chapter2
... carbon and hydrogen Saturated hydrocarbon: a hydrocarbon containing only single bonds Alkane: a saturated hydrocarbon whose carbons are arranged in an open chain Aliphatic hydrocarbon: another name for an alkane ...
... carbon and hydrogen Saturated hydrocarbon: a hydrocarbon containing only single bonds Alkane: a saturated hydrocarbon whose carbons are arranged in an open chain Aliphatic hydrocarbon: another name for an alkane ...
Unsaturated hydrocarbons Alkenes
... The second class of simple hydrocarbons the alkenes, consists of molecules that contain at least one doublebonded carbon pair. The chemical formula for the simple alkenes follows the expression: ...
... The second class of simple hydrocarbons the alkenes, consists of molecules that contain at least one doublebonded carbon pair. The chemical formula for the simple alkenes follows the expression: ...
Chapter 22 and 23 Study Guide
... notes or help from anyone. You should review homework, quizzes and other assignments for complete test preparation. These questions are only examples. Different chemicals will be on the test. ...
... notes or help from anyone. You should review homework, quizzes and other assignments for complete test preparation. These questions are only examples. Different chemicals will be on the test. ...
IUPAC System of Nomenclature
... Monocyclic alkanes or alkenes, those where the carbon chain forms a single closed ring, are named by putting the prefix cyclo- in front of the name based on the number of carbons in the ring. All other rules for substituents and numbering are the same, and written as prefixes to the ring name. ...
... Monocyclic alkanes or alkenes, those where the carbon chain forms a single closed ring, are named by putting the prefix cyclo- in front of the name based on the number of carbons in the ring. All other rules for substituents and numbering are the same, and written as prefixes to the ring name. ...
Aldehydes and Ketones
... Naming Aldehydes IUPAC Replace the -e in the alkane name with –al Common Add aldehyde to the prefixes form (1C), acet (2C), propion(3), and butry(4C) O ...
... Naming Aldehydes IUPAC Replace the -e in the alkane name with –al Common Add aldehyde to the prefixes form (1C), acet (2C), propion(3), and butry(4C) O ...
Chemistry 30 - SharpSchool
... 30-C1.2sts explain that science and technology have influenced, and been influenced by, historical development and societal needs 30-C1.3s follow appropriated IUPAC guidelines when writing the names and formulas of organic compounds ...
... 30-C1.2sts explain that science and technology have influenced, and been influenced by, historical development and societal needs 30-C1.3s follow appropriated IUPAC guidelines when writing the names and formulas of organic compounds ...
Naming Organic Compounds
... CH3-CH2Cl + KOH CH3-CH2OH + KCl CH3-CH2OH + HCl CH3-CH2Cl + H2O CH3CH3 + Cl2 CH3CH2Cl + HCl ...
... CH3-CH2Cl + KOH CH3-CH2OH + KCl CH3-CH2OH + HCl CH3-CH2Cl + H2O CH3CH3 + Cl2 CH3CH2Cl + HCl ...
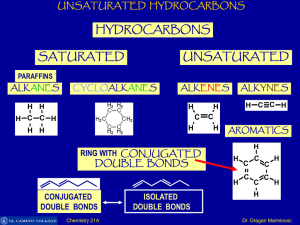
Unsaturated Hydrocarbons
... C-atom overlap with two sp2-hybrid orbital of two other C-atoms to form sigma bonds. In this way there are six sigma bonds are formed between six C-atoms which are 120o apart. Remaining six sp2-orbital of six C-atoms overlap with 1s orbital of six H-atoms individually to form six sigma bonds. Since ...
... C-atom overlap with two sp2-hybrid orbital of two other C-atoms to form sigma bonds. In this way there are six sigma bonds are formed between six C-atoms which are 120o apart. Remaining six sp2-orbital of six C-atoms overlap with 1s orbital of six H-atoms individually to form six sigma bonds. Since ...
Revised organic chemistry
... USE:-This method is also suitable for the preparation of symmetrical alkanes. Properties of Alkanes: Physical Properties: 1) First four members (methane to butane) are colourless, odourles gases , next thirteen (C3 to C17)are colourless, odourles liquids while higher alkanes are colourless wax like ...
... USE:-This method is also suitable for the preparation of symmetrical alkanes. Properties of Alkanes: Physical Properties: 1) First four members (methane to butane) are colourless, odourles gases , next thirteen (C3 to C17)are colourless, odourles liquids while higher alkanes are colourless wax like ...
organic intro
... (i) a prefix which comes from the number of carbon atoms in the longest straight chain in the molecule. Apart from the first four, which have trivial names, the number of carbons atoms is indicated by a prefix derived from the Greek numbering system. Prefix methethpropbutpenthexheptoctnondec- ...
... (i) a prefix which comes from the number of carbon atoms in the longest straight chain in the molecule. Apart from the first four, which have trivial names, the number of carbons atoms is indicated by a prefix derived from the Greek numbering system. Prefix methethpropbutpenthexheptoctnondec- ...
Chapter 25 Organic and Biological Chemistry
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prentice- ...
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prentice- ...
Chapter 25 Organic and Biological Chemistry
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prenti ...
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prenti ...
Chapter 25 Organic and Biological Chemistry
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prentice- ...
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prentice- ...
Chapter 25 Organic and Biological Chemistry
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prentice- ...
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prentice- ...
Chapter 25 Organic and Biological Chemistry
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prentice- ...
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prentice- ...
Chapter 25 Organic and Biological Chemistry
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prentice- ...
... How to Name a Compound 1. Find the longest chain in the molecule. 2. Number the chain from the end nearest the first substituent encountered. 3. List the substituents as a prefix along with the number(s) of the carbon(s) to which they are attached. Organic and Biological Chemistry © 2009, Prentice- ...
CHAPTER 15
... (1) In the IUPAC nomenclature system, an aldehyde group has priority over a ketone group. (2) Addition of an alcohol molecule across the carbon-oxygen double bond of an aldehyde produces a compound in which the carbonyl carbon atom bears both an alkoxy group and a hydroxy group. (3) 2-Propenal conta ...
... (1) In the IUPAC nomenclature system, an aldehyde group has priority over a ketone group. (2) Addition of an alcohol molecule across the carbon-oxygen double bond of an aldehyde produces a compound in which the carbonyl carbon atom bears both an alkoxy group and a hydroxy group. (3) 2-Propenal conta ...
chm121 tutorial kit - Covenant University
... The following are common drying agent for organic solutions but which of them has high capacity, fast speed, and good efficiency as its chemical property. (a) CaSO4 (b) CaCl 2 (c) K2CO3 (d) MgSO4 All except ------------- is not employed as an adsorbents in column chromatography (a) Starch (b) Silica ...
... The following are common drying agent for organic solutions but which of them has high capacity, fast speed, and good efficiency as its chemical property. (a) CaSO4 (b) CaCl 2 (c) K2CO3 (d) MgSO4 All except ------------- is not employed as an adsorbents in column chromatography (a) Starch (b) Silica ...
Alcohols, Phenols, and Ethers
... (3) Rubbing alcohol is a 50-50 mixture by volume of isopropyl alcohol and water. a) All three statements are true. b) Two of the three statements are true. c) Only one of the statements is true. d) None of the statements is true. 14.38 c - FTF Statements: (1) Glycerin, a three-carbon diol, has a gre ...
... (3) Rubbing alcohol is a 50-50 mixture by volume of isopropyl alcohol and water. a) All three statements are true. b) Two of the three statements are true. c) Only one of the statements is true. d) None of the statements is true. 14.38 c - FTF Statements: (1) Glycerin, a three-carbon diol, has a gre ...
Organic Chemistry II
... Alkyl Halides We did talk about halo-alkanes (called alkyl halides) which are alkanes with a halogen attached. These molecules do, in fact, have polar bonds: C-Br, C-I, C-Cl are all polar bonds. Carbon is slightly positive, the halogen is slightly negative. ...
... Alkyl Halides We did talk about halo-alkanes (called alkyl halides) which are alkanes with a halogen attached. These molecules do, in fact, have polar bonds: C-Br, C-I, C-Cl are all polar bonds. Carbon is slightly positive, the halogen is slightly negative. ...
Aromatic Hydrocarbon Tutorial
... these compounds display physicochemical and reactivity profiles very different from dipolar compounds. Because of their atomic composition, aromatic hydrocarbons are classified as “non-polar" compounds or substituents and the only significant intermolecular bonding possible are relatively weak van d ...
... these compounds display physicochemical and reactivity profiles very different from dipolar compounds. Because of their atomic composition, aromatic hydrocarbons are classified as “non-polar" compounds or substituents and the only significant intermolecular bonding possible are relatively weak van d ...
Alkenes notes
... -bond. Since these orbitals overlap in two places, i -bond about its axis without breaking the bonds. There is thus restricted rotation about the double bond. If both carbon atoms on either side of the bond are attached to different groups, then two different structures arise which cannot be interco ...
... -bond. Since these orbitals overlap in two places, i -bond about its axis without breaking the bonds. There is thus restricted rotation about the double bond. If both carbon atoms on either side of the bond are attached to different groups, then two different structures arise which cannot be interco ...
Ultimate Analysis - Cheresources.com
... The presence of sulfur compounds in finished petroleum products often produces harmful effects. For example, in gasoline, sulfur compounds are believed to promote corrosion of engine parts, especially under winter conditions, when water containing sulfur dioxide from the combustion may accumulate in ...
... The presence of sulfur compounds in finished petroleum products often produces harmful effects. For example, in gasoline, sulfur compounds are believed to promote corrosion of engine parts, especially under winter conditions, when water containing sulfur dioxide from the combustion may accumulate in ...
CHM 103 Lecture 24 S07
... • contains an ─O─ between two carbon groups (R-O-R’). • has a common name that gives the alkyl names of the attached groups followed by ether. CH3─O─CH3 CH3─CH2─O─CH3 ...
... • contains an ─O─ between two carbon groups (R-O-R’). • has a common name that gives the alkyl names of the attached groups followed by ether. CH3─O─CH3 CH3─CH2─O─CH3 ...
CH 2 CH(CH 3 ) - Parkway C-2
... Always include both the double and triple bond in the longest chain – even if it isn’t the most number of carbons! Start counting from the end closest to the double or triple bond – whichever has the lowest number If there is a tie, and only then, do double bonds take priority over triple ...
... Always include both the double and triple bond in the longest chain – even if it isn’t the most number of carbons! Start counting from the end closest to the double or triple bond – whichever has the lowest number If there is a tie, and only then, do double bonds take priority over triple ...
Alkane
In organic chemistry, an alkane, or paraffin (a historical name that also has other meanings), is a saturated hydrocarbon. Alkanes consist only of hydrogen and carbon atoms and all bonds are single bonds. Alkanes (technically, always acyclic or open-chain compounds) have the general chemical formula CnH2n+2. For example, Methane is CH4, in which n=1 (n being the number of Carbon atoms). Alkanes belong to a homologous series of organic compounds in which the members differ by a molecular mass of 14.03u (mass of a methanediyl group, —CH2—, one carbon atom of mass 12.01u, and two hydrogen atoms of mass ≈1.01u each). There are two main commercial sources: petroleum (crude oil) and natural gas.Each carbon atom has 4 bonds (either C-H or C-C bonds), and each hydrogen atom is joined to a carbon atom (H-C bonds). A series of linked carbon atoms is known as the carbon skeleton or carbon backbone. The number of carbon atoms is used to define the size of the alkane e.g., C2-alkane.An alkyl group, generally abbreviated with the symbol R, is a functional group or side-chain that, like an alkane, consists solely of single-bonded carbon and hydrogen atoms, for example a methyl or ethyl group.The simplest possible alkane (the parent molecule) is methane, CH4. There is no limit to the number of carbon atoms that can be linked together, the only limitation being that the molecule is acyclic, is saturated, and is a hydrocarbon. Waxes include examples of larger alkanes where the number of carbons in the carbon backbone is greater than about 17, above which the compounds are solids at standard ambient temperature and pressure (SATP).Alkanes are not very reactive and have little biological activity. All alkanes are colourless and odourless. Alkanes can be viewed as a molecular tree upon which can be hung the more biologically active/reactive portions (functional groups) of the molecule.