... monocarboxylic acids (including butyric acid) and the good solubility of the first four solid dicarboxylic acids (including glutaric acid). Acid strength. Most monocarboxylic acids of an aliphatic or aromatic series are acids of moderate strength with pKa values in the range from 4 to 5 (Table 9.1). ...
organic revision nots
... Although amino group is o– and p– directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline. 9. Direct nitration of aniline is not carried out. Explain why? 10. NH2 group of aniline acetylated is before carrying out nitration? 11. A ...
... Although amino group is o– and p– directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline. 9. Direct nitration of aniline is not carried out. Explain why? 10. NH2 group of aniline acetylated is before carrying out nitration? 11. A ...
chemistry ch4 - The Student Room
... In this reaction ammonia reacts as a nucleophile. State what is meant by the term nucleophile. ...
... In this reaction ammonia reacts as a nucleophile. State what is meant by the term nucleophile. ...
snc 2do unit: chemistry unit test review questions
... B) potassium + oxygen ------> _________________ C) magnesium carbonate -----> magnesium oxide + carbon dioxide D) ___________ + ________________ iron (III) hydroxide + potassium nitrate 6. Consider a solution with a pH of 3 and a solution with a pH of 5. Which is more acidic? How much more acidic ...
... B) potassium + oxygen ------> _________________ C) magnesium carbonate -----> magnesium oxide + carbon dioxide D) ___________ + ________________ iron (III) hydroxide + potassium nitrate 6. Consider a solution with a pH of 3 and a solution with a pH of 5. Which is more acidic? How much more acidic ...
Fisher Esterification - OpenBU
... which has an apple fragrance/flavor. First, protonation of the carbonyl oxygen lowers the carbonyl LUMO (π*) sufficiently for addition of the weakly nucleophilic solvent, methanol. Within the tetrahedral intermediate, proton transfer occurs to generate water as a good leaving group. Reformation of t ...
... which has an apple fragrance/flavor. First, protonation of the carbonyl oxygen lowers the carbonyl LUMO (π*) sufficiently for addition of the weakly nucleophilic solvent, methanol. Within the tetrahedral intermediate, proton transfer occurs to generate water as a good leaving group. Reformation of t ...
Exam 3 Key - Chemistry
... 11. (3) The first step of the reaction between a ketone and a Grignard reagent is: a) initial protonation of the C=O oxygen by H+ b) attack of the C=O oxygen upon the Mg c) attack by R- upon the C=O oxygen d) attack by R- upon the C=O carbon e) protonation of the Grignard reagent 12. (4) One of the ...
... 11. (3) The first step of the reaction between a ketone and a Grignard reagent is: a) initial protonation of the C=O oxygen by H+ b) attack of the C=O oxygen upon the Mg c) attack by R- upon the C=O oxygen d) attack by R- upon the C=O carbon e) protonation of the Grignard reagent 12. (4) One of the ...
Chapter 20 reactions of carbonyls
... [1] Convert the OH group into another functional group that does not interfere with the desired reaction. • This new blocking group is called a protecting group, and the reaction that creates it is called “protection”. [2] Carry out the desired reaction. [3] Remove the protecting group. • This react ...
... [1] Convert the OH group into another functional group that does not interfere with the desired reaction. • This new blocking group is called a protecting group, and the reaction that creates it is called “protection”. [2] Carry out the desired reaction. [3] Remove the protecting group. • This react ...
Document
... The chemical shift of the -OH proton occurs over a large range (2.0 - 5.5 ppm). It chemical shift is dependent upon the sample concentration and temperature. This proton is often observed as a broad singlet (br s). Exchangable protons are often not to be observed at all. ...
... The chemical shift of the -OH proton occurs over a large range (2.0 - 5.5 ppm). It chemical shift is dependent upon the sample concentration and temperature. This proton is often observed as a broad singlet (br s). Exchangable protons are often not to be observed at all. ...
Nucleophilic acyl substitution

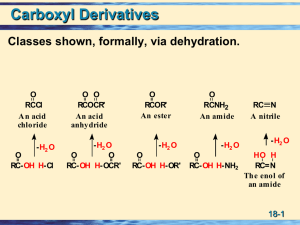
Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.