PYRUVATE OXIDATION, KREBS CYCLE agnes je... 583KB Nov 04
... Quick Review -In glycolysis, the first stage of cellular respiration, glucose, a 6-C chain molecule was broken down into 2 pyruvate molecules in a series of 10 steps. ...
... Quick Review -In glycolysis, the first stage of cellular respiration, glucose, a 6-C chain molecule was broken down into 2 pyruvate molecules in a series of 10 steps. ...
Lecture Eighteen - Personal Webspace for QMUL
... NOTE: Fatty acid oxidation, pyruvate dehydrogenase complex, the citric acid cycle => located in the _________ NOTE: Respiratory electron transfer, oxidative phosphorylation, and solute transfer in and out of mitochondria => located on the IMM The Endosymbiotic Theory of the Evolution of Mitoch ...
... NOTE: Fatty acid oxidation, pyruvate dehydrogenase complex, the citric acid cycle => located in the _________ NOTE: Respiratory electron transfer, oxidative phosphorylation, and solute transfer in and out of mitochondria => located on the IMM The Endosymbiotic Theory of the Evolution of Mitoch ...
TCA (Krebs) Cycle
... No part of this presentation may be reproduced by any mechanical, photographic, or electronic process, or in the form of a phonographic recording, nor may it be stored in a retrieval system, transmitted, or otherwise copied for public or private use, without written permission from the publisher. ...
... No part of this presentation may be reproduced by any mechanical, photographic, or electronic process, or in the form of a phonographic recording, nor may it be stored in a retrieval system, transmitted, or otherwise copied for public or private use, without written permission from the publisher. ...
BSC 2010 - Exam I Lectures and Text Pages Citric Acid Cycle • Citric
... • Complete oxidation of 1 mole of glucose releases 686 kcal of energy • Phosphorylation of ADP → ATP stores 7.3 ...
... • Complete oxidation of 1 mole of glucose releases 686 kcal of energy • Phosphorylation of ADP → ATP stores 7.3 ...
The Presence and Function of Cytochromes in
... absence and presence of potassium nitrate (50 mM). Veillonella alcalescens was grown with sodium lactate (30 or 40 mM) or sodium lactate (30 mM) and potassium nitrate (100mM). j- Values, expressed as the percentage of the total amount of fermentation products, represent the mean of the percentages f ...
... absence and presence of potassium nitrate (50 mM). Veillonella alcalescens was grown with sodium lactate (30 or 40 mM) or sodium lactate (30 mM) and potassium nitrate (100mM). j- Values, expressed as the percentage of the total amount of fermentation products, represent the mean of the percentages f ...
File - Groby Bio Page
... is decarboxylated (CO2) to form Start with Pyruvate produced in Acetate Glycolysis This produces Acetate Acetyl coenzyme It is also dehydrogenated (H+Aremoved) The Acetyl coenzyme A now enters Krebs cycle ...
... is decarboxylated (CO2) to form Start with Pyruvate produced in Acetate Glycolysis This produces Acetate Acetyl coenzyme It is also dehydrogenated (H+Aremoved) The Acetyl coenzyme A now enters Krebs cycle ...
triose phosphate
... organisms mainly bacteria can only respire anaerobically others can switch to anaerobic when oxygen levels are low. ...
... organisms mainly bacteria can only respire anaerobically others can switch to anaerobic when oxygen levels are low. ...
Chapter 9 Cell Respiration
... • If ATP concentration begins to drop, respiration speeds up; when there is plenty of ATP, respiration slows down • Control of catabolism is based mainly on regulating the activity of enzymes at strategic points in the catabolic pathway ...
... • If ATP concentration begins to drop, respiration speeds up; when there is plenty of ATP, respiration slows down • Control of catabolism is based mainly on regulating the activity of enzymes at strategic points in the catabolic pathway ...
Respiration: Occurs in two places in the cell Cytoplasm and
... How is ATP made during Glycolysis? Substrate level Phosphorylation Transfer of phosphate from substrate directly to ADP ...
... How is ATP made during Glycolysis? Substrate level Phosphorylation Transfer of phosphate from substrate directly to ADP ...
Document
... - to ethanol (fermentation) via ethanol dehydrogenase • Aerobic pathway – through citric acid cycle and respiration; Enough O2,this pathway yields far more energy NADH + O2 NAD+ + energy Pyruvate + O2 3CO2 + energy Oxygen availability determines fate of Pyruvate ...
... - to ethanol (fermentation) via ethanol dehydrogenase • Aerobic pathway – through citric acid cycle and respiration; Enough O2,this pathway yields far more energy NADH + O2 NAD+ + energy Pyruvate + O2 3CO2 + energy Oxygen availability determines fate of Pyruvate ...
Cellular Respiration
... • If ATP concentration begins to drop, respiration speeds up; when there is plenty of ATP, respiration slows down • Control of catabolism is based mainly on regulating the activity of enzymes at strategic points in the catabolic pathway ...
... • If ATP concentration begins to drop, respiration speeds up; when there is plenty of ATP, respiration slows down • Control of catabolism is based mainly on regulating the activity of enzymes at strategic points in the catabolic pathway ...
Chapter 9
... • If ATP concentration begins to drop, respiration speeds up; when there is plenty of ATP, respiration slows down • Control of catabolism is based mainly on regulating the activity of enzymes at strategic points in the catabolic pathway ...
... • If ATP concentration begins to drop, respiration speeds up; when there is plenty of ATP, respiration slows down • Control of catabolism is based mainly on regulating the activity of enzymes at strategic points in the catabolic pathway ...
U4L23 starvation - The University of Sydney
... Glucose Accounting • The brain needs ~120g/day, • Substrates for gluconeogenesis – ~30g glucose from glycerol per day – Glucose from lactate is just recycling – Alanine from muscle/tissue proteolysis • Would need to provide 90 g/day • Or 180 g protein per day, just for the brain ...
... Glucose Accounting • The brain needs ~120g/day, • Substrates for gluconeogenesis – ~30g glucose from glycerol per day – Glucose from lactate is just recycling – Alanine from muscle/tissue proteolysis • Would need to provide 90 g/day • Or 180 g protein per day, just for the brain ...
Lactic acid fermentation
... is usually done through an electron transport chain in a process called oxidative phosphorylation; however, this mechanism is not available without oxygen.[3][4] Instead, the NADH donates its extra electrons to the pyruvate molecules formed during glycolysis. Since the NADH has lost electrons, NAD+ ...
... is usually done through an electron transport chain in a process called oxidative phosphorylation; however, this mechanism is not available without oxygen.[3][4] Instead, the NADH donates its extra electrons to the pyruvate molecules formed during glycolysis. Since the NADH has lost electrons, NAD+ ...
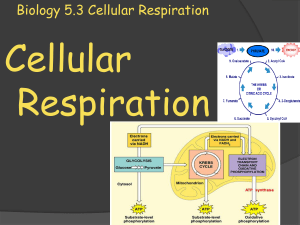
Biology 5.3 Cellular Respiration
... waste products of respiration. A simple formula to show respiration looks like this: Glucose + oxygen carbon dioxide (waste) + water (waste) + energy ...
... waste products of respiration. A simple formula to show respiration looks like this: Glucose + oxygen carbon dioxide (waste) + water (waste) + energy ...
WEEK 8 - WordPress.com
... Advantages/Disadvantages of Fermentation • Lactic acid fermentation is critical for certain animals and tissues • Animals use lactic acid for rapid bursts of energy • Lactic acid fermentation provides continued production of ATP in absence of oxygen via the cycling of NAD+ • Lactic acid and alcohol ...
... Advantages/Disadvantages of Fermentation • Lactic acid fermentation is critical for certain animals and tissues • Animals use lactic acid for rapid bursts of energy • Lactic acid fermentation provides continued production of ATP in absence of oxygen via the cycling of NAD+ • Lactic acid and alcohol ...
PPT - Chris Anthony
... Systems involving methylated amino acids In ‘non-pigmented pseudomonads’: Ps. aminovorans; Pseudomonas MA, & Hyphomicrobium Two step system Methylamine + glutamate N-methylglutamate + PMS + H2O ...
... Systems involving methylated amino acids In ‘non-pigmented pseudomonads’: Ps. aminovorans; Pseudomonas MA, & Hyphomicrobium Two step system Methylamine + glutamate N-methylglutamate + PMS + H2O ...
The energy-less red blood cell is lost: erythrocyte
... The subunits are expressed in a tissue-specific manner and, in erythrocytes, 5 isoenzymes of varying subunit composition (M4, M3L1, M2L2, ML3, and L4) can be identified.37 The gene encoding the M subunit (PFKM) has been assigned to chromosome 12q13.3 and spans 30 kb. It contains 27 exons and at leas ...
... The subunits are expressed in a tissue-specific manner and, in erythrocytes, 5 isoenzymes of varying subunit composition (M4, M3L1, M2L2, ML3, and L4) can be identified.37 The gene encoding the M subunit (PFKM) has been assigned to chromosome 12q13.3 and spans 30 kb. It contains 27 exons and at leas ...
Cellular Respiration
... • 2.Kreb’s Cycle: mitochondrial matrix; pyruvate into carbon dioxide • 3.Electron Transport Chain: inner membrane of mitochondrion; electrons passed to oxygen ...
... • 2.Kreb’s Cycle: mitochondrial matrix; pyruvate into carbon dioxide • 3.Electron Transport Chain: inner membrane of mitochondrion; electrons passed to oxygen ...
Chapter 8
... Enzymes that catalyze near-equilibrium reactions tend to – act quickly – restore equilibrium concentrations – net rates of such reactions are effectively regulated by the relative concentration of substrates and products ...
... Enzymes that catalyze near-equilibrium reactions tend to – act quickly – restore equilibrium concentrations – net rates of such reactions are effectively regulated by the relative concentration of substrates and products ...
Document
... • Irreversible inhibitors usually covalently modify an enzyme, and inhibition cannot therefore be reversed. • Irreversible inhibitors often contain reactive functional groups react with amino acid side chains in the enzyme active sites to form covalent adducts. • The side chain of amino acid may be ...
... • Irreversible inhibitors usually covalently modify an enzyme, and inhibition cannot therefore be reversed. • Irreversible inhibitors often contain reactive functional groups react with amino acid side chains in the enzyme active sites to form covalent adducts. • The side chain of amino acid may be ...
Document
... - anaerobic = no requirement of oxygen - cytoplasmic location - 10 step enzymatic pathway hexose --> 2 PYR + 4ATP (2 net) + 2NADH • energy investment phase (coupled Rx's) phosphorylation of low energy intermediates ...
... - anaerobic = no requirement of oxygen - cytoplasmic location - 10 step enzymatic pathway hexose --> 2 PYR + 4ATP (2 net) + 2NADH • energy investment phase (coupled Rx's) phosphorylation of low energy intermediates ...
Lactate dehydrogenase

A lactate dehydrogenase (LDH or LD) is an enzyme found in nearly all living cells (animals, plants, and prokaryotes). LDH catalyzes the conversion of pyruvate to lactate and back, as it converts NADH to NAD+ and back. A dehydrogenase is an enzyme that transfers a hydride from one molecule to another.LDH exist in four distinct enzyme classes. This article is about the common NAD(P)-dependent L-lactate dehydrogenase. Other LDHs act on D-lactate and/or are dependent on cytochrome c: D-lactate dehydrogenase (cytochrome)) and L-lactate (L-lactate dehydrogenase (cytochrome)). LDH has been of medical significance because it is found extensively in body tissues, such as blood cells and heart muscle. Because it is released during tissue damage, it is a marker of common injuries and disease such as heart failure.