Process for preparing polycarbonates
... then cooled to 130° C. and 30 grams of ?nely divided 60 The reaction conditions of Examples 1 and 2 were magnesium silicate and 60 grams of celite clay were again duplicated except that a mixture of 44 grams (0.5 added to purify the mixture. The mixture was continu moles) of ethylene carbonate, 6.3 ...
... then cooled to 130° C. and 30 grams of ?nely divided 60 The reaction conditions of Examples 1 and 2 were magnesium silicate and 60 grams of celite clay were again duplicated except that a mixture of 44 grams (0.5 added to purify the mixture. The mixture was continu moles) of ethylene carbonate, 6.3 ...
Electrophilic Additions: Alkenes Addition of Hydrogen Halides
... In a regioselective reaction, one constitutional isomer is the major or the only product. I: early transition state (Like reactants) ...
... In a regioselective reaction, one constitutional isomer is the major or the only product. I: early transition state (Like reactants) ...
9: Formation of Alkenes and Alkynes. Elimination Reactions
... C in substitution reactions (and form C-Nu bonds) react with C-H protons to form Nu-H bonds. That is why we use the term "base" to describe nucleophiles that also remove protons from carbon in elimination reactions. E1 and E2 Reactions Can Compete (9.2B) E1 and E2 reactions may occur simultaneously ...
... C in substitution reactions (and form C-Nu bonds) react with C-H protons to form Nu-H bonds. That is why we use the term "base" to describe nucleophiles that also remove protons from carbon in elimination reactions. E1 and E2 Reactions Can Compete (9.2B) E1 and E2 reactions may occur simultaneously ...
Pincer Complexes. Applications in Catalysis
... employed in the activation of C-H bonds of several substrates (vide supra) [21]; probably the most notable case being the ...
... employed in the activation of C-H bonds of several substrates (vide supra) [21]; probably the most notable case being the ...
Chapter 2 Phenols
... 5- Reactions of Phenols A hydroxyl group is very powerful activating substituent, and electrophilic aromatic substitution in phenol occurs far faster, and under milder condition, than in benzene. ...
... 5- Reactions of Phenols A hydroxyl group is very powerful activating substituent, and electrophilic aromatic substitution in phenol occurs far faster, and under milder condition, than in benzene. ...
Chapter 9
... • The most common reaction of aromatic compounds • This reaction is characteristic of all aromatic rings ...
... • The most common reaction of aromatic compounds • This reaction is characteristic of all aromatic rings ...
Palladium-Catalyzed Synthesis and Transformation of Organoboranes
... important class of organic reagents because of the high stereoselectivity of the coupling reactions with electrophiles. However, these compounds are relatively unstable and therefore preparation of functionalized allylboranes is often a challenging synthetic task. This thesis is focused on the devel ...
... important class of organic reagents because of the high stereoselectivity of the coupling reactions with electrophiles. However, these compounds are relatively unstable and therefore preparation of functionalized allylboranes is often a challenging synthetic task. This thesis is focused on the devel ...
CHM 235 Course Outline and Homework in McMurry (6th ed.)
... Bond dissociation energies (Ho = energy used to break bonds–energy gained by making bonds) Energy diagrams (reaction coordinates, transition states, reaction intermediates, RDS) ...
... Bond dissociation energies (Ho = energy used to break bonds–energy gained by making bonds) Energy diagrams (reaction coordinates, transition states, reaction intermediates, RDS) ...
Chapter 15
... more substituted – and more stable – carbocation as predicted by Markovnikov’s rule (the proton adds to the carbon with the most H’s). In the second step water attacks the carbocation and in a third, fast step the extra proton on the water is transferred to another molecule of water to regenerate th ...
... more substituted – and more stable – carbocation as predicted by Markovnikov’s rule (the proton adds to the carbon with the most H’s). In the second step water attacks the carbocation and in a third, fast step the extra proton on the water is transferred to another molecule of water to regenerate th ...
Chapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions
... Canizzaro Reaction Enzymes catalyze the reduction of aldehydes and ketones using NADH as the source of the equivalent of H The transfer resembles that in the Cannizzaro reaction but the ...
... Canizzaro Reaction Enzymes catalyze the reduction of aldehydes and ketones using NADH as the source of the equivalent of H The transfer resembles that in the Cannizzaro reaction but the ...
Chapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions
... Canizzaro Reaction Enzymes catalyze the reduction of aldehydes and ketones using NADH as the source of the equivalent of H The transfer resembles that in the Cannizzaro reaction but the ...
... Canizzaro Reaction Enzymes catalyze the reduction of aldehydes and ketones using NADH as the source of the equivalent of H The transfer resembles that in the Cannizzaro reaction but the ...
Nuggets of Knowledge for Chapter 10 – Alkyl Halides II Chem 2310 I
... ◦ Electronegativity: Atoms that are less electronegative are stronger nucleophiles and stronger bases. This is because their electrons are not attracted to the atom as strongly, and are easier to donate. In the case of negative nucleophiles and bases, they are also more reactive because it is harder ...
... ◦ Electronegativity: Atoms that are less electronegative are stronger nucleophiles and stronger bases. This is because their electrons are not attracted to the atom as strongly, and are easier to donate. In the case of negative nucleophiles and bases, they are also more reactive because it is harder ...
Novel Synthesis of Schiff bases Bearing Glucosamine Moiety
... Mannich-Michael13. Also Schiff bases derived amino sugars with salicylaldehyde have been shown to give stable complexes with transition-metal ions such as Cu (II), Fe (III), and Co (II) and can be applied as asymmetric catalysts14. On the other hand Pérez et al. have reported the first detailed stud ...
... Mannich-Michael13. Also Schiff bases derived amino sugars with salicylaldehyde have been shown to give stable complexes with transition-metal ions such as Cu (II), Fe (III), and Co (II) and can be applied as asymmetric catalysts14. On the other hand Pérez et al. have reported the first detailed stud ...
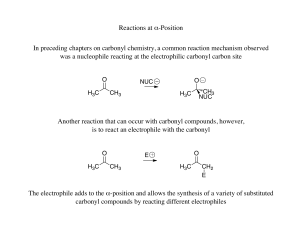
Reactions at α-Position In preceding chapters on carbonyl chemistry
... While reacting two different ketones with alkoxide base is impractical due to the variety of products obtained, the desired product would only be obtained in low yield after a difficult separation, there are methods to react two different carbonyls in an aldol reaction efficiently ...
... While reacting two different ketones with alkoxide base is impractical due to the variety of products obtained, the desired product would only be obtained in low yield after a difficult separation, there are methods to react two different carbonyls in an aldol reaction efficiently ...
Chem 33 Lab - Santa Clara University
... bp will only correspond to literature values if the compound is pure and these values are measured accurately. Purity should be determined by TLC for solids and GC for liquids. High-boiling liquids can be analyzed by TLC as well. For synthesis experiments, you should calculate the percent yield of y ...
... bp will only correspond to literature values if the compound is pure and these values are measured accurately. Purity should be determined by TLC for solids and GC for liquids. High-boiling liquids can be analyzed by TLC as well. For synthesis experiments, you should calculate the percent yield of y ...
Acidity of Alcohols
... If the groups attached to the double bond are different, we distinguish the two isomers by adding the prefix Z (same side) or E (opposite sides) depending on the atomic number of the atoms attached to each end of the double bond ...
... If the groups attached to the double bond are different, we distinguish the two isomers by adding the prefix Z (same side) or E (opposite sides) depending on the atomic number of the atoms attached to each end of the double bond ...
Latest Publication (still not complete)
... observed reaction pathways and hence is crucial to the total understanding of the chemistry of chromium-pentacarbonyl carbene complexes. Fischer carbene complexes exhibit two characteristic features that are important in understanding their respective chemistry. The first of these features is the fa ...
... observed reaction pathways and hence is crucial to the total understanding of the chemistry of chromium-pentacarbonyl carbene complexes. Fischer carbene complexes exhibit two characteristic features that are important in understanding their respective chemistry. The first of these features is the fa ...
13-Elimination Reactions
... the SN1 and SN2 reaction mechanisms, which are covered in Chapter 12. The E1 reaction mechanism is a two-step process that, as with the SN1 mechanism, usually loses all the stereochemical information of the substrate as the reaction proceeds. The E2 mechanism, similar to the SN2 mechanism, is a conc ...
... the SN1 and SN2 reaction mechanisms, which are covered in Chapter 12. The E1 reaction mechanism is a two-step process that, as with the SN1 mechanism, usually loses all the stereochemical information of the substrate as the reaction proceeds. The E2 mechanism, similar to the SN2 mechanism, is a conc ...
General and Selective Synthesis of (Z)-3
... (Z)- and (E)-isomers was obtained by decreasing the amount of MeOH to 0.2 mL (entry 3).9 Encouraged by the results, we then examined a series of solvents (entries 3-6), and DCE (1,2-dichloroethane) was the most effective (entry 7). In the presence of 5 mol % of PdBr2 and 5 equiv of CuBr2, treatment ...
... (Z)- and (E)-isomers was obtained by decreasing the amount of MeOH to 0.2 mL (entry 3).9 Encouraged by the results, we then examined a series of solvents (entries 3-6), and DCE (1,2-dichloroethane) was the most effective (entry 7). In the presence of 5 mol % of PdBr2 and 5 equiv of CuBr2, treatment ...
f8560d95306293b
... and reeducing agents. Cleavage takes place under quite vigorous conditions ...
... and reeducing agents. Cleavage takes place under quite vigorous conditions ...
Cl3CCN/PPh3 and CBr4/PPh3: two efficient reagent systems for the
... Br3CCOCBr3 were promising candidates in terms of new brominating agents for N-heteroaromatic hydroxy compounds, affording 2-bromopyridine in 15% and 45% yield, respectively (entries 2 and 3). Although, the use of Br3CCOCBr3 gave rise to the bromide in higher yield than CBr4, several by-products were ...
... Br3CCOCBr3 were promising candidates in terms of new brominating agents for N-heteroaromatic hydroxy compounds, affording 2-bromopyridine in 15% and 45% yield, respectively (entries 2 and 3). Although, the use of Br3CCOCBr3 gave rise to the bromide in higher yield than CBr4, several by-products were ...
Learning materials
... Nucleophilicity is the ability to donate a pair of electrons to a non-hydrogen atom. Nucleophilicity depends on the ability of the nucleophile to donate a pair of electrons to H+ (basicity) and on the influence on such ability from the substrate (usually polarizability). Polarizability is characte ...
... Nucleophilicity is the ability to donate a pair of electrons to a non-hydrogen atom. Nucleophilicity depends on the ability of the nucleophile to donate a pair of electrons to H+ (basicity) and on the influence on such ability from the substrate (usually polarizability). Polarizability is characte ...
Selective Oxidation Reactions of Natural Compounds with
... 1-Phenanthrenecarboxylic acid, 1,2,3,4,4a,4b,5,6,8a,9,10,10a-dodecahydro-1,4a-dimethyl-7-(1methylethyl)-9-oxo-, methyl ester, [1R,(1α,4αβ,4βα,8αβ,10αα)] (8). This compound has been identified on the basis of the comparison of its 1H- and 13C-NMR, EIMS, and IR data with those reported in the literatu ...
... 1-Phenanthrenecarboxylic acid, 1,2,3,4,4a,4b,5,6,8a,9,10,10a-dodecahydro-1,4a-dimethyl-7-(1methylethyl)-9-oxo-, methyl ester, [1R,(1α,4αβ,4βα,8αβ,10αα)] (8). This compound has been identified on the basis of the comparison of its 1H- and 13C-NMR, EIMS, and IR data with those reported in the literatu ...
Aromatic Compounds
... • Nucleophilic substitutions are favored by electronwithdrawing substituents which stabilize a carbanion intermediate • Electron-withdrawing groups that deactivate rings for electrophilic substitution (nitro, carbonyl, cyano, and so on) activate rings for nucleophilic substitution ...
... • Nucleophilic substitutions are favored by electronwithdrawing substituents which stabilize a carbanion intermediate • Electron-withdrawing groups that deactivate rings for electrophilic substitution (nitro, carbonyl, cyano, and so on) activate rings for nucleophilic substitution ...
Chloroperbenzoic_aci..
... Epoxidations of alkenes with m-CPBA are usually carried out by mixing the reactants in CH2 Cl2 or CHCl3 at 0–25 ◦ C.9 After the reaction is complete the reaction mixture is cooled in an ice bath and the precipitated m-chlorobenzoic acid is removed by filtration. The organic layer is washed with sodi ...
... Epoxidations of alkenes with m-CPBA are usually carried out by mixing the reactants in CH2 Cl2 or CHCl3 at 0–25 ◦ C.9 After the reaction is complete the reaction mixture is cooled in an ice bath and the precipitated m-chlorobenzoic acid is removed by filtration. The organic layer is washed with sodi ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.