Proteins & Nucleic Acids - St. Mary Catholic Secondary School
... Tertiary shape is held together by R-group bonding within the chain and R-group interactions with the environment. Tertiary structure is also aided by prosthetic groups that are inorganic compounds that act as a central point for bonding within the protein. Quaternary structure occurs when a few ter ...
... Tertiary shape is held together by R-group bonding within the chain and R-group interactions with the environment. Tertiary structure is also aided by prosthetic groups that are inorganic compounds that act as a central point for bonding within the protein. Quaternary structure occurs when a few ter ...
Proteins & Nucleic Acids - St. Mary Catholic Secondary School
... Ladder shape – Rails - A series of alternating phosphates and sugars linked by covalent bonds known as phosphodiester bonds. Rungs of the ladder are made of the nitrogenous bases and their hydrogen bonds. The nitrogenous bases involved with DNA are adenine, cytosine, guanine and thymine. The adenine ...
... Ladder shape – Rails - A series of alternating phosphates and sugars linked by covalent bonds known as phosphodiester bonds. Rungs of the ladder are made of the nitrogenous bases and their hydrogen bonds. The nitrogenous bases involved with DNA are adenine, cytosine, guanine and thymine. The adenine ...
04-05 Biochem review sheet answers ws
... 18. List some of the many functions of proteins. transport, hormones, receptors, defense, etc. 19. How do amino acids differ? R group 20. What is a peptide bond? covalent bond between two amino acids 21. Discuss the connection between the term peptide and polypeptide. peptide is the monomer of a pro ...
... 18. List some of the many functions of proteins. transport, hormones, receptors, defense, etc. 19. How do amino acids differ? R group 20. What is a peptide bond? covalent bond between two amino acids 21. Discuss the connection between the term peptide and polypeptide. peptide is the monomer of a pro ...
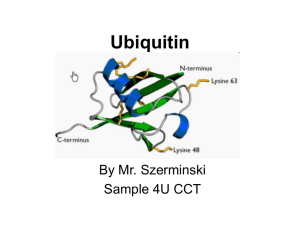
Ubiquitin
... eukaryotes - one of its functions: it directs protein recycling - can attach to proteins and label them for destruction. - discovery won the Nobel Prize for chemistry in 2004 ...
... eukaryotes - one of its functions: it directs protein recycling - can attach to proteins and label them for destruction. - discovery won the Nobel Prize for chemistry in 2004 ...
Practice Problems
... E. Peptide Bond 10. If a protein is amphipathic it is likely A. to be made of many amino acids. B. found in a membrane. C. to have quaternary structure. D. to be an enzyme. E. to be at or near it’s Vmax value. 11. Protein fill-in-the-blank: A. The “back” end of a protein is better called the _______ ...
... E. Peptide Bond 10. If a protein is amphipathic it is likely A. to be made of many amino acids. B. found in a membrane. C. to have quaternary structure. D. to be an enzyme. E. to be at or near it’s Vmax value. 11. Protein fill-in-the-blank: A. The “back” end of a protein is better called the _______ ...
BL 616 Test 1 study guide. The test will probably have 20 multiple
... Structures of major macromolecules – be able to draw generic nucleotide, fatty acid, phospholipid, triacylglycerol, glucose, and disaccharide, cAMP, Ch 6-7 amino acids, protein structure Be able to draw generic amino acid, peptide bond between two amino acids You will be provided with a diagram of t ...
... Structures of major macromolecules – be able to draw generic nucleotide, fatty acid, phospholipid, triacylglycerol, glucose, and disaccharide, cAMP, Ch 6-7 amino acids, protein structure Be able to draw generic amino acid, peptide bond between two amino acids You will be provided with a diagram of t ...
Nick Grishin "Evolutionary Classification of Protein Domains
... Homology is frequently obscured by sequence divergence, spatial structure changes and resemblance between unrelated 3D structures. We have developed a hierarchical evolutionary classification of all proteins with experimentally determined spatial structures. ECOD (Evolutionary Classification of prot ...
... Homology is frequently obscured by sequence divergence, spatial structure changes and resemblance between unrelated 3D structures. We have developed a hierarchical evolutionary classification of all proteins with experimentally determined spatial structures. ECOD (Evolutionary Classification of prot ...
Proteins and amino acids
... Amino acids – Hydrogen bonds The amino acids can make hydrogen bonds with their backbone, and some with their side chain A hydrogen bond needs a donor: donates a hydrogen (N-H or O-H) an acceptor: accepts the donated hydrogen (N or O) Example ...
... Amino acids – Hydrogen bonds The amino acids can make hydrogen bonds with their backbone, and some with their side chain A hydrogen bond needs a donor: donates a hydrogen (N-H or O-H) an acceptor: accepts the donated hydrogen (N or O) Example ...
A One- or Two-Day Course for Your Campus on
... Visual exploration of the 3D structures of macromolecules, such as proteins bound to ligands or nucleic acids. Where to find protein structures related to your research, how they are determined, how much of the genome is (and is not) known, and why. Teaching protein 3D structure, ligand interactions ...
... Visual exploration of the 3D structures of macromolecules, such as proteins bound to ligands or nucleic acids. Where to find protein structures related to your research, how they are determined, how much of the genome is (and is not) known, and why. Teaching protein 3D structure, ligand interactions ...
Amino acids and Protein Structure
... -SH more reactive than -OH. -SH pairs form disulfide bonds (aka bridges), key role stabilizing proteins ...
... -SH more reactive than -OH. -SH pairs form disulfide bonds (aka bridges), key role stabilizing proteins ...
Building Secondary Structures
... 2. Add the hydrogen bonds between an oxygen and a nitrogen. a. What effect does the addition of the hydrogen bonds have on the stability of these secondary structures? The structure is more stable with hydrogen bonds. b. Why does a hydrogen bond have to be between an oxygen and a nitrogen? A hydroge ...
... 2. Add the hydrogen bonds between an oxygen and a nitrogen. a. What effect does the addition of the hydrogen bonds have on the stability of these secondary structures? The structure is more stable with hydrogen bonds. b. Why does a hydrogen bond have to be between an oxygen and a nitrogen? A hydroge ...
Macromolecules Part 2
... B. Names usually end with the suffix “lin” (i.e. Insulin) for proteins and “ase” for enzymes (i.e. Sucrase) C. The monomer “building blocks” are called Amino Acids (There are 20 different Amino Acids that can be involved in making proteins. Proteins and enzymes usually have hundreds to thousands of ...
... B. Names usually end with the suffix “lin” (i.e. Insulin) for proteins and “ase” for enzymes (i.e. Sucrase) C. The monomer “building blocks” are called Amino Acids (There are 20 different Amino Acids that can be involved in making proteins. Proteins and enzymes usually have hundreds to thousands of ...
Complex Protein Structure
... a) three polypeptide chains held in a helix by hydrogen bonds b) found in collagen, the most abundant protein found in higher vertebrates (33 % of the body) collagen makes up skin, tendons, ligaments, bone connective tissue and the cornea of the eye. ...
... a) three polypeptide chains held in a helix by hydrogen bonds b) found in collagen, the most abundant protein found in higher vertebrates (33 % of the body) collagen makes up skin, tendons, ligaments, bone connective tissue and the cornea of the eye. ...
Cell Building Blocks
... aldehydes and ketones with many hyçjroxyl groups addcd, usually one on each carbon except the functional group. Other carbohydrates are composed of monosaccharide units and break down under hydrolysis. These may be classified as disaccharides, oligosaccharides, or polysaccharides, depending on wheth ...
... aldehydes and ketones with many hyçjroxyl groups addcd, usually one on each carbon except the functional group. Other carbohydrates are composed of monosaccharide units and break down under hydrolysis. These may be classified as disaccharides, oligosaccharides, or polysaccharides, depending on wheth ...
BNFO 602 Lecture 1 - New Jersey Institute of Technology
... • Since A (adenosine) always pairs with T (thymine) and C (cytosine) always pairs with G (guanine) knowing only one side of the ladder is enough • We represent DNA as a sequence of letters where each letter could be A,C,G, or T. • For example, for the helix shown here we would represent this as CAGT ...
... • Since A (adenosine) always pairs with T (thymine) and C (cytosine) always pairs with G (guanine) knowing only one side of the ladder is enough • We represent DNA as a sequence of letters where each letter could be A,C,G, or T. • For example, for the helix shown here we would represent this as CAGT ...
proteins——Echo,Jason,Philip
... Proteins are complex molecules made up of C,H,Obut they also contain N,S. ...
... Proteins are complex molecules made up of C,H,Obut they also contain N,S. ...
Protein structure prediction

Protein structure prediction is the prediction of the three-dimensional structure of a protein from its amino acid sequence — that is, the prediction of its folding and its secondary, tertiary, and quaternary structure from its primary structure. Structure prediction is fundamentally different from the inverse problem of protein design. Protein structure prediction is one of the most important goals pursued by bioinformatics and theoretical chemistry; it is highly important in medicine (for example, in drug design) and biotechnology (for example, in the design of novel enzymes). Every two years, the performance of current methods is assessed in the CASP experiment (Critical Assessment of Techniques for Protein Structure Prediction). A continuous evaluation of protein structure prediction web servers is performed by the community project CAMEO3D.