Triosephosphate Isomerase (T2507) - Datasheet - Sigma
... dihydroxyacetone phosphate is favored by a ratio of 20:1 over the reverse reaction.1 A deficiency in TPI is an autosomal recessive disorder in children under five characterized by cardiomyopathy, congenital hemolytic anemia, and susceptibility to bacterial infection. Most children with this disorder ...
... dihydroxyacetone phosphate is favored by a ratio of 20:1 over the reverse reaction.1 A deficiency in TPI is an autosomal recessive disorder in children under five characterized by cardiomyopathy, congenital hemolytic anemia, and susceptibility to bacterial infection. Most children with this disorder ...
K - UCLA Chemistry and Biochemistry
... Side note: Gluconeogenesis is the reverse of glycolysis. The 7 reversible enzymes are the same, but 4 gluconeogeneic enzymes are needed to catalyze the opposite of the 3 irrevesible glycolytic enzymes. To get back to glucose, cells liberate 2 phosphate groups. Why use hydrolyases (glucose-6-phosphat ...
... Side note: Gluconeogenesis is the reverse of glycolysis. The 7 reversible enzymes are the same, but 4 gluconeogeneic enzymes are needed to catalyze the opposite of the 3 irrevesible glycolytic enzymes. To get back to glucose, cells liberate 2 phosphate groups. Why use hydrolyases (glucose-6-phosphat ...
pptx
... Side note: Gluconeogenesis is the reverse of glycolysis. The 7 reversible enzymes are the same, but 4 gluconeogeneic enzymes are needed to catalyze the opposite of the 3 irrevesible glycolytic enzymes. To get back to glucose, cells liberate 2 phosphate groups. Why use hydrolyases (glucose-6-phosphat ...
... Side note: Gluconeogenesis is the reverse of glycolysis. The 7 reversible enzymes are the same, but 4 gluconeogeneic enzymes are needed to catalyze the opposite of the 3 irrevesible glycolytic enzymes. To get back to glucose, cells liberate 2 phosphate groups. Why use hydrolyases (glucose-6-phosphat ...
$doc.title
... Glucose + oxygen Carbon dioxide + water +energy • Oxygen is the best electron acceptor, but what if it is not around? ...
... Glucose + oxygen Carbon dioxide + water +energy • Oxygen is the best electron acceptor, but what if it is not around? ...
- Wiley Online Library
... persistent dormant form of the bacilli. The generation of alanine is accompanied by the oxidation of NADH. This aspect of alanine synthesis might play a role under the oxygen limiting conditions encountered after de£ection from aerobic growth and later during anaerobic dormancy. Therefore, we propos ...
... persistent dormant form of the bacilli. The generation of alanine is accompanied by the oxidation of NADH. This aspect of alanine synthesis might play a role under the oxygen limiting conditions encountered after de£ection from aerobic growth and later during anaerobic dormancy. Therefore, we propos ...
Importance of pH Homeostasis in Metabolic Health and Diseases
... the resting state. Their actions are caused by elevation of activity and expression of related enzymes in skeletal muscles [59– 61]. Since the energy consumed in muscle during exercise is mainly supplied by carbohydrates and lipids, the exerciseinduced lipid utilization may decrease the energy obtai ...
... the resting state. Their actions are caused by elevation of activity and expression of related enzymes in skeletal muscles [59– 61]. Since the energy consumed in muscle during exercise is mainly supplied by carbohydrates and lipids, the exerciseinduced lipid utilization may decrease the energy obtai ...
Exam 2
... d. Coenzyme Q is an electron carrier molecule between Complex I and Complex III. e. None of the above 19. The followings are electron some carriers. ____ carries two electrons at one time. a. CoQ b. 2Fe-2S c. FMNH2 d. NADH e. None of them 20. The followings are some descriptions of ATP and NAD+. a. ...
... d. Coenzyme Q is an electron carrier molecule between Complex I and Complex III. e. None of the above 19. The followings are electron some carriers. ____ carries two electrons at one time. a. CoQ b. 2Fe-2S c. FMNH2 d. NADH e. None of them 20. The followings are some descriptions of ATP and NAD+. a. ...
Chapter 9 Cellular Respiration (working)
... • Most cellular respiration requires O2 to produce ATP • Glycolysis can produce ATP with or without O2 (in aerobic or anaerobic conditions), therefore this series of reactions evolved very early in prokaryotic organisms before oxygen was present in the atomosphere. • In the absence of O2, glycolysis ...
... • Most cellular respiration requires O2 to produce ATP • Glycolysis can produce ATP with or without O2 (in aerobic or anaerobic conditions), therefore this series of reactions evolved very early in prokaryotic organisms before oxygen was present in the atomosphere. • In the absence of O2, glycolysis ...
9/2/08 Transcript I - UAB School of Optometry
... Utilized in "Fight or Flight"- If confronted by a lion then you will fight or flee and use this type of process because it does not require any set up time or oxygen. There are 10 rxns which are the same in all cells, but may not happen at same rate. 2 Phases: 1. Converts glucose to two Glycer ...
... Utilized in "Fight or Flight"- If confronted by a lion then you will fight or flee and use this type of process because it does not require any set up time or oxygen. There are 10 rxns which are the same in all cells, but may not happen at same rate. 2 Phases: 1. Converts glucose to two Glycer ...
Hormonal regulation and pathologies of carbohydrate metabolism
... High levels of ATP allosterically inhibit the phosphofructokinase 1 in the liver lowering its affinity for fructose 6-phosphate. AMP reverses the inhibitory action of ATP, and so the activity of the enzyme increases when the ATP/AMP ratio is lowered (glycolysis is stimulated as the energy charge fa ...
... High levels of ATP allosterically inhibit the phosphofructokinase 1 in the liver lowering its affinity for fructose 6-phosphate. AMP reverses the inhibitory action of ATP, and so the activity of the enzyme increases when the ATP/AMP ratio is lowered (glycolysis is stimulated as the energy charge fa ...
Enzymes - Dr. Hamad Ali Yaseen
... • active site - a region of an enzyme comprised of different amino acids where catalysis occurs (determined by the tertiary and quaternary structure of each enzyme) • substrate - the molecule being utilized and/or modified by a particular enzyme at its active site • co-factor - organic or inorganic ...
... • active site - a region of an enzyme comprised of different amino acids where catalysis occurs (determined by the tertiary and quaternary structure of each enzyme) • substrate - the molecule being utilized and/or modified by a particular enzyme at its active site • co-factor - organic or inorganic ...
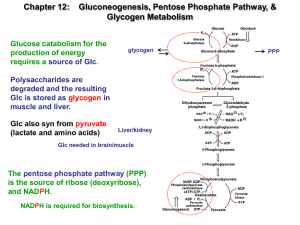
Gluconeogensis
... ii. it’s not phosphorylated in liver or kidney b/c these organs are responsible for gluconeogenesis 1. only these two organs can do gluconeogenesis XXVI. Glucose-6-Phosphate (S26) a. In the liver cell endoplasmic reticulum there is an enzyme called glucose-6-phosphatase i. This enzyme is only found ...
... ii. it’s not phosphorylated in liver or kidney b/c these organs are responsible for gluconeogenesis 1. only these two organs can do gluconeogenesis XXVI. Glucose-6-Phosphate (S26) a. In the liver cell endoplasmic reticulum there is an enzyme called glucose-6-phosphatase i. This enzyme is only found ...
O 2 - Madison Public Schools
... proteins all catabolized through same pathways enter at different points cell extracts energy from every source ...
... proteins all catabolized through same pathways enter at different points cell extracts energy from every source ...
Lb. curvatus
... This defect on the surfaces of cheese does not pose any health hazards, but may be mistaken by consumers as mold, resulting in economic loss to the industry. It is postulated that certain non-starter lactic acid bacteria (NSLAB) that grow in cheese are responsible. The conversion of pyruvate to L(+) ...
... This defect on the surfaces of cheese does not pose any health hazards, but may be mistaken by consumers as mold, resulting in economic loss to the industry. It is postulated that certain non-starter lactic acid bacteria (NSLAB) that grow in cheese are responsible. The conversion of pyruvate to L(+) ...
Word
... 20) Which of the following correctly describes galactose metabolism? A) Galactose is a substrate for hexokinase. B) Primarily ingested in the form of sucrose C) Galactokinase and galactose 1-phosphate uridyl transferase deficiencies cause mental retardation D) Galactose 1-phosphate uridyl transfera ...
... 20) Which of the following correctly describes galactose metabolism? A) Galactose is a substrate for hexokinase. B) Primarily ingested in the form of sucrose C) Galactokinase and galactose 1-phosphate uridyl transferase deficiencies cause mental retardation D) Galactose 1-phosphate uridyl transfera ...
Slide 1
... UDP-Glc synthases in protists, animals, and fungi. ADP-Glc synthase in plants. Primer of 4 to 8 Glc on a Tyr (-OH) of glycogenin. 1st Glc from UDP-Glc via Glc transferase. Remaining Glc’s tranferred by glycogenin. Amylo-(1,4 1,6)-transglycolase catalyzes the branch point. (Alpha 1-6 link) ...
... UDP-Glc synthases in protists, animals, and fungi. ADP-Glc synthase in plants. Primer of 4 to 8 Glc on a Tyr (-OH) of glycogenin. 1st Glc from UDP-Glc via Glc transferase. Remaining Glc’s tranferred by glycogenin. Amylo-(1,4 1,6)-transglycolase catalyzes the branch point. (Alpha 1-6 link) ...
PowerPoint Presentation - Mediterranean Emergency Medicine
... Recommended Dietary Thiamine 1 mg/day 0.5 mg/1000kcal Thiamine depletion develops within 18 days in thiamine free diet. Normally: organ meats, yeast, eggs, green leafy vegetables. Poorly absorbed in the presence of ethanol. J Nutr 1965;85:297-304. ...
... Recommended Dietary Thiamine 1 mg/day 0.5 mg/1000kcal Thiamine depletion develops within 18 days in thiamine free diet. Normally: organ meats, yeast, eggs, green leafy vegetables. Poorly absorbed in the presence of ethanol. J Nutr 1965;85:297-304. ...
Chapter 9. Cellular Respiration STAGE 1: Glycolysis
... 2. Glycolysis is a pathway in which One Six-Carbon Molecule of GLUCOSE is Oxidized to Produce Two ThreeCarbon Molecules of PYRUVIC ACID OR PYRUVATE. 3. The word "GLYCOLYSIS" means "The Splitting of Glucose". In a series of Ten Reactions, a molecule of Glucose is split into Two identical smaller ...
... 2. Glycolysis is a pathway in which One Six-Carbon Molecule of GLUCOSE is Oxidized to Produce Two ThreeCarbon Molecules of PYRUVIC ACID OR PYRUVATE. 3. The word "GLYCOLYSIS" means "The Splitting of Glucose". In a series of Ten Reactions, a molecule of Glucose is split into Two identical smaller ...
File E-Leraning : METABOLISME
... • Electrons from NADH and FADH2 were passed down the ETC • As the electrons move down, energy released moves protons to create electrochemical gradient • Protons move through proton channels, and release energy to synthesize ATP from ADP and Pi • The many processes of ATP synthesis are all continuou ...
... • Electrons from NADH and FADH2 were passed down the ETC • As the electrons move down, energy released moves protons to create electrochemical gradient • Protons move through proton channels, and release energy to synthesize ATP from ADP and Pi • The many processes of ATP synthesis are all continuou ...
12-Glycolysis2016-11-15 13:225.6 MB
... energy production during short, intense exercise, providing energy for a period ranging from 10 seconds to 2 minutes. ...
... energy production during short, intense exercise, providing energy for a period ranging from 10 seconds to 2 minutes. ...
Electron-Transport Chain and ATP production
... Electron-Transport Chain and ATP production Occurs in the inner mitochondrial membrane where NADH and FADH2 are oxidized back to NAD+ and FAD. They transfer their e- in a series of steps and ultimately to O2: O2 + 4e- + 4H+ → 2H2O The energy released in these e- transfers is used to pump H+ (protons ...
... Electron-Transport Chain and ATP production Occurs in the inner mitochondrial membrane where NADH and FADH2 are oxidized back to NAD+ and FAD. They transfer their e- in a series of steps and ultimately to O2: O2 + 4e- + 4H+ → 2H2O The energy released in these e- transfers is used to pump H+ (protons ...
Biochemistry The Citric Acid Cycle Chapter 17:
... used for synthesis – Anaplerotic reaction – reaction that replenishes a citric acid cycle intermediate – [Oxaloacetate] must allow acetyl-CoA to enter cycle – In mammals mammals, Pyruvate + CO2 + ATP + H2O → oxaloacetate + ADP + Pi + 2 H+ ...
... used for synthesis – Anaplerotic reaction – reaction that replenishes a citric acid cycle intermediate – [Oxaloacetate] must allow acetyl-CoA to enter cycle – In mammals mammals, Pyruvate + CO2 + ATP + H2O → oxaloacetate + ADP + Pi + 2 H+ ...
Lactate dehydrogenase

A lactate dehydrogenase (LDH or LD) is an enzyme found in nearly all living cells (animals, plants, and prokaryotes). LDH catalyzes the conversion of pyruvate to lactate and back, as it converts NADH to NAD+ and back. A dehydrogenase is an enzyme that transfers a hydride from one molecule to another.LDH exist in four distinct enzyme classes. This article is about the common NAD(P)-dependent L-lactate dehydrogenase. Other LDHs act on D-lactate and/or are dependent on cytochrome c: D-lactate dehydrogenase (cytochrome)) and L-lactate (L-lactate dehydrogenase (cytochrome)). LDH has been of medical significance because it is found extensively in body tissues, such as blood cells and heart muscle. Because it is released during tissue damage, it is a marker of common injuries and disease such as heart failure.