
doc 3.4.2 protein synthesis checklist
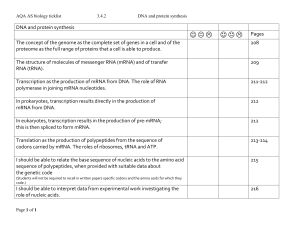
... Transcription as the production of mRNA from DNA. The role of RNA polymerase in joining mRNA nucleotides. ...
... Transcription as the production of mRNA from DNA. The role of RNA polymerase in joining mRNA nucleotides. ...
Biological Molecules
... The major classes of organic compounds are Proteins, Carbohydrates, Lipids and Nucleic acids. •All of these organic molecules always contain the elements Carbon (C), Hydrogen (H) and Oxygen (O). Proteins contain Nitrogen as well, and sometimes sulfur. Nucleic acids have C, H, O, N and phosphorus (P ...
... The major classes of organic compounds are Proteins, Carbohydrates, Lipids and Nucleic acids. •All of these organic molecules always contain the elements Carbon (C), Hydrogen (H) and Oxygen (O). Proteins contain Nitrogen as well, and sometimes sulfur. Nucleic acids have C, H, O, N and phosphorus (P ...
Essential Question: What is biochemistry
... Dehydration Synthesis-process by which a polymer is formed by the removal of water. Hydrolysis-process by which a polymer is broken into smaller units by the addition of water. ...
... Dehydration Synthesis-process by which a polymer is formed by the removal of water. Hydrolysis-process by which a polymer is broken into smaller units by the addition of water. ...
Cellular Respiration - Peoria Public Schools
... Humans and Anaerobic Respiration • Occurs in muscles during vigorous exercise: - if not enough O2 gets to the muscle then anaerobic resp. occurs - lactic acid builds up in muscles - once O2 is available lactic acid is converted back to pyruvic acid and aerobic resp. takes place ...
... Humans and Anaerobic Respiration • Occurs in muscles during vigorous exercise: - if not enough O2 gets to the muscle then anaerobic resp. occurs - lactic acid builds up in muscles - once O2 is available lactic acid is converted back to pyruvic acid and aerobic resp. takes place ...
Metabolism PPT File
... • Metabolism is the total of all the chemical processes that take place in the body. These chemical processes convert the food you eat into the energy and materials needed for all life processes. ...
... • Metabolism is the total of all the chemical processes that take place in the body. These chemical processes convert the food you eat into the energy and materials needed for all life processes. ...
Carbohydrates & Begin Lipids
... • Animals convert excess carbohydrates into fats and store the fat molecules as droplets in cells of adipose (fat) tissue. ...
... • Animals convert excess carbohydrates into fats and store the fat molecules as droplets in cells of adipose (fat) tissue. ...
Questions for week 2 - Seattle Central College
... Questions for week 2 1. What organelle is the main site of cellular energy (ATP) production? What organic molecule do cells commonly metabolize (split/burn) to make ATP molecules? What are the names for those processes? What inorganic molecule must be present for a cell to produce LOTS of ATP molecu ...
... Questions for week 2 1. What organelle is the main site of cellular energy (ATP) production? What organic molecule do cells commonly metabolize (split/burn) to make ATP molecules? What are the names for those processes? What inorganic molecule must be present for a cell to produce LOTS of ATP molecu ...
The Structure and Function of Macromolecules
... monomers into a polysaccharide sugar. Place the remaining carbohydrate monomers into a chain. Use the triangle water to point to the bond site. Draw an arrow to show if water is being added or released during this reaction. ...
... monomers into a polysaccharide sugar. Place the remaining carbohydrate monomers into a chain. Use the triangle water to point to the bond site. Draw an arrow to show if water is being added or released during this reaction. ...
Cell Metabolism - s3.amazonaws.com
... Figure 4-3 Breakdown of glucose. A, Anaerobic: to lactic acid. B, Aerobic: to carbon dioxide, water, and ATP. Elsevier items and derived items © 2007, 2003, 2000 by Saunders, an imprint of Elsevier Inc. ...
... Figure 4-3 Breakdown of glucose. A, Anaerobic: to lactic acid. B, Aerobic: to carbon dioxide, water, and ATP. Elsevier items and derived items © 2007, 2003, 2000 by Saunders, an imprint of Elsevier Inc. ...
Biology * Introduction to Organic Chemistry
... most other biological molecules, lipids are hydrophobic (waterfearing). You can see this chemical behavior in an unshaken bottle of salad dressing. The oil (a type of lipid) separates from the vinegar (which is mostly water). ...
... most other biological molecules, lipids are hydrophobic (waterfearing). You can see this chemical behavior in an unshaken bottle of salad dressing. The oil (a type of lipid) separates from the vinegar (which is mostly water). ...
Organic and Biochemical Compounds (5.4) Notes
... Organic and Biochemical Compounds (5.4) Notes Organic Compounds ...
... Organic and Biochemical Compounds (5.4) Notes Organic Compounds ...
ch4 reading guide key
... 4. The nucleotides of the anticodon bind to nucleotides of the codon. 5. There are twenty types of amino acids. 6. There are sixty-four codons possible. 7. Three codons provide a stop signal. 8. A stop signal indicates the end of protein synthesis. 9. More than one type of tRNA can correspond to th ...
... 4. The nucleotides of the anticodon bind to nucleotides of the codon. 5. There are twenty types of amino acids. 6. There are sixty-four codons possible. 7. Three codons provide a stop signal. 8. A stop signal indicates the end of protein synthesis. 9. More than one type of tRNA can correspond to th ...
Chapter 16.3: Anaerobic Respiration
... – Reoxygenation of hemoglobin in the blood – High metabolic rate (as many organs are operating at above resting levels) ...
... – Reoxygenation of hemoglobin in the blood – High metabolic rate (as many organs are operating at above resting levels) ...
II. Control of Metabolic Reactions
... 4. The nucleotides of the anticodon bind to nucleotides of the codon. 5. There are 20 types of amino acids. 6. There are 64 codons possible. 7. Three codons provide a stop signal. 8. A stop signal indicates the end of protein synthesis. 9. More than one type of tRNA can correspond to the same amino ...
... 4. The nucleotides of the anticodon bind to nucleotides of the codon. 5. There are 20 types of amino acids. 6. There are 64 codons possible. 7. Three codons provide a stop signal. 8. A stop signal indicates the end of protein synthesis. 9. More than one type of tRNA can correspond to the same amino ...
4.1_Proteins_Amino_Acids_2011
... chain. The peptide bond is planar (gray shading) and does not permit rotation. By contrast, rotation can occur about the Cα–C bond, whose angle of rotation is called psi (ψ), and about the N–Cα bond, whose angle of rotation is called phi (ϕ). By convention, an R group is often used to denote an amin ...
... chain. The peptide bond is planar (gray shading) and does not permit rotation. By contrast, rotation can occur about the Cα–C bond, whose angle of rotation is called psi (ψ), and about the N–Cα bond, whose angle of rotation is called phi (ϕ). By convention, an R group is often used to denote an amin ...
Digestive System Learning Targets 6-10
... Mitochondria use glucose to produce a constant supply of ATP for the cell Essential fatty acids like Ω6 (linoleic acid) form plasma membranes Essential amino acids are used to construct proteins such as enzymes to carry out metabolism, & body structures – hair, nails, DNA ...
... Mitochondria use glucose to produce a constant supply of ATP for the cell Essential fatty acids like Ω6 (linoleic acid) form plasma membranes Essential amino acids are used to construct proteins such as enzymes to carry out metabolism, & body structures – hair, nails, DNA ...
MOLECULES IN CELLS - SITH-ITB
... same proportion as human protein: meat, dairy products Low quality protein: plant proteins Essential amino acids (8) cannot be synthesized by the body Right combination of food can provide the essential amino acids ...
... same proportion as human protein: meat, dairy products Low quality protein: plant proteins Essential amino acids (8) cannot be synthesized by the body Right combination of food can provide the essential amino acids ...
Amino Acids as Protein Building Blocks [2]
... Amino acids are most logically grouped according to the physical properties of their side chains. ...
... Amino acids are most logically grouped according to the physical properties of their side chains. ...
BIOCHEM MID SEM EXAM 2014 The Foundations of Biochemistry
... Qu. What is Biochemistry? Provide a brief definition in 30 words or less. The study of biomolecules inside & outside of cells from living systems and how they interact to maintain & perpetuate life via universal physical & chemical laws. Qu. What are the cellular foundations of biochemistry? - Cells ...
... Qu. What is Biochemistry? Provide a brief definition in 30 words or less. The study of biomolecules inside & outside of cells from living systems and how they interact to maintain & perpetuate life via universal physical & chemical laws. Qu. What are the cellular foundations of biochemistry? - Cells ...
BIO 330 Cell Biology Spring 2011 Lecture Outline Chemistry of the
... 4 – Activation involves coupling of monomer to carrier molecule 5 – Energy needed for condensation reaction is delivered by ATP 6 – Macromolecules have inherent directionality E. Degradation of macromolecules Hydrolysis reactions use water to break bonds between adjacent monomers V. The Importance o ...
... 4 – Activation involves coupling of monomer to carrier molecule 5 – Energy needed for condensation reaction is delivered by ATP 6 – Macromolecules have inherent directionality E. Degradation of macromolecules Hydrolysis reactions use water to break bonds between adjacent monomers V. The Importance o ...
2.24 MB - KFUPM Resources v3
... chains of nucleotides twisted together into a double helix with links of hydrogen bonds Contains all inherited information necessary to build/maintain an organism, coded in the order of the nucleotide bases, with each three base “letters” forming a genetic code “word” All living organisms have D ...
... chains of nucleotides twisted together into a double helix with links of hydrogen bonds Contains all inherited information necessary to build/maintain an organism, coded in the order of the nucleotide bases, with each three base “letters” forming a genetic code “word” All living organisms have D ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.

















![Amino Acids as Protein Building Blocks [2]](http://s1.studyres.com/store/data/001231379_1-1c71e58dd2b4a4d156753bc870eff68b-300x300.png)


