Nitrogen Metabolism During Fermentation*
... metabolism. For example, NADP dependent glutamate dehydrogenase enzymes catalyze the reaction, which fixes an ammonium ion in α-ketoglutarate to produce glutamate (Fig. 3.). In the case of a transamination reaction, the amino group is transferred from a donor molecule to a receptor molecule. Glutama ...
... metabolism. For example, NADP dependent glutamate dehydrogenase enzymes catalyze the reaction, which fixes an ammonium ion in α-ketoglutarate to produce glutamate (Fig. 3.). In the case of a transamination reaction, the amino group is transferred from a donor molecule to a receptor molecule. Glutama ...
Molecuar Structure of DNA Questions
... 5. How many DNA nucleotides are there? List them. Also indicate which are purines, and which are pyrimidines. ...
... 5. How many DNA nucleotides are there? List them. Also indicate which are purines, and which are pyrimidines. ...
Respiration of Glucose: The first stage of glucose metabolism is: is
... Respiration of Glucose: The first stage of glucose metabolism is: All steps are reversible except step #s ...
... Respiration of Glucose: The first stage of glucose metabolism is: All steps are reversible except step #s ...
2. Pyruvate Oxidation
... One molecule of NADH produced One molecule of CO2 is released The product of pyruvate oxidation is acetylCoA In cellular respiration, acetyl-CoA enters Krebs Cycle to eventually produce molecules of ATP ...
... One molecule of NADH produced One molecule of CO2 is released The product of pyruvate oxidation is acetylCoA In cellular respiration, acetyl-CoA enters Krebs Cycle to eventually produce molecules of ATP ...
Chapter 9. Cellular Respiration STAGE 1: Glycolysis
... 2. Some organisms that are exposed to oxygen, but switch to fermentation when oxygen is scarce. AP Biology ...
... 2. Some organisms that are exposed to oxygen, but switch to fermentation when oxygen is scarce. AP Biology ...
Anaerobic and Aerobic Glycolysis
... It turns out breakdown of glucose to pyruvate within muscle cells is not the primary source of ATP synthesis. ATP is the immediate source of energy for muscle contraction. Although a muscle fiber contains only enough ATP to power a few twitches, its ATP "pool" is replenished as needed. There are th ...
... It turns out breakdown of glucose to pyruvate within muscle cells is not the primary source of ATP synthesis. ATP is the immediate source of energy for muscle contraction. Although a muscle fiber contains only enough ATP to power a few twitches, its ATP "pool" is replenished as needed. There are th ...
Protein Synthesis - NCEA Level 2 Biology
... Transfer RNA (tRNA) • This is a clover-leaf shaped molecule. • It has a 3 base anticodon code, (complementary to the codon on mRNA) at one end, and at the other end there is a particular amino acid. • The function of tRNA is to pick up amino acids specific to the anticodon and carry them to the rib ...
... Transfer RNA (tRNA) • This is a clover-leaf shaped molecule. • It has a 3 base anticodon code, (complementary to the codon on mRNA) at one end, and at the other end there is a particular amino acid. • The function of tRNA is to pick up amino acids specific to the anticodon and carry them to the rib ...
Proteins - Many Structures, Many Functions
... foreign substances that fit their binding sites. – Enzyme recognize and bind to specific substrates, facilitating a chemical reaction. – Neurotransmitters pass signals from one cell to another by binding to receptor sites on proteins in the membrane of the receiving cell. ...
... foreign substances that fit their binding sites. – Enzyme recognize and bind to specific substrates, facilitating a chemical reaction. – Neurotransmitters pass signals from one cell to another by binding to receptor sites on proteins in the membrane of the receiving cell. ...
Energy Systems
... • The longer it takes a person to reach their threshold, the more “endurance” they have •Elite endurance athletes will have a high lactate threshold •Untrained individuals will have a low lactate threshold Therefore, the lower your LT, the less efficient your energy systems are working, or the poo ...
... • The longer it takes a person to reach their threshold, the more “endurance” they have •Elite endurance athletes will have a high lactate threshold •Untrained individuals will have a low lactate threshold Therefore, the lower your LT, the less efficient your energy systems are working, or the poo ...
Krebs (Citric Acid) Cycle
... of biomolecules like amino acids, nucleotides, chlorophyll, cytochromes and fats etc. 2. Intermediate like succinyl CoA takes part in the formation of chlorophyll. 3. Amino Acids are formed from α-Ketoglutaric acid, pyruvic acids and oxaloacetic acid. 4. Krebs cycle (citric acid cycle) releases plen ...
... of biomolecules like amino acids, nucleotides, chlorophyll, cytochromes and fats etc. 2. Intermediate like succinyl CoA takes part in the formation of chlorophyll. 3. Amino Acids are formed from α-Ketoglutaric acid, pyruvic acids and oxaloacetic acid. 4. Krebs cycle (citric acid cycle) releases plen ...
Review Guide for Third Exam in Biochemistry 507 (1997)
... 3. The reaction catalyzed by glutamate dehydrogenase 4. The reactions in the synthesis of carbamoyl phosphate 5. The sequence of steps, and the names (not structures) of the intermediates of the urea cycle 6. The reaction catalyzed by arginase (structures) 7. Role of the urea cycle in mammals Lectur ...
... 3. The reaction catalyzed by glutamate dehydrogenase 4. The reactions in the synthesis of carbamoyl phosphate 5. The sequence of steps, and the names (not structures) of the intermediates of the urea cycle 6. The reaction catalyzed by arginase (structures) 7. Role of the urea cycle in mammals Lectur ...
Lecture 023--Photosynthesis 2 (Dark Reactions)
... What will the C6H12O6 be used for? Where did the O2 come from? Where will the O2 go? What else is involved that is not listed in this equation? ...
... What will the C6H12O6 be used for? Where did the O2 come from? Where will the O2 go? What else is involved that is not listed in this equation? ...
Cellular Respiration
... • 4 high energy electrons are also removed and added to NAD+ (nicotinamide adenine dinucleotide) creating NADH, which helps pass energy from glucose to other pathways in the cell. ...
... • 4 high energy electrons are also removed and added to NAD+ (nicotinamide adenine dinucleotide) creating NADH, which helps pass energy from glucose to other pathways in the cell. ...
The Basics of Cellular Respiration
... given off, and NADH is formed • 4) 4C acid is “rearranged” 1ATP, and FADH2 is formed. • 5) 4C acid becomes oxaloacetate, and NADH is formed, The oxaloacetate is involved in a series of reactions and the cycle begins ...
... given off, and NADH is formed • 4) 4C acid is “rearranged” 1ATP, and FADH2 is formed. • 5) 4C acid becomes oxaloacetate, and NADH is formed, The oxaloacetate is involved in a series of reactions and the cycle begins ...
Cellular Respiration (CR
... eukaryotic cells (location and names of processes involved)? How many ATP’s are produced from each cell type? --------------------------------------------Define: The process of releasing energy (ATP) from food (glucose). Both consumers AND producers need to go through cellular respiration to turn th ...
... eukaryotic cells (location and names of processes involved)? How many ATP’s are produced from each cell type? --------------------------------------------Define: The process of releasing energy (ATP) from food (glucose). Both consumers AND producers need to go through cellular respiration to turn th ...
Chapter 8: The Origins of Life
... o Living systems can evolve Composition of Life o Life is based on carbon; many molecules of living organisms are based on long chains of carbon atoms o Carbon is prominent because it formed early during nucleosynthesis, so it is abundant in the universe; it is also versatile, forming one or more ...
... o Living systems can evolve Composition of Life o Life is based on carbon; many molecules of living organisms are based on long chains of carbon atoms o Carbon is prominent because it formed early during nucleosynthesis, so it is abundant in the universe; it is also versatile, forming one or more ...
CO2 would move across a plasma membrane more quickly than
... Something is inhibiting his cells from using oxygen. Glycolysis occurs, but with no usable oxygen respiration cannot continue. ...
... Something is inhibiting his cells from using oxygen. Glycolysis occurs, but with no usable oxygen respiration cannot continue. ...
Pset 1 Solutions
... If the size of each base pair is 0.34 nm, the total length of the genome in ALL the cells in a human is = (5 X 1013) x (6X 109) X 0.34nm or 1.02 X 1023 nm So the number of times this length of DNA could travel to and from the Sun is : (1.02 X 1023 nm / 1.496 x 1020 nm)/2 = (0.682 X 103)/2 = 681/ 2 = ...
... If the size of each base pair is 0.34 nm, the total length of the genome in ALL the cells in a human is = (5 X 1013) x (6X 109) X 0.34nm or 1.02 X 1023 nm So the number of times this length of DNA could travel to and from the Sun is : (1.02 X 1023 nm / 1.496 x 1020 nm)/2 = (0.682 X 103)/2 = 681/ 2 = ...
Lecture 17/18 - Aerobic and Anaerobic Metabolism
... 4.) What is the energy yield of the citric acid cycle? What type of phosphorylation produces the GTP? 5.) Summarize the overall net energy yield for 1 molecule of glucose that undergoes glycolysis and the CAC—It may be helpful to include the energy reactants and products 6.) Explain how the concent ...
... 4.) What is the energy yield of the citric acid cycle? What type of phosphorylation produces the GTP? 5.) Summarize the overall net energy yield for 1 molecule of glucose that undergoes glycolysis and the CAC—It may be helpful to include the energy reactants and products 6.) Explain how the concent ...
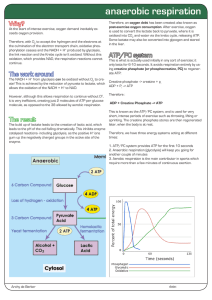
anaerobic respiration
... At the start of intense exercise, oxygen demand inevitably exceeds oxygen provision. Therefore, with O2 to accept the hydrogen and the electrons at the culmination of the electron transport chain, oxidative phosphorylation ceases and the NADH + H+ produced by glycolysis, the link reaction and the Kr ...
... At the start of intense exercise, oxygen demand inevitably exceeds oxygen provision. Therefore, with O2 to accept the hydrogen and the electrons at the culmination of the electron transport chain, oxidative phosphorylation ceases and the NADH + H+ produced by glycolysis, the link reaction and the Kr ...
General Definitions and Basic Concepts Describing Cancer
... images are called enantiomers. Molecules with chiral centers rotate plane-polarized light and therefore have optical activity. The importance of chirality will become evident when we discuss metabolism, because metabolizing enzymes have chiral active sites and therefore selectively generate one out ...
... images are called enantiomers. Molecules with chiral centers rotate plane-polarized light and therefore have optical activity. The importance of chirality will become evident when we discuss metabolism, because metabolizing enzymes have chiral active sites and therefore selectively generate one out ...
Lecture 023--Photosynthesis 2 (Dark Reactions)
... What will the C6H12O6 be used for? Where did the O2 come from? Where will the O2 go? What else is involved that is not listed in this equation? ...
... What will the C6H12O6 be used for? Where did the O2 come from? Where will the O2 go? What else is involved that is not listed in this equation? ...
Distinguish between mRNA, rRNA, and tRNA. What molecule does
... information carried by a gene. mRNA is transcribed from a DNA template, and carries information to the sites of protein synthesis: the ribosome. ...
... information carried by a gene. mRNA is transcribed from a DNA template, and carries information to the sites of protein synthesis: the ribosome. ...
No Slide Title
... • Occurs in the cytoplasm of all organisms. • 6 Carbon Glucose splits into 2 molecules each containing 3 carbons (called pyruvate) • Because Glucose is a pretty stable molecule, energy is needed to start this reaction (2ATP) ...
... • Occurs in the cytoplasm of all organisms. • 6 Carbon Glucose splits into 2 molecules each containing 3 carbons (called pyruvate) • Because Glucose is a pretty stable molecule, energy is needed to start this reaction (2ATP) ...
Biochemistry
_and_Carl_Ferdinand_Cori.jpg?width=300)
Biochemistry, sometimes called biological chemistry, is the study of chemical processes within and relating to living organisms. By controlling information flow through biochemical signaling and the flow of chemical energy through metabolism, biochemical processes give rise to the complexity of life. Over the last decades of the 20th century, biochemistry has become so successful at explaining living processes that now almost all areas of the life sciences from botany to medicine to genetics are engaged in biochemical research. Today, the main focus of pure biochemistry is in understanding how biological molecules give rise to the processes that occur within living cells, which in turn relates greatly to the study and understanding of whole organisms.Biochemistry is closely related to molecular biology, the study of the molecular mechanisms by which genetic information encoded in DNA is able to result in the processes of life. Depending on the exact definition of the terms used, molecular biology can be thought of as a branch of biochemistry, or biochemistry as a tool with which to investigate and study molecular biology.Much of biochemistry deals with the structures, functions and interactions of biological macromolecules, such as proteins, nucleic acids, carbohydrates and lipids, which provide the structure of cells and perform many of the functions associated with life. The chemistry of the cell also depends on the reactions of smaller molecules and ions. These can be inorganic, for example water and metal ions, or organic, for example the amino acids which are used to synthesize proteins. The mechanisms by which cells harness energy from their environment via chemical reactions are known as metabolism. The findings of biochemistry are applied primarily in medicine, nutrition, and agriculture. In medicine, biochemists investigate the causes and cures of disease. In nutrition, they study how to maintain health and study the effects of nutritional deficiencies. In agriculture, biochemists investigate soil and fertilizers, and try to discover ways to improve crop cultivation, crop storage and pest control.