Development of Multi-Component Reactions using Catalytically Generated Allyl Metal Reagents
... definitions are found.3-6 A thorough taxonomy of tandem catalysis has been reviewed by Fogg and co-workers.4 Accordingly, in tandem or domino catalyses, all catalytic species must be present from the outset. When only one catalytic transformation is performed, followed by another stoichiometric tran ...
... definitions are found.3-6 A thorough taxonomy of tandem catalysis has been reviewed by Fogg and co-workers.4 Accordingly, in tandem or domino catalyses, all catalytic species must be present from the outset. When only one catalytic transformation is performed, followed by another stoichiometric tran ...
Full Text - Verlag der Zeitschrift für Naturforschung
... alcohols (steroids, monoterpenoids) or phenols (Ar-O-CO-CH3), and methyl (or ethyl) esters of acids with an aromatic or an aromatic-aliphatic system (Ar-CO-OR, where Ar means either naphthyl or a substituted phenyl ring). The results of these transformations are presented in Table I and II [the yiel ...
... alcohols (steroids, monoterpenoids) or phenols (Ar-O-CO-CH3), and methyl (or ethyl) esters of acids with an aromatic or an aromatic-aliphatic system (Ar-CO-OR, where Ar means either naphthyl or a substituted phenyl ring). The results of these transformations are presented in Table I and II [the yiel ...
Document
... Hydrolysis of amides: This reaction involves breaking the C-N bond at the carbonyl carbon. a. acid hydrolysis: forms “ammonium” ion and carboxylic acid b. alkaline hydrolysis: forms carboxylate ion and amine ...
... Hydrolysis of amides: This reaction involves breaking the C-N bond at the carbonyl carbon. a. acid hydrolysis: forms “ammonium” ion and carboxylic acid b. alkaline hydrolysis: forms carboxylate ion and amine ...
Colorimetric Assay of Alditols in Complex Biological Samples
... The formation of color was completed in 1 min at 100 °C and bleaching began after 3 min at that temperature. Thus, 2 min in boiling water was chosen as the optimal condition for the color reaction. On the other hand, changing the pH of the reaction medium from 2.0 to 7.0 had only a minor effect in t ...
... The formation of color was completed in 1 min at 100 °C and bleaching began after 3 min at that temperature. Thus, 2 min in boiling water was chosen as the optimal condition for the color reaction. On the other hand, changing the pH of the reaction medium from 2.0 to 7.0 had only a minor effect in t ...
FULL PAPER Observations on the Influence of Precursor
... chemical yield and concentration was also addressed lately.10 In order to harvest the potential of macrocycles for screening campaigns, one has to face the challenges posed by this compound class: the chemistry of cyclization is also the chemistry of oligomerization, which often compels that high di ...
... chemical yield and concentration was also addressed lately.10 In order to harvest the potential of macrocycles for screening campaigns, one has to face the challenges posed by this compound class: the chemistry of cyclization is also the chemistry of oligomerization, which often compels that high di ...
Organic Chemistry Notes by Jim Maxka jim.maxka
... The Grignard reacts with a carbonyl compound just like the hydride. We must keep acid away from Grignards. Finish the reaction of CH3CH2-MgBr + H-OH Æ Predict the direction and extent of the reaction. A general Grignard reaction away from water is: ...
... The Grignard reacts with a carbonyl compound just like the hydride. We must keep acid away from Grignards. Finish the reaction of CH3CH2-MgBr + H-OH Æ Predict the direction and extent of the reaction. A general Grignard reaction away from water is: ...
Organic Chemistry 145 CHEM
... - Bromine adds as follows; In the first step, the addition occurs mainly trans. ...
... - Bromine adds as follows; In the first step, the addition occurs mainly trans. ...
New Exp8
... Limitations of E1 Reaction: Acid-Catalyzed Dehydrations Competition can occur with SN1 reaction if reaction conditions are not ‘controlled’ (when protic solvents, non-basic nucleophiles are used). Mixtures of products form with the E1 reaction (also SN1). Unsymmetrical reagents and rearrangements po ...
... Limitations of E1 Reaction: Acid-Catalyzed Dehydrations Competition can occur with SN1 reaction if reaction conditions are not ‘controlled’ (when protic solvents, non-basic nucleophiles are used). Mixtures of products form with the E1 reaction (also SN1). Unsymmetrical reagents and rearrangements po ...
An Epoxidation Reaction: The Epoxidation of Cholesterol to 5 ,6
... Esters are acid derivatives and contain a carbonyl group; whereas, ethers are water or alcohol derivatives and do not contain a carbonyl group. Alcohols are named by finding the longest carbon chain to which the OH group is bonded and naming the alcohol accordingly. Ethers have two R groups, which m ...
... Esters are acid derivatives and contain a carbonyl group; whereas, ethers are water or alcohol derivatives and do not contain a carbonyl group. Alcohols are named by finding the longest carbon chain to which the OH group is bonded and naming the alcohol accordingly. Ethers have two R groups, which m ...
Alcohols, Diols, and Thiols
... We’ll start by discussing in more detail a class of compounds already familiar to us, alcohols. Alcohols were introduced in Chapter 4 and have appeared regularly since then. With this chapter we extend our knowledge of alcohols, particularly with respect to their relationship to carbonyl-containing ...
... We’ll start by discussing in more detail a class of compounds already familiar to us, alcohols. Alcohols were introduced in Chapter 4 and have appeared regularly since then. With this chapter we extend our knowledge of alcohols, particularly with respect to their relationship to carbonyl-containing ...
Chpt 23Final7e
... of alanine (a) How do you account for the fact that the -NH3 + group of the conjugate acid of alanine is a stronger acid than the -NH3 + group of the conjugate acid of isopropylamine? The electron-withdrawing properties of the carboxyl group adjacent to the amine of alanine make the conjugate acid o ...
... of alanine (a) How do you account for the fact that the -NH3 + group of the conjugate acid of alanine is a stronger acid than the -NH3 + group of the conjugate acid of isopropylamine? The electron-withdrawing properties of the carboxyl group adjacent to the amine of alanine make the conjugate acid o ...
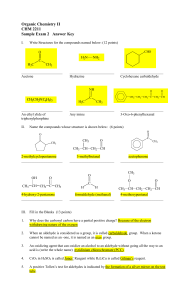
Organic Chemistry II CHM 2211 Sample Exam 2 Answer Key
... Why does the carbonyl carbon have a partial positive charge? Because of the electron withdrawing nature of the oxygen ...
... Why does the carbonyl carbon have a partial positive charge? Because of the electron withdrawing nature of the oxygen ...
ETHERS
... ROH + R'OH )))))))))> ROR + ROR' + R'OR' + H2O However, if one alcohol is t-BuOH, t-Bu-O-R will be the major product owing to rapid formation of t-Bu+ carbocation and inability to form t-Bu-O-t-Bu. ...
... ROH + R'OH )))))))))> ROR + ROR' + R'OR' + H2O However, if one alcohol is t-BuOH, t-Bu-O-R will be the major product owing to rapid formation of t-Bu+ carbocation and inability to form t-Bu-O-t-Bu. ...
Chapter 12. Aldehydes, Ketones and Carboxylic Acids
... [Fehling solution is a mixture of Fehling solution A and Fehling solution B in 1: 1 ratio. Fehling solution A is aqueous copper sulphate and Fehling solution B is alkaline sodium potassium tartrate which is also called, Rochelle salt.] (c) Benedict solution With it, aldehydes (except benzaldehyde) a ...
... [Fehling solution is a mixture of Fehling solution A and Fehling solution B in 1: 1 ratio. Fehling solution A is aqueous copper sulphate and Fehling solution B is alkaline sodium potassium tartrate which is also called, Rochelle salt.] (c) Benedict solution With it, aldehydes (except benzaldehyde) a ...
Practice Problem - HCC Southeast Commons
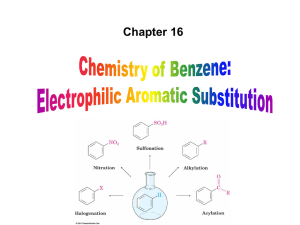
... Three rules for the additive effects of two different groups: 1. If the directing effects of the two groups are the same, the result is additive 2. If the directing effects of two groups oppose each other, the more powerful activating group determines the principal outcome 3. The position between th ...
... Three rules for the additive effects of two different groups: 1. If the directing effects of the two groups are the same, the result is additive 2. If the directing effects of two groups oppose each other, the more powerful activating group determines the principal outcome 3. The position between th ...
Direct organocatalytic enantioselective a-aminomethylation
... Trost and co-workers12 developed di-nuclear zinc organometallic complexes as catalyst for the direct catalytic enantioselective Mannich-type reactions between hydroxyarylketones and preformed imines. In addition, Jørgensen and co-workers have developed elegant direct asymmetric Mannich reactions inv ...
... Trost and co-workers12 developed di-nuclear zinc organometallic complexes as catalyst for the direct catalytic enantioselective Mannich-type reactions between hydroxyarylketones and preformed imines. In addition, Jørgensen and co-workers have developed elegant direct asymmetric Mannich reactions inv ...
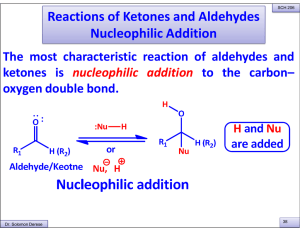
Reactions of Ketones and Aldehydes Nucleophilic Addition
... I. Addition of the organometalics to formaldehyde (CH2=O) forms a 1° alcohol. II. Addition of the organometalics to all other aldehydes forms a 2° alcohol. III.Addition of the organometalics to ketones forms a 3° alcohol. Dr. Solomon Derese ...
... I. Addition of the organometalics to formaldehyde (CH2=O) forms a 1° alcohol. II. Addition of the organometalics to all other aldehydes forms a 2° alcohol. III.Addition of the organometalics to ketones forms a 3° alcohol. Dr. Solomon Derese ...
Ch 10- Alcohols and Ethers
... priority over halogens, double bonds, triple bonds, and alkyl groups in numbering and naming. ...
... priority over halogens, double bonds, triple bonds, and alkyl groups in numbering and naming. ...
Wafer-Level Artificial Photosynthesis for CO2 Reduction into CH4
... mechanism is debatable among researchers6-8. With the input energy from photons in CO2 photoreduction, the bond C=O can be broken to form C-H bonds, which leads to the formation of different fuels such as methane (CH4). H2 formation is a very competitive process in CO2 photochemical and photoelectro ...
... mechanism is debatable among researchers6-8. With the input energy from photons in CO2 photoreduction, the bond C=O can be broken to form C-H bonds, which leads to the formation of different fuels such as methane (CH4). H2 formation is a very competitive process in CO2 photochemical and photoelectro ...
Nucleophilic Substitution on the Carbonyl Group
... The ease with which a leaving group leaves a compound is inversely proportional to its basicity. Thus, the more basic the leaving group, the less readily it leaves. A stronger base is more willing to donate its electron pair to an electrophile that, in this case, is the carbonyl carbon. In the carbo ...
... The ease with which a leaving group leaves a compound is inversely proportional to its basicity. Thus, the more basic the leaving group, the less readily it leaves. A stronger base is more willing to donate its electron pair to an electrophile that, in this case, is the carbonyl carbon. In the carbo ...
Green synthesis of 2-amino-7-hydroxy-4-aryl-4H
... resorcinol, and malononitrile to afford 2-amino-7-hydroxy-4-aryl-4H -chromene-3-carbonitrile derivatives. Comparatively few methods have been described for the synthesis of 2-amino-7-hydroxy-4-aryl/alkyl-4H chromene-3-carbonitriles applying the use of potassium phthalimide-N-oxyl, 18 2,2,2-trifluoro ...
... resorcinol, and malononitrile to afford 2-amino-7-hydroxy-4-aryl-4H -chromene-3-carbonitrile derivatives. Comparatively few methods have been described for the synthesis of 2-amino-7-hydroxy-4-aryl/alkyl-4H chromene-3-carbonitriles applying the use of potassium phthalimide-N-oxyl, 18 2,2,2-trifluoro ...
Year 13 rings polymers and Analysis past papers 1
... Predict the m/z values of the two most abundant fragments in the mass spectrum of pentan-2-one. Fragment 1 ............................................................................................................. Fragment 2 ........................................................................ ...
... Predict the m/z values of the two most abundant fragments in the mass spectrum of pentan-2-one. Fragment 1 ............................................................................................................. Fragment 2 ........................................................................ ...
Anaysis exam questions
... Predict the m/z values of the two most abundant fragments in the mass spectrum of pentan-2-one. Fragment 1 ............................................................................................................. Fragment 2 ........................................................................ ...
... Predict the m/z values of the two most abundant fragments in the mass spectrum of pentan-2-one. Fragment 1 ............................................................................................................. Fragment 2 ........................................................................ ...
Document
... other cases we were unable to isolate the corresponding ester in any detectable amount. Of course, iodosobenzene was quantitively reduced to iodobenzene. A plausible mechanism of this IOB mediated conversion of 1 to 2, outlined in Scheme 2, is analogous to that suggested by Kita et al. for the oxida ...
... other cases we were unable to isolate the corresponding ester in any detectable amount. Of course, iodosobenzene was quantitively reduced to iodobenzene. A plausible mechanism of this IOB mediated conversion of 1 to 2, outlined in Scheme 2, is analogous to that suggested by Kita et al. for the oxida ...
molecules Palladium and Organocatalysis: An Excellent Recipe for Asymmetric Synthesis
... a variety of precursors and protocols [31]. Clearly, the generation of tertiary carbon stereocenters by using the very small proton atom is a very interesting and attractive process; however, it is also very challenging, because it normally involves undesired racemization reactions. The enantioselec ...
... a variety of precursors and protocols [31]. Clearly, the generation of tertiary carbon stereocenters by using the very small proton atom is a very interesting and attractive process; however, it is also very challenging, because it normally involves undesired racemization reactions. The enantioselec ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.