Advanced Practical Organic Chemistry
... of possible compounds the constituent atoms and their exact combination determines the chemical and physical properties of compounds and hence, their suitability for applications. To understand life as we know it, we must first understand a little bit of organic chemistry. Organic molecules contain ...
... of possible compounds the constituent atoms and their exact combination determines the chemical and physical properties of compounds and hence, their suitability for applications. To understand life as we know it, we must first understand a little bit of organic chemistry. Organic molecules contain ...
Efficient hydrogenation of organic carbonates, carbamates and
... complexes 1–4 as catalysts. Reaction of methyl formate and dihydrogen (7 atm) catalysed by 1 (0.1 mol%) at 145 8C in 1,4dioxane yielded 71% of methanol after 30 h (Table 3, entry 1). Performing the reaction under 9 atm of H2 resulted in complete conversion of methyl formate to methanol after 36 h wi ...
... complexes 1–4 as catalysts. Reaction of methyl formate and dihydrogen (7 atm) catalysed by 1 (0.1 mol%) at 145 8C in 1,4dioxane yielded 71% of methanol after 30 h (Table 3, entry 1). Performing the reaction under 9 atm of H2 resulted in complete conversion of methyl formate to methanol after 36 h wi ...
Alcohols, Phenols, and Thiols
... acidity is due to charge delocalization (resonance) in phenoxide ions. Electron-withdrawing groups, such as F and NO2, increase acidity, through either an inductive or a resonance effect, or both. Alkoxides, the conjugate bases of alcohols, are prepared from alcohols by reaction with reactive meta ...
... acidity is due to charge delocalization (resonance) in phenoxide ions. Electron-withdrawing groups, such as F and NO2, increase acidity, through either an inductive or a resonance effect, or both. Alkoxides, the conjugate bases of alcohols, are prepared from alcohols by reaction with reactive meta ...
organic chemistry - Sakshieducation.com
... 3. Explain the structure and chemical nature of ethers. Ans: Structure : In CH3OCH3, the central oxygen atom is sp3 hybridized with two completely filled sp3 orbitals having lone pair of electrons and two half filled sp3 hybridized orbitals. Also carbon atoms are sp3 hybridized and both the half fil ...
... 3. Explain the structure and chemical nature of ethers. Ans: Structure : In CH3OCH3, the central oxygen atom is sp3 hybridized with two completely filled sp3 orbitals having lone pair of electrons and two half filled sp3 hybridized orbitals. Also carbon atoms are sp3 hybridized and both the half fil ...
Iodine and Lipase Based Green Oxidation Technology
... Ease of recovery of reagents and catalysts is important for two reasons: (1) complete removal of catalyst is necessary to prevent contamination of the product. (2) It allows the recycling of the catalyst. Heterogenization is a commonly applied procedure that allows easy and efficient r ...
... Ease of recovery of reagents and catalysts is important for two reasons: (1) complete removal of catalyst is necessary to prevent contamination of the product. (2) It allows the recycling of the catalyst. Heterogenization is a commonly applied procedure that allows easy and efficient r ...
Transition Metal Catalyzed Selective Oxidation of Sugars and Polyols
... oxidation of a secondary alcohol of sugar substrates to keto-sugars using transition metal catalysis were investigated. Research results demonstrate that Shvo’s hydrogen-transfer catalyst, [(η4-C4Ph4CO)(CO)2Ru]2, selectively oxidized unprotected sugars to δ-lactones under very mild conditions. This ...
... oxidation of a secondary alcohol of sugar substrates to keto-sugars using transition metal catalysis were investigated. Research results demonstrate that Shvo’s hydrogen-transfer catalyst, [(η4-C4Ph4CO)(CO)2Ru]2, selectively oxidized unprotected sugars to δ-lactones under very mild conditions. This ...
New Stereoselective Approaches to Highly Substituted
... developed by Kazmaier is described below, in addition to the modifications of the procedure conducted by Sharland. ...
... developed by Kazmaier is described below, in addition to the modifications of the procedure conducted by Sharland. ...
AMIDES AND AMINES: ORGANIC NITROGEN COMPOUNDS
... are odorless and colorless. Low molar-mass amides are water soluble. Solubility in water decreases as the molar mass increases. Amides are neutral compounds. The group is capable of hydrogen bonding. Amines: Low molar-mass amines are flammable gases with an ammonia-like odor. Aliphatic amines up to ...
... are odorless and colorless. Low molar-mass amides are water soluble. Solubility in water decreases as the molar mass increases. Amides are neutral compounds. The group is capable of hydrogen bonding. Amines: Low molar-mass amines are flammable gases with an ammonia-like odor. Aliphatic amines up to ...
Synthesis of alternating hydroxy-and methyl
... cyclopropane in ligating the mercuric salt, which leads to exceedingly long reaction times. Treatment of free alcohol 10a and its corresponding tert-butyldiphenylsilyl ether 10b with mercuric salts led to complex product mixtures. Reaction of acetate 1Oc with mercuric trifluoroacetate produced diol ...
... cyclopropane in ligating the mercuric salt, which leads to exceedingly long reaction times. Treatment of free alcohol 10a and its corresponding tert-butyldiphenylsilyl ether 10b with mercuric salts led to complex product mixtures. Reaction of acetate 1Oc with mercuric trifluoroacetate produced diol ...
Chapter 7. Alcohols, Thiols, Phenols, Ethers
... There are many examples of the use of Grignard reactions. The example above could be expanded into many variations just by changing the halide and the carbonyl compound. Variations with other types of organometallic compound are also available. The reaction is given in more detail in chapter 8 on al ...
... There are many examples of the use of Grignard reactions. The example above could be expanded into many variations just by changing the halide and the carbonyl compound. Variations with other types of organometallic compound are also available. The reaction is given in more detail in chapter 8 on al ...
OChem1 Course Pack
... The secondary carbocation that is formed undergoes rearrangement to produce a better tertiary cation that no longer has the strained cyclobutyl ring involved. The tertiary carbocation is then deprotonated to give the most highly substituted alkene which is stabilized by four electron-donating alkyl ...
... The secondary carbocation that is formed undergoes rearrangement to produce a better tertiary cation that no longer has the strained cyclobutyl ring involved. The tertiary carbocation is then deprotonated to give the most highly substituted alkene which is stabilized by four electron-donating alkyl ...
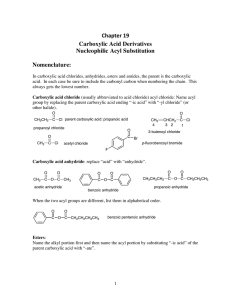
Chapter 19 Carboxylic Acid Derivatives Nucleophilic Acyl
... withdraws electrons from the carbonyl carbon inductively (i.e. through the bond) but oxygen is a first row element and the C-O bond is considerably shorter than the C-Cl bond. Consequently there is much better overlap of the oxygen lone pair with the π-orbital of the carbonyl. But since there are tw ...
... withdraws electrons from the carbonyl carbon inductively (i.e. through the bond) but oxygen is a first row element and the C-O bond is considerably shorter than the C-Cl bond. Consequently there is much better overlap of the oxygen lone pair with the π-orbital of the carbonyl. But since there are tw ...
RUMPLE-DISSERTATION-2014 - SMARTech Home
... I would like to begin by thanking my advisors Dr. Charles Liotta and Dr. Charles Eckert. The opportunity to work with such skilled scientists and kind mentors is a rare one, and I am extremely glad I had the opportunity to learn from them. I have always been in awe of their brilliance (I’m pretty su ...
... I would like to begin by thanking my advisors Dr. Charles Liotta and Dr. Charles Eckert. The opportunity to work with such skilled scientists and kind mentors is a rare one, and I am extremely glad I had the opportunity to learn from them. I have always been in awe of their brilliance (I’m pretty su ...
Alkyl Halides02
... This is also a qualitative test for identifying alcohols, i.e, the Lucas test. HCl with ZnCl 2 catalyst are used. 3 alcohols react quickly, 2 slowly, and 1 don’t react. Note that 1 and 2 alcohols will react with special reagents to produce alkyl halides (i.e., thionyl chloride, SOCl2, or PBr3) ...
... This is also a qualitative test for identifying alcohols, i.e, the Lucas test. HCl with ZnCl 2 catalyst are used. 3 alcohols react quickly, 2 slowly, and 1 don’t react. Note that 1 and 2 alcohols will react with special reagents to produce alkyl halides (i.e., thionyl chloride, SOCl2, or PBr3) ...
An efficient acetylation of dextran using in situ activated acetic
... Regarding the esterification of polysaccharides, a few methods are reported in the literature, such as using acetyl chloride with pyridine,14 acetic anhydride with pyridine and 4-(dimethylamino)pyridine,15,16 carboxylic acids in situ activated with tosyl chloride,17–19 iminium chloride20 and 1,1’-ca ...
... Regarding the esterification of polysaccharides, a few methods are reported in the literature, such as using acetyl chloride with pyridine,14 acetic anhydride with pyridine and 4-(dimethylamino)pyridine,15,16 carboxylic acids in situ activated with tosyl chloride,17–19 iminium chloride20 and 1,1’-ca ...
Excited State Reactions of Carbonyl Compounds
... In the pp* state the lone pair (n-orbital) on the oxygen remains intact, and there are electronic changes only on the p-face. Although on the basis of first principles one might expect the pp* excited carbonyl to behave similar to an alkene, the fact that the electron distribution in p and p* orbita ...
... In the pp* state the lone pair (n-orbital) on the oxygen remains intact, and there are electronic changes only on the p-face. Although on the basis of first principles one might expect the pp* excited carbonyl to behave similar to an alkene, the fact that the electron distribution in p and p* orbita ...
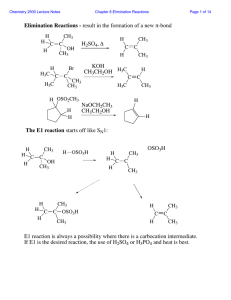
Dehydration of t-Amyl Alcohol (2-Methyl-2
... In the presence of a strong acid, alcohols will protonate. This turns a bad leaving group (hydroxide) into a good leaving group (water). After protonation of a tertiary alcohol, a water molecule leaves with a pair of electrons creating a carbocation. H+ ...
... In the presence of a strong acid, alcohols will protonate. This turns a bad leaving group (hydroxide) into a good leaving group (water). After protonation of a tertiary alcohol, a water molecule leaves with a pair of electrons creating a carbocation. H+ ...
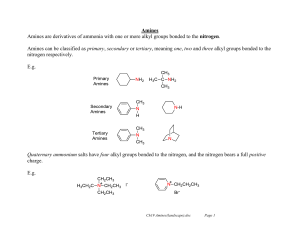
Amines Amines are derivatives of ammonia with one or more alkyl
... The reaction of amines with nitrous acid (HNO2) is a very useful synthetic reaction. Nitrous acid is unstable and needs to be generated in situ by reaction of sodium nitrite and hydrochloric acid. Na+ -O-N=O + HCl H-O-N=O + Na+ClIn very acidic media, nitrous acid can become protonated and lose wat ...
... The reaction of amines with nitrous acid (HNO2) is a very useful synthetic reaction. Nitrous acid is unstable and needs to be generated in situ by reaction of sodium nitrite and hydrochloric acid. Na+ -O-N=O + HCl H-O-N=O + Na+ClIn very acidic media, nitrous acid can become protonated and lose wat ...
Compounds with Oxygen Atoms
... Compounds of phenol are the active ingredients in the essential oils of nutmeg, thyme, cloves, and vanilla. ...
... Compounds of phenol are the active ingredients in the essential oils of nutmeg, thyme, cloves, and vanilla. ...
Handout 3
... attached to the carbon with the most hydrogens. Negatively charged ion is considered a nucleophile. However, neutral H2O molecule (or ROH) may react as a nucleophile also using lone pair’s electrons of oxygen atom. Addition of X2 or X & OH: anti-addition (trans product). ...
... attached to the carbon with the most hydrogens. Negatively charged ion is considered a nucleophile. However, neutral H2O molecule (or ROH) may react as a nucleophile also using lone pair’s electrons of oxygen atom. Addition of X2 or X & OH: anti-addition (trans product). ...
C 1 hapter
... adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through transition metal complexes. There are several challenges involved in applying chiral catalysts in the hydroboration reaction: the catalytic performance must be excellent, the dev ...
... adduct C*-B. In addition, there are a wide range of unsaturated substrates, which can react with a borane reagent through transition metal complexes. There are several challenges involved in applying chiral catalysts in the hydroboration reaction: the catalytic performance must be excellent, the dev ...
Organic Chemistry
... • Cis‐alkenes have similar higher priority elements or group in the chain on the same side of the molecule (or Z‐isomer i.e. have higher priority elements but not necessarily the same on the same side of the molecule) • Trans‐alkenes have similar higher priority elements or group in the chain on opp ...
... • Cis‐alkenes have similar higher priority elements or group in the chain on the same side of the molecule (or Z‐isomer i.e. have higher priority elements but not necessarily the same on the same side of the molecule) • Trans‐alkenes have similar higher priority elements or group in the chain on opp ...
Amino Acids and Proteins
... 28.1A General Features of α-Amino Acids The 20 amino acids that occur naturally in proteins differ in the identity of the R group bonded to the α carbon. The R group is called the side chain of the amino acid. The simplest amino acid, called glycine, has R = H. All other amino acids (R ñ H) have a s ...
... 28.1A General Features of α-Amino Acids The 20 amino acids that occur naturally in proteins differ in the identity of the R group bonded to the α carbon. The R group is called the side chain of the amino acid. The simplest amino acid, called glycine, has R = H. All other amino acids (R ñ H) have a s ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.