Oxygen containing organic compound
... Are they soluble? aldehydes and ketones • electronegative O atom of carbonyl group •aldehydes and ketones •hydrogen bonds with water •not between each other, thus ...
... Are they soluble? aldehydes and ketones • electronegative O atom of carbonyl group •aldehydes and ketones •hydrogen bonds with water •not between each other, thus ...
Development of Catalytic Ester Condensations and Hydrolysis of
... Since a variety of esters are synthesized on an industrial scale all over the world, large amounts of byproducts are also generated during synthetic processes. Therefore, the use of stoichiometric dehydrating reagents, activated carboxylic acid derivatives, excess amounts of substrates, or solvents ...
... Since a variety of esters are synthesized on an industrial scale all over the world, large amounts of byproducts are also generated during synthetic processes. Therefore, the use of stoichiometric dehydrating reagents, activated carboxylic acid derivatives, excess amounts of substrates, or solvents ...
Organic Chemistry Fifth Edition
... chemical equations showing the elementary steps. An elementary step is a reaction that proceeds by way of a single transition state. Mechanisms can be shown likely to be correct, but cannot be proven correct. ...
... chemical equations showing the elementary steps. An elementary step is a reaction that proceeds by way of a single transition state. Mechanisms can be shown likely to be correct, but cannot be proven correct. ...
Document
... Markovnikov’s rule (1869) in the addition of HX to an unsymmetrical alkene, the H atom adds to the less substituted carbon atom—that is, the carbon that has the greater number of H atoms to ...
... Markovnikov’s rule (1869) in the addition of HX to an unsymmetrical alkene, the H atom adds to the less substituted carbon atom—that is, the carbon that has the greater number of H atoms to ...
Boron and Metal Catalyzed CC and CH Bond Formation
... dialkynylation of aryl aldehydes using dialkynylboron chloride. Numerous applications of these novel reactions have been developed. These include alternate routes to diphenylmethanes and 1,4-diynes from easily prepared dialkynylboron chlorides. In addition, E and Z alkenyl halides can now be prepare ...
... dialkynylation of aryl aldehydes using dialkynylboron chloride. Numerous applications of these novel reactions have been developed. These include alternate routes to diphenylmethanes and 1,4-diynes from easily prepared dialkynylboron chlorides. In addition, E and Z alkenyl halides can now be prepare ...
Chemistry of DOW Glycol Ether Products
... one moles of ethylene oxide reacted. The Butyl designates the alcohol from which the glycol ether was produced, in this case n-butanol. Since Butyl CELLOSOLVE contains an alcohol group, it can be reacted with additional ethylene oxide to yield Butyl CARBITOL™ diethylene glycol butyl ether. The CARBI ...
... one moles of ethylene oxide reacted. The Butyl designates the alcohol from which the glycol ether was produced, in this case n-butanol. Since Butyl CELLOSOLVE contains an alcohol group, it can be reacted with additional ethylene oxide to yield Butyl CARBITOL™ diethylene glycol butyl ether. The CARBI ...
Synthesis of Natural Products and Related Compounds using Enyne
... our group (Scheme 6).[22] Reactions of five- to sevenmembered cycloalkenes 14 having the substituent at the 3-position of the cycloalkene with 1c under ethylene gas afforded the cyclic compounds 15 in good yields. This reaction could proceed via the highly strained ruthenacyclobutane 16. In each cas ...
... our group (Scheme 6).[22] Reactions of five- to sevenmembered cycloalkenes 14 having the substituent at the 3-position of the cycloalkene with 1c under ethylene gas afforded the cyclic compounds 15 in good yields. This reaction could proceed via the highly strained ruthenacyclobutane 16. In each cas ...
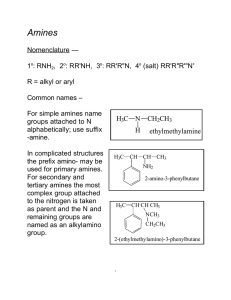
226 amines lec
... than other carbonyl compounds. This can also be explained by resonance. [In the table to the left, the most basic compound is at the top and the least basic is at the bottom. The conjugate acids of these bases are also shown, along with their pKas. The weakest conjugate acid is at the top of the lis ...
... than other carbonyl compounds. This can also be explained by resonance. [In the table to the left, the most basic compound is at the top and the least basic is at the bottom. The conjugate acids of these bases are also shown, along with their pKas. The weakest conjugate acid is at the top of the lis ...
Palladium and Ruthenium Catalyzed Reactions By Bryan Jaksic
... activity of commonly used precatalysts with the newly synthesized precatalyst, Pd(η5-C5H5)(η3-1Ph-C3H4), for Sonogashira cross-coupling reactions. Sonogashira reactions are important as they provide a simple method for the formation of substituted alkynes, a commonly found functionality within impor ...
... activity of commonly used precatalysts with the newly synthesized precatalyst, Pd(η5-C5H5)(η3-1Ph-C3H4), for Sonogashira cross-coupling reactions. Sonogashira reactions are important as they provide a simple method for the formation of substituted alkynes, a commonly found functionality within impor ...
Ch 9 Lecture 2
... 2) No Hydrogen Bonding is possible in R—O—R 3) Boiling Points are much lower than alcohols, more like haloalkanes 4) Water solubility much less than alcohols a) MeOMe and EtOEt have some water solubility b) Larger ethers are insoluble, very much like alkanes 5) Fairly unreactive, nonpolar solvents f ...
... 2) No Hydrogen Bonding is possible in R—O—R 3) Boiling Points are much lower than alcohols, more like haloalkanes 4) Water solubility much less than alcohols a) MeOMe and EtOEt have some water solubility b) Larger ethers are insoluble, very much like alkanes 5) Fairly unreactive, nonpolar solvents f ...
Amidations of Rosin with Isocyanates
... Color is an important indication of modified rosin products, which somewhat reflects oxidation of rosin (Hind et aZ. 1954; Nakamura, 1970). In this study, we used color number according to JIS K5902 to monitor color changes of the reaction products of rosin with phenyl isocyanate as shown in Table 3 ...
... Color is an important indication of modified rosin products, which somewhat reflects oxidation of rosin (Hind et aZ. 1954; Nakamura, 1970). In this study, we used color number according to JIS K5902 to monitor color changes of the reaction products of rosin with phenyl isocyanate as shown in Table 3 ...
lecture 7 reductive eliminations
... OXIDATIVE ADDITION – CONCERTED MECHANISM • Concerted, or three‐center, oxidative addition is really an associative reaction in which the incoming ligand first binds as a σ complex and then undergoes bond breaking as a result of strong back donation from the metal into the * orbital. • Non‐polar re ...
... OXIDATIVE ADDITION – CONCERTED MECHANISM • Concerted, or three‐center, oxidative addition is really an associative reaction in which the incoming ligand first binds as a σ complex and then undergoes bond breaking as a result of strong back donation from the metal into the * orbital. • Non‐polar re ...
Alcohols
... • Nucleophilic substitution of OH- on alkyl halide • Hydration of alkenes water in acid solution (not very effective) oxymercuration - demercuration hydroboration - oxidation ...
... • Nucleophilic substitution of OH- on alkyl halide • Hydration of alkenes water in acid solution (not very effective) oxymercuration - demercuration hydroboration - oxidation ...
Synthesis of Nitrogen-, Oxygen- and Sulphur
... ditionally, we prepared 1,3,5-tris((4-phenyl-1-H-1,2,3-triazol1-yl)methyl)benzene (9) by addition of three equivalents of phenylacetylene to 7 in the presence of a catalytic amount of CuI (Scheme 5) [13]. Examination of the reactivity of compound 8 in Michael addition reactions revealed that the exp ...
... ditionally, we prepared 1,3,5-tris((4-phenyl-1-H-1,2,3-triazol1-yl)methyl)benzene (9) by addition of three equivalents of phenylacetylene to 7 in the presence of a catalytic amount of CuI (Scheme 5) [13]. Examination of the reactivity of compound 8 in Michael addition reactions revealed that the exp ...
Organic Chemistry II Introduction
... Alcohols and Phenols • Important solvents and intermediates • Phenols contain an OH group connected to a carbon in a benzene ...
... Alcohols and Phenols • Important solvents and intermediates • Phenols contain an OH group connected to a carbon in a benzene ...
06_10_13.html
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
Slide 1
... Elimination Reactions involving Amines Amines can under go two different types of elimination reactions that give alkenes. Hofmann elimination: An amine is exhaustively methylated to the quaternary ammonium salt. The halide salt is converted to the hydroxide salt by treatment with silver oxide. The ...
... Elimination Reactions involving Amines Amines can under go two different types of elimination reactions that give alkenes. Hofmann elimination: An amine is exhaustively methylated to the quaternary ammonium salt. The halide salt is converted to the hydroxide salt by treatment with silver oxide. The ...
Efficient and Convenient Procedure for Protection of Hydroxyl
... processes whereby the protecting groups may be obtained under milder conditions. Results and Discussion Room temperature ionic liquids (RTILs) could be suitable and environmentally safer replacements for the volatile, toxic and flammable organic solvents currently used in synthetic and catalytic rea ...
... processes whereby the protecting groups may be obtained under milder conditions. Results and Discussion Room temperature ionic liquids (RTILs) could be suitable and environmentally safer replacements for the volatile, toxic and flammable organic solvents currently used in synthetic and catalytic rea ...
Improved Synthesis, Separation, Transition Metal Coordination and
... The 6-10.5 ppm region of the 1H NMR spectra: sample from reaction with 1-hexene, meso-(et,ph-P4) in acetone-d6/ D2O under N2, 24 hours (blue spectrum), same as above recorded 1.5 hours after exposure to O2(red spectrum).......................................................... 68 ...
... The 6-10.5 ppm region of the 1H NMR spectra: sample from reaction with 1-hexene, meso-(et,ph-P4) in acetone-d6/ D2O under N2, 24 hours (blue spectrum), same as above recorded 1.5 hours after exposure to O2(red spectrum).......................................................... 68 ...
CHAPTER 11 BONDING AND MOLECULAR STRUCTURE:
... adds to the carbon with the most hydrogen. For alkynes, the addition is always two mole to one mole of alkyne, the product being a substituted alkane. If hydrogen gas is added, the process is called hydrogenation. ...
... adds to the carbon with the most hydrogen. For alkynes, the addition is always two mole to one mole of alkyne, the product being a substituted alkane. If hydrogen gas is added, the process is called hydrogenation. ...
Recent developments in electrochemical and photoelectrochemical
... semiconducting p-type diamond electrodes for CO2 reduction, because hydrogen evolution is extremely inhibited on such surfaces.19 Indeed, using methanol as the solvent with high-pressure CO2, we found that essentially no hydrogen was produced; CO, methyl formate and hydrocarbons were the major produ ...
... semiconducting p-type diamond electrodes for CO2 reduction, because hydrogen evolution is extremely inhibited on such surfaces.19 Indeed, using methanol as the solvent with high-pressure CO2, we found that essentially no hydrogen was produced; CO, methyl formate and hydrocarbons were the major produ ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.