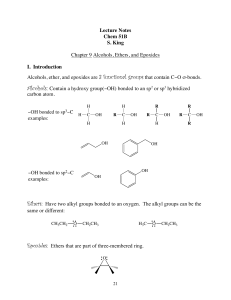
Lecture Notes Chem 51B S. King Chapter 9 Alcohols, Ethers, and
... alcohols go undergo an E2 reaction. All three (1°, 2° and 3°) have the same first step, protonation of the hydroxyl group by the strong acid to make it a better LG: ...
... alcohols go undergo an E2 reaction. All three (1°, 2° and 3°) have the same first step, protonation of the hydroxyl group by the strong acid to make it a better LG: ...
Chapter 12 Organic Compounds with Oxygen and Sulfur
... Stereoisomers have identical molecular formulas, but they are not structural isomers. In stereoisomers, the atoms are bonded in the same sequence but differ in the way they are arranged in space. When stereoisomers have mirror images that are different, they are said to have “handedness.” ...
... Stereoisomers have identical molecular formulas, but they are not structural isomers. In stereoisomers, the atoms are bonded in the same sequence but differ in the way they are arranged in space. When stereoisomers have mirror images that are different, they are said to have “handedness.” ...
Lithium Iodide Original Commentary - Groupe Charette
... LiI as an Additive for Organometallic-mediated Transformations. Diastereoselectivity in the cyclization of 5-hexenyllithiums was shown to be influenced by LiI.34 As an additive in the reduction of α,β-unsaturated ketones by Bu2 SnH2 /Bu2 SnF2 , LiI has a dramatic effect on the selectivity for 1,2- ve ...
... LiI as an Additive for Organometallic-mediated Transformations. Diastereoselectivity in the cyclization of 5-hexenyllithiums was shown to be influenced by LiI.34 As an additive in the reduction of α,β-unsaturated ketones by Bu2 SnH2 /Bu2 SnF2 , LiI has a dramatic effect on the selectivity for 1,2- ve ...
Aldehydes, Ketones, & Chiral Molecules
... The negative part of the added molecule bonds to the positive carbonyl carbon. The positive part of the added molecule bonds to the negative carbonyl oxygen. | + + —C=O + X—Y ...
... The negative part of the added molecule bonds to the positive carbonyl carbon. The positive part of the added molecule bonds to the negative carbonyl oxygen. | + + —C=O + X—Y ...
Elimination Reactions
... • E1 reactions occur under essentially neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 ...
... • E1 reactions occur under essentially neutral conditions with polar solvents, such as water, ethyl alcohol or acetic acid. • E1 reactions can also occur with strong bases, but only at low concentration, about 0.01 to 0.1 M or below. • E2 reactions require strong base in high concentration, about 1 ...
Part I Carbohydrate Auxiliaries - Wiley-VCH
... the stereocenter stemming from the amino aldehyde part. The diastereoselectivities were 96% and 82% de, respectively, indicating only a small matched/mismatched effect between carbohydrate auxiliary and the stereocenters from the amino-aldehyde substrates. The bis-hydrochlorides 17a and 17b were obt ...
... the stereocenter stemming from the amino aldehyde part. The diastereoselectivities were 96% and 82% de, respectively, indicating only a small matched/mismatched effect between carbohydrate auxiliary and the stereocenters from the amino-aldehyde substrates. The bis-hydrochlorides 17a and 17b were obt ...
THE FRIEDEL-CRAFTS BENZYLATION OF ARENES AND THE
... was the selective activation of a N-methyl hydroxamic acid leaving group with BF3•OEt2; an activation most likely proceeding in a reversible manner that avoids build up of highly reactive carbocations. This new Friedel-Crafts approach is therefore cleaner, more selective, and more easily executed th ...
... was the selective activation of a N-methyl hydroxamic acid leaving group with BF3•OEt2; an activation most likely proceeding in a reversible manner that avoids build up of highly reactive carbocations. This new Friedel-Crafts approach is therefore cleaner, more selective, and more easily executed th ...
Preparation and reactions of some lower tungsten halides and
... foam), resulted from the interference of the tungsten mineral in tin ores in the smelting of tin by slagging or eating up the tin. ...
... foam), resulted from the interference of the tungsten mineral in tin ores in the smelting of tin by slagging or eating up the tin. ...
Chiral Enolate Equivalents
... necessarily regarded as such. The term ‘equivalent’ was mostly reserved for closely related reactions such as those involving chiral metallated enamines or hydrazones.5 For example, while the addition of allylic metals to aldehydes (Eq. 1) was immediately recognized as a close relative of the aldol ...
... necessarily regarded as such. The term ‘equivalent’ was mostly reserved for closely related reactions such as those involving chiral metallated enamines or hydrazones.5 For example, while the addition of allylic metals to aldehydes (Eq. 1) was immediately recognized as a close relative of the aldol ...
Lorell Thesis Final Version in PDF S
... overall. To my parents José and Betzaida, who always supported me in all aspects of life and made me what I am today. To my brother, José and sisters, Cheryl and Lorna for being there for me and giving me the unconditional love that only brothers can give. To my parents in law, Hilda and Luis, for a ...
... overall. To my parents José and Betzaida, who always supported me in all aspects of life and made me what I am today. To my brother, José and sisters, Cheryl and Lorna for being there for me and giving me the unconditional love that only brothers can give. To my parents in law, Hilda and Luis, for a ...
Ethylene glycol is a major constituent of motor vehicle antifreeze
... glycolates. These are then hydrogenated to glycol in the presence of a catalyst containing copper and magnesium oxides at temperatures of 125-3250C and pressure above 100 atm. Ethylene glycol is recovered from the reaction mixture by fractional distillation. III. Ethylene glycol has been produced in ...
... glycolates. These are then hydrogenated to glycol in the presence of a catalyst containing copper and magnesium oxides at temperatures of 125-3250C and pressure above 100 atm. Ethylene glycol is recovered from the reaction mixture by fractional distillation. III. Ethylene glycol has been produced in ...
Chapter 4 Alcohols and Alkyl Halides
... (1) Thionyl Chloride: We can treat alcohols with boiling thionyl chloride (SOCl2) to convert the alcohol to an alkyl chloride. This method is considerably milder than using concentrated hydrochloric acid and is useful when the molecule contains sensitive functional groups that would react with the s ...
... (1) Thionyl Chloride: We can treat alcohols with boiling thionyl chloride (SOCl2) to convert the alcohol to an alkyl chloride. This method is considerably milder than using concentrated hydrochloric acid and is useful when the molecule contains sensitive functional groups that would react with the s ...
- University at Albany
... does the acidity. Therefore, acetylenic protons are relatively acidic, with a pKa of ~ 25. Acetylenic protons are more acidic than vinyl (pKa = 44) or alkane (pka = 50) protons. Strong bases such as -NH (but not alkoxide and hydroxide ions) can ...
... does the acidity. Therefore, acetylenic protons are relatively acidic, with a pKa of ~ 25. Acetylenic protons are more acidic than vinyl (pKa = 44) or alkane (pka = 50) protons. Strong bases such as -NH (but not alkoxide and hydroxide ions) can ...
4134gdisk doc..4134gdisk chapter .. Page501
... by [Ru(terpy)(bpy)O]2+ type complexes have been found to be in the same order as the redox potentials, i.e. [Ru(4A-Cl-terpy)(bpy)O]2+ > [Ru(terpy)(bpy)O]2+ > [Ru(terpy)(4,4A-Me2-bpy)O]2+ > [Ru(terpy)(4,4A-EtO-bpy)O]2+.83 There has also been a study of the oxidation of guanines in DNA from calf thymu ...
... by [Ru(terpy)(bpy)O]2+ type complexes have been found to be in the same order as the redox potentials, i.e. [Ru(4A-Cl-terpy)(bpy)O]2+ > [Ru(terpy)(bpy)O]2+ > [Ru(terpy)(4,4A-Me2-bpy)O]2+ > [Ru(terpy)(4,4A-EtO-bpy)O]2+.83 There has also been a study of the oxidation of guanines in DNA from calf thymu ...
2007 Nov Paper 1 - A Level Tuition
... N-methyl-D-aspartic acid has no alcohol group and so, cannot react with ethanoyl chloride to form an ester. (ans) © Step-by-Step ...
... N-methyl-D-aspartic acid has no alcohol group and so, cannot react with ethanoyl chloride to form an ester. (ans) © Step-by-Step ...
On The catalytic Hydrogenation of Co2 and Carboxylic acid esters
... transformation, a new concept of formate-based hydrogen battery was put forward and realized practically on a lab-scale. Beller and co-workers reported on the possibility of a cyclic storage of H2 by CO2 hydrogenation and subsequent H2 release by HCOOH decomposition in a catalytic system comprised o ...
... transformation, a new concept of formate-based hydrogen battery was put forward and realized practically on a lab-scale. Beller and co-workers reported on the possibility of a cyclic storage of H2 by CO2 hydrogenation and subsequent H2 release by HCOOH decomposition in a catalytic system comprised o ...
Project Overview
... William Tam and his wife, Dr. Phillis Chang. Professor William Tam received his B.Sc. at the University of Hong Kong in 1990 and his Ph.D. at the University of Toronto (Canada) in 1995. He was an NSERC postdoctoral fellow at the Imperial College (UK) and at Harvard University (USA). He joined the De ...
... William Tam and his wife, Dr. Phillis Chang. Professor William Tam received his B.Sc. at the University of Hong Kong in 1990 and his Ph.D. at the University of Toronto (Canada) in 1995. He was an NSERC postdoctoral fellow at the Imperial College (UK) and at Harvard University (USA). He joined the De ...
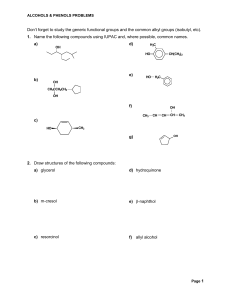
Don`t forget to study the generic functional groups and the common
... 3. Give IUPAC names and, where possible, common names for the following compounds. a) ...
... 3. Give IUPAC names and, where possible, common names for the following compounds. a) ...
Comprehensive Organic Reactions in Aqueous Media
... introduction, pericyclic reactions, nucleophilic additions and substitutions, metal-mediated reactions, transition-metal catalyzed reactions, oxidations and reductions, and industrial applications. At that time, only a limited number of references existed in the literature on this subject, and such ...
... introduction, pericyclic reactions, nucleophilic additions and substitutions, metal-mediated reactions, transition-metal catalyzed reactions, oxidations and reductions, and industrial applications. At that time, only a limited number of references existed in the literature on this subject, and such ...
aa-2005-38-71-negishi - University of Windsor
... alkynyl, allyl, benzyl, propargyl, alkyl, cyano, or enoxy; while the R2 group of R2X can be aryl, alkenyl, alkynyl, allyl, benzyl, propargyl, alkyl, or acyl. Use of other related carbon groups as R1 and/or R2 is not only conceivable, but also known in the literature. Even if only those nine types of ...
... alkynyl, allyl, benzyl, propargyl, alkyl, cyano, or enoxy; while the R2 group of R2X can be aryl, alkenyl, alkynyl, allyl, benzyl, propargyl, alkyl, or acyl. Use of other related carbon groups as R1 and/or R2 is not only conceivable, but also known in the literature. Even if only those nine types of ...
Alcohols, Phenols and Ethers
... AIIMS,CBSE,AIPMT, AFMC,Bio.Tech & PMT, Contact :- 9438559863, Mail at :- [email protected],www.ibiokaare.com,compiled by AKB ...
... AIIMS,CBSE,AIPMT, AFMC,Bio.Tech & PMT, Contact :- 9438559863, Mail at :- [email protected],www.ibiokaare.com,compiled by AKB ...
Transition Metal Reagents and Catalysts
... important types of reactions classi®ed mainly by representative substrates such as organic halides and allylic derivatives are surveyed with pertinent examples. For this purpose, I cited many references; these were selected from a much larger number which I have collected over the years. I wanted to ...
... important types of reactions classi®ed mainly by representative substrates such as organic halides and allylic derivatives are surveyed with pertinent examples. For this purpose, I cited many references; these were selected from a much larger number which I have collected over the years. I wanted to ...
C−C, C−O, C−N Bond Formation on sp2 Carbon by Pd(II)
... mechanisms of these processes have been extensively studied. A typical reaction with alkenes starts with the complexation of the olefin by the Pd(II) salt, as shown in Scheme 1, left-hand side. The resulting π-olefin complex A can undergo an intermolecular or intramolecular nucleophilic attack, usua ...
... mechanisms of these processes have been extensively studied. A typical reaction with alkenes starts with the complexation of the olefin by the Pd(II) salt, as shown in Scheme 1, left-hand side. The resulting π-olefin complex A can undergo an intermolecular or intramolecular nucleophilic attack, usua ...
06. Alcohols. Phenols. Ethers
... are called monohydric alcohols. These are further classified as primary (1'), secondary (2'), and tertiary (3') according as the ОН group is attached to primary, secondary and tertiary carbon atoms respectively. For example: ...
... are called monohydric alcohols. These are further classified as primary (1'), secondary (2'), and tertiary (3') according as the ОН group is attached to primary, secondary and tertiary carbon atoms respectively. For example: ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.