Reduction of Aldehydes and Ketones
... Benedict’s Test Benedict’s test • gives a positive result with compounds that have an aldehyde functional group and an adjacent hydroxyl group. • utilizes Benedict’s solution, which contains Cu2+ (CuSO4). When the solution is added to this type of aldehyde and heated, a brick-red solid of Cu2O form ...
... Benedict’s Test Benedict’s test • gives a positive result with compounds that have an aldehyde functional group and an adjacent hydroxyl group. • utilizes Benedict’s solution, which contains Cu2+ (CuSO4). When the solution is added to this type of aldehyde and heated, a brick-red solid of Cu2O form ...
Exam III
... stereospecific reactions, be able to show the structures for the alkene reactants. -given the structures of the reactants and the products be able to state whether a reaction is syn- or anti-. ...
... stereospecific reactions, be able to show the structures for the alkene reactants. -given the structures of the reactants and the products be able to state whether a reaction is syn- or anti-. ...
Predicting Products online assistance #3
... 2. decomposition - one reactant decomposes, or breaks apart, into two or more products. 3. single replacement - an element replaces another in a compound. 4. double replacement - the elements in two compounds switch partners to form two new compounds. Writing Balanced Equations A chemical reaction i ...
... 2. decomposition - one reactant decomposes, or breaks apart, into two or more products. 3. single replacement - an element replaces another in a compound. 4. double replacement - the elements in two compounds switch partners to form two new compounds. Writing Balanced Equations A chemical reaction i ...
Chemical Reaction Th..
... rxn (2) won’t complete (with a feed CH4=CO2=1 & CO=H2=0, max. conv.=63% at 1000K) ...
... rxn (2) won’t complete (with a feed CH4=CO2=1 & CO=H2=0, max. conv.=63% at 1000K) ...
Chapter 1 Chemical Bonding and Chemical Structure
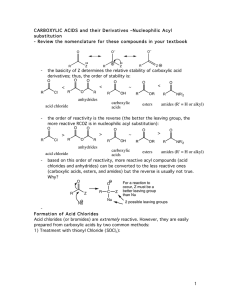
... • Recall priorities: carboxylic acid > anhydride > ester > acid halide > amide > nitrile > aldehyde > ketone > alcohol (phenol) > thiol > amine 16.1 Nomenclature of Benzene Derivatives ...
... • Recall priorities: carboxylic acid > anhydride > ester > acid halide > amide > nitrile > aldehyde > ketone > alcohol (phenol) > thiol > amine 16.1 Nomenclature of Benzene Derivatives ...
Chapter 19 Reaction Rates And Equilibrium
... molecules) must collide in order for reactions to occur • An effective collision occurs when reactant particles approach each other: – at the proper angle – With enough Energy (Force) ...
... molecules) must collide in order for reactions to occur • An effective collision occurs when reactant particles approach each other: – at the proper angle – With enough Energy (Force) ...
File - IGCSE STUDY BANK
... The rate of a reaction that produces a gas can also be measured by following the mass loss as the gas is formed and escapes from the reaction flask. o The method is ok for reactions producing carbon dioxide or oxygen, o but not very accurate for reactions giving hydrogen (low mass loss). ...
... The rate of a reaction that produces a gas can also be measured by following the mass loss as the gas is formed and escapes from the reaction flask. o The method is ok for reactions producing carbon dioxide or oxygen, o but not very accurate for reactions giving hydrogen (low mass loss). ...
ORGSEQPP.pps
... It may be surprising that water needs such vigorous conditions to react with ethene. It is a highly polar molecule and you would expect it to be a good electrophile. ...
... It may be surprising that water needs such vigorous conditions to react with ethene. It is a highly polar molecule and you would expect it to be a good electrophile. ...
C 2 H 5 OH(l)
... It may be surprising that water needs such vigorous conditions to react with ethene. It is a highly polar molecule and you would expect it to be a good electrophile. ...
... It may be surprising that water needs such vigorous conditions to react with ethene. It is a highly polar molecule and you would expect it to be a good electrophile. ...
Chapter 1 Structure and Bonding
... The carboxy carbon is attacked by nucleophiles (like other carbonyls) 1) In aldehydes and ketones, addition is followed by aqueous workup to give alcohol 2) In Carboxylic Acids and derivatives, there is a potential leaving group: elimination ...
... The carboxy carbon is attacked by nucleophiles (like other carbonyls) 1) In aldehydes and ketones, addition is followed by aqueous workup to give alcohol 2) In Carboxylic Acids and derivatives, there is a potential leaving group: elimination ...
hc1(8)notes
... • To complete the process of writing a correct equation, the law of conservation of mass must be taken into account. • The relative amounts of reactants and products represented in the equation must be adjusted so that the numbers and types of atoms are the same on both sides of the equation. • This ...
... • To complete the process of writing a correct equation, the law of conservation of mass must be taken into account. • The relative amounts of reactants and products represented in the equation must be adjusted so that the numbers and types of atoms are the same on both sides of the equation. • This ...
A-level Paper 2 Practice Paper 6 - A
... Identify compounds H and S and give reagents and conditions for Steps 1 and 2. State the type of compound of which W is an example. W reacts with a large excess of bromomethane to form a solid product. Draw the structure of this product and name the type of mechanism for this reaction. ...
... Identify compounds H and S and give reagents and conditions for Steps 1 and 2. State the type of compound of which W is an example. W reacts with a large excess of bromomethane to form a solid product. Draw the structure of this product and name the type of mechanism for this reaction. ...
Chemical reactions unit
... 1. Determine number of atoms for each element. 2. Pick an element that is not equal on both sides of the equation. 3. Add a coefficient in front of the formula with that element and adjust your counts. 4. Continue adding coefficients to get the same number of atoms of each element on each side. ...
... 1. Determine number of atoms for each element. 2. Pick an element that is not equal on both sides of the equation. 3. Add a coefficient in front of the formula with that element and adjust your counts. 4. Continue adding coefficients to get the same number of atoms of each element on each side. ...
A Model for Catalytically Active Zinc(I1) Ion in Liver
... 1,4,7,10-tetraazacyclododecane,L2),Zn"[ 14]aneN4, 3 ([ 14]aneN4 = 1,4,8,1I-tetraazacyclotetradecane,LJ,and free Zn" salts, 4. Variations in Zn" acidity and coordination environment in these complexes result in varying degrees of catalytic activity in the reduction of p-nitrobenzaldehyde (9) and an N ...
... 1,4,7,10-tetraazacyclododecane,L2),Zn"[ 14]aneN4, 3 ([ 14]aneN4 = 1,4,8,1I-tetraazacyclotetradecane,LJ,and free Zn" salts, 4. Variations in Zn" acidity and coordination environment in these complexes result in varying degrees of catalytic activity in the reduction of p-nitrobenzaldehyde (9) and an N ...
Chemical reactions unit
... 1. You CANNOT balance an equation by changing the chemical formulas. You must leave the subscripts in the formulas alone!! 2. An equation can be balanced only by: putting numbers, called coefficients, in front of the chemical formulas. The coefficients tell how many molecules of that compound ar ...
... 1. You CANNOT balance an equation by changing the chemical formulas. You must leave the subscripts in the formulas alone!! 2. An equation can be balanced only by: putting numbers, called coefficients, in front of the chemical formulas. The coefficients tell how many molecules of that compound ar ...
1. Natures Chemistry Unit Questions
... (ii) How would the reaction mixture be heated? (1) (c) Aldehydes can also be formed by the reaction of some alcohols with copper(II) oxide. Name the type of alcohol that would react with copper(II) oxide to form an aldehyde. ...
... (ii) How would the reaction mixture be heated? (1) (c) Aldehydes can also be formed by the reaction of some alcohols with copper(II) oxide. Name the type of alcohol that would react with copper(II) oxide to form an aldehyde. ...
ppt
... Conclusion • Very simple strategy for engaging CO2 in C–C bond formation that does not require synthetic or biological catalysts. • The ability to deprotonate unactivated C–H bonds opens the possibility of using this approach to prepare numerous high-volume targets. ...
... Conclusion • Very simple strategy for engaging CO2 in C–C bond formation that does not require synthetic or biological catalysts. • The ability to deprotonate unactivated C–H bonds opens the possibility of using this approach to prepare numerous high-volume targets. ...
Asymmetric induction

Asymmetric induction (also enantioinduction) in stereochemistry describes the preferential formation in a chemical reaction of one enantiomer or diastereoisomer over the other as a result of the influence of a chiral feature present in the substrate, reagent, catalyst or environment. Asymmetric induction is a key element in asymmetric synthesis.Asymmetric induction was introduced by Hermann Emil Fischer based on his work on carbohydrates. Several types of induction exist.Internal asymmetric induction makes use of a chiral center bound to the reactive center through a covalent bond and remains so during the reaction. The starting material is often derived from chiral pool synthesis. In relayed asymmetric induction the chiral information is introduced in a separate step and removed again in a separate chemical reaction. Special synthons are called chiral auxiliaries. In external asymmetric induction chiral information is introduced in the transition state through a catalyst of chiral ligand. This method of asymmetric synthesis is economically most desirable.