Synthesis of DiamidoPyrrolyl Molybdenum Complexes Relevant to Reduction DOI: 10.1021/ic100856n
... decomposition of [(Art-BuN)2Pyr]Mo-NdNH through a variety of pathways. Three attempts to reduce dinitrogen catalytically with [(Art-BuN)2Pyr]Mo(N) as a “catalyst” yielded an average of 1.02 ( 0.12 equiv of NH3. ...
... decomposition of [(Art-BuN)2Pyr]Mo-NdNH through a variety of pathways. Three attempts to reduce dinitrogen catalytically with [(Art-BuN)2Pyr]Mo(N) as a “catalyst” yielded an average of 1.02 ( 0.12 equiv of NH3. ...
- University of Bath Opus
... borrowing hydrogen methodology with masked ammonia sources. This unique approach allows selective access to primary amines without the over alkylation seen when using other ammonia sources. Furthermore, the borrowing hydrogen reaction can be coupled with an in-situ palladium catalysed hydrogenolysis ...
... borrowing hydrogen methodology with masked ammonia sources. This unique approach allows selective access to primary amines without the over alkylation seen when using other ammonia sources. Furthermore, the borrowing hydrogen reaction can be coupled with an in-situ palladium catalysed hydrogenolysis ...
24 Gastrointestinal protein and amino acid metabolism in
... intermediates; and (3) complete oxidation to CO2. In the first two pathways, amino acids can be deposited and recycled by the body for purposes of growth or other biological functions. In the case of some amino acids, namely threonine and cysteine, incorporation into endogenous secretions that are fe ...
... intermediates; and (3) complete oxidation to CO2. In the first two pathways, amino acids can be deposited and recycled by the body for purposes of growth or other biological functions. In the case of some amino acids, namely threonine and cysteine, incorporation into endogenous secretions that are fe ...
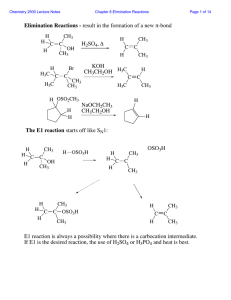
Reactions of Alkyl Halides (SN1, SN2, E1, and E2 reactions)
... Polar solvents, both protic and aprotic, will solvate and stabilize the charged transition state (C+ intermediate), lowering the activation energy and accelerating SN1 reactions. Nonpolar solvents do not lower the activation energy and thus make SN1 reactions relatively slower The relative rates ...
... Polar solvents, both protic and aprotic, will solvate and stabilize the charged transition state (C+ intermediate), lowering the activation energy and accelerating SN1 reactions. Nonpolar solvents do not lower the activation energy and thus make SN1 reactions relatively slower The relative rates ...
Derivatization - Sigma
... performance and peak shape. The derivative should give a single symmetrical peak corresponding to the parent compound. The functional group of polar compounds do not chromatograph well as they tend to adsorb on the active surfaces of the column wall causing peak tailing, poor response or no response ...
... performance and peak shape. The derivative should give a single symmetrical peak corresponding to the parent compound. The functional group of polar compounds do not chromatograph well as they tend to adsorb on the active surfaces of the column wall causing peak tailing, poor response or no response ...
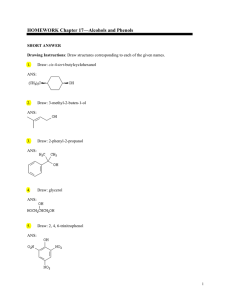
Alcohols phenols
... alcohols as the latter are easily available. It is a nucleophilic substitution reaction in which hydroxide ion substitutes halide ions. Among alkyl halides, alkyl Iodides undergo nucleophilic substitution at the fastest rate. The mode of mechanism SN1 and SN2 depends on the nature of alkyl group. Te ...
... alcohols as the latter are easily available. It is a nucleophilic substitution reaction in which hydroxide ion substitutes halide ions. Among alkyl halides, alkyl Iodides undergo nucleophilic substitution at the fastest rate. The mode of mechanism SN1 and SN2 depends on the nature of alkyl group. Te ...
Unsaturated hydrocarbons Alkenes
... and other oxidizing agents. When alkenes are added to the purple alkaline potassium manganate, there is a color change to brown manganese dioxide (MnO2) as the alkenes are being oxidized. This reaction is called Baeyer test for the unsaturation. The General equation C C ...
... and other oxidizing agents. When alkenes are added to the purple alkaline potassium manganate, there is a color change to brown manganese dioxide (MnO2) as the alkenes are being oxidized. This reaction is called Baeyer test for the unsaturation. The General equation C C ...
An efficient acetylation of dextran using in situ activated acetic
... Bruker 400 MHz instrument in suitable deuterated solvents. Thermogravimetric (TG) analysis and derivative thermogravimetric (DTG) analysis were realized on a SDT Q600 (USA) thermal analyzer. Thermal decomposition temperatures (td) of the dextran esters were determined from the DTG curves. The td is ...
... Bruker 400 MHz instrument in suitable deuterated solvents. Thermogravimetric (TG) analysis and derivative thermogravimetric (DTG) analysis were realized on a SDT Q600 (USA) thermal analyzer. Thermal decomposition temperatures (td) of the dextran esters were determined from the DTG curves. The td is ...
C1 polymerization and related C-C bond forming - UvA-DARE
... stability only a few reports describe the polymerization (or oligomerization) of αcarbonyl stabilized ‘carbenes’ from diazocarbonyl compounds (vide infra). Ylides of sulfoxides (e.g. ylides of DMSO) are also valuable synthetic precursors to carbenes. Elimination of a stable neutral fragment from eit ...
... stability only a few reports describe the polymerization (or oligomerization) of αcarbonyl stabilized ‘carbenes’ from diazocarbonyl compounds (vide infra). Ylides of sulfoxides (e.g. ylides of DMSO) are also valuable synthetic precursors to carbenes. Elimination of a stable neutral fragment from eit ...
Ch-6-Alcohols and phenols - Home
... Methanol and ethanol can be used as an alternative to fossil fuels as they burn very cleanly, producing only carbon dioxide and water. Ethanol is considered a renewable fuel as it can be made from renewable sources such a sugar cane and can be used as a fuel in its own right, or in mixtures with pet ...
... Methanol and ethanol can be used as an alternative to fossil fuels as they burn very cleanly, producing only carbon dioxide and water. Ethanol is considered a renewable fuel as it can be made from renewable sources such a sugar cane and can be used as a fuel in its own right, or in mixtures with pet ...
chm121 tutorial kit - Covenant University
... structure of an alkyne. (c) Addition of water and zinc dust to ozonide gives aldehydes and or ketones. (d) Ethane can add on to itself to form an addition polymer. ...
... structure of an alkyne. (c) Addition of water and zinc dust to ozonide gives aldehydes and or ketones. (d) Ethane can add on to itself to form an addition polymer. ...
OChem1 Course Pack
... The secondary carbocation that is formed undergoes rearrangement to produce a better tertiary cation that no longer has the strained cyclobutyl ring involved. The tertiary carbocation is then deprotonated to give the most highly substituted alkene which is stabilized by four electron-donating alkyl ...
... The secondary carbocation that is formed undergoes rearrangement to produce a better tertiary cation that no longer has the strained cyclobutyl ring involved. The tertiary carbocation is then deprotonated to give the most highly substituted alkene which is stabilized by four electron-donating alkyl ...
acetyl coenzyme A - Fakultas Farmasi Unand
... Dimethylallyl pyrophosphate has a leaving group (pyrophosphate) at an allylic carbon; it is reactive toward nucleophilic substitution at this position. Dr. Wolf's CHM 424 ...
... Dimethylallyl pyrophosphate has a leaving group (pyrophosphate) at an allylic carbon; it is reactive toward nucleophilic substitution at this position. Dr. Wolf's CHM 424 ...
FUNCTIONAL GROUPS OF TREE FERNS (CYATHEA) USING FT
... may date back to thousands of years i.e. from the time of using wild plants as a source of medicine [15]. In the earlier days, only morphological characters were used to identify the drug. But now, identification of plants based on morphological parameters is very tenuous. To overcome this, molecula ...
... may date back to thousands of years i.e. from the time of using wild plants as a source of medicine [15]. In the earlier days, only morphological characters were used to identify the drug. But now, identification of plants based on morphological parameters is very tenuous. To overcome this, molecula ...
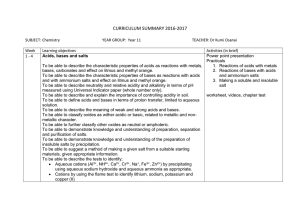
CURRICULUM SUMMARY 2016-2017 SUBJECT: Chemistry YEAR
... on health and discuss why these pollutants are of global concern. To be able to state the conditions required for the rusting of iron. To be able to describe ad explain methods of rust prevention, specifically paint and other coating to exclude oxygen. To be able to describe and explain sacrificial ...
... on health and discuss why these pollutants are of global concern. To be able to state the conditions required for the rusting of iron. To be able to describe ad explain methods of rust prevention, specifically paint and other coating to exclude oxygen. To be able to describe and explain sacrificial ...
The behaviour of esters in the presence of
... to improve the gas chromatographi~ resolution of mixtures of fatty acids. In a number of more recent papers [7-lo] natural esters were converted to their methyl esters by tetramethyl- or other tetraalkylammonium hydroxides under pyrolysis conditions. It was indicated that the methyl esters were the ...
... to improve the gas chromatographi~ resolution of mixtures of fatty acids. In a number of more recent papers [7-lo] natural esters were converted to their methyl esters by tetramethyl- or other tetraalkylammonium hydroxides under pyrolysis conditions. It was indicated that the methyl esters were the ...
Aldehydes And Ketones
... • the parent alkane is the longest chain that contains the carbonyl group • for ketones, change the suffix -e to -one • number the chain to give C=O the smaller number • the IUPAC retains the common names acetone, acetophenone, and benzophenone O ...
... • the parent alkane is the longest chain that contains the carbonyl group • for ketones, change the suffix -e to -one • number the chain to give C=O the smaller number • the IUPAC retains the common names acetone, acetophenone, and benzophenone O ...
aa-2005-38-71-negishi - University of Windsor
... alkylation had been achieved by using alkylmetals. The latter is still of much broader synthetic applicability. However, some recent developments suggest that this generalization may have to be significantly modified in the future, as discussed in Section 2.6. Another group of categorically difficul ...
... alkylation had been achieved by using alkylmetals. The latter is still of much broader synthetic applicability. However, some recent developments suggest that this generalization may have to be significantly modified in the future, as discussed in Section 2.6. Another group of categorically difficul ...
15alcpp - Knockhardy
... It is a green fuel, does not contribute to the carbon dioxide (CO2) burden and produces drastically reduced engine emissions. It is non-toxic and ...
... It is a green fuel, does not contribute to the carbon dioxide (CO2) burden and produces drastically reduced engine emissions. It is non-toxic and ...
Petasis reaction

The Petasis reaction (alternatively called the Petasis borono–Mannich (PBM) reaction) is the chemical reaction of an amine, aldehyde, and vinyl- or aryl-boronic acid to form substituted amines.Reported in 1993 by Nicos Petasis as a practical method towards the synthesis of a geometrically pure antifungal agent, naftifine, the Petasis reaction can be described as a variation of the Mannich reaction. Rather than generating an enolate to form the substituted amine product, in the Petasis reaction, the vinyl group of the organoboronic acid serves as the nucleophile. In comparison to other methods of generating allyl amines, the Petasis reaction tolerates a multifunctional scaffold, with a variety of amines and organoboronic acids as potential starting materials. Additionally, the reaction does not require anhydrous or inert conditions. As a mild, selective synthesis, the Petasis reaction is useful in generating α-amino acids, and is utilized in combinatorial chemistry and drug discovery.