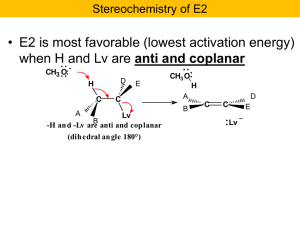
Nucleophilic Substitution and b
... Here is the crux of the matter: how can the non-reacting carbon change its configuration??? Further it does not always change but only if configuration of the reacting carbon changes!! We got a mixture of enantiomers, a racemic mixture. Something strange is happening!! Expect sulfur to attack the C- ...
... Here is the crux of the matter: how can the non-reacting carbon change its configuration??? Further it does not always change but only if configuration of the reacting carbon changes!! We got a mixture of enantiomers, a racemic mixture. Something strange is happening!! Expect sulfur to attack the C- ...
Cerium(IV) Ammonium Nitrate as a Catalyst in
... José Carlos Menéndez was born in Madrid (1960) and obtained degrees in Pharmacy from Universidad Complutense at Madrid, UCM (1982) and Chemistry from UNED (1985), followed by a Ph.D. in Pharmacy from UCM in 1988, under the supervision of Dr. Mónica M. Söllhuber. In August 1988, he joined the gro ...
... José Carlos Menéndez was born in Madrid (1960) and obtained degrees in Pharmacy from Universidad Complutense at Madrid, UCM (1982) and Chemistry from UNED (1985), followed by a Ph.D. in Pharmacy from UCM in 1988, under the supervision of Dr. Mónica M. Söllhuber. In August 1988, he joined the gro ...
EXPERIMENT 2 Properties of Alkanes, Alkenes, and Alcohols
... Boiling Point. The bp of a compound depends on the strength of the intermolecular forces between individual molecules of the compound. Compounds that can form hydrogen bonds between their molecules have higher bps than those that cannot. Non-polar compounds, such as alkanes and alkenes, have relativ ...
... Boiling Point. The bp of a compound depends on the strength of the intermolecular forces between individual molecules of the compound. Compounds that can form hydrogen bonds between their molecules have higher bps than those that cannot. Non-polar compounds, such as alkanes and alkenes, have relativ ...
- Sacramento - California State University
... enantiomeric excess for asymmetric epoxidations was only 25% ee. Since epoxides are useful building blocks for synthesis, further progress towards higher enantioselective epoxidations was required. Before Sharpless began work on the more popular titanium catalyzed epoxidations8, he synthesized hydro ...
... enantiomeric excess for asymmetric epoxidations was only 25% ee. Since epoxides are useful building blocks for synthesis, further progress towards higher enantioselective epoxidations was required. Before Sharpless began work on the more popular titanium catalyzed epoxidations8, he synthesized hydro ...
Alcohols, Phenols and Ethers
... 38. Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation. 39. How can propan-2-one be converted into tert- butyl alcohol? ...
... 38. Name the enzymes and write the reactions involved in the preparation of ethanol from sucrose by fermentation. 39. How can propan-2-one be converted into tert- butyl alcohol? ...
Mechanism of intra - Chemical Engineering Labs
... occur much faster than the initial alkanal turnovers and lead to diverse methyl- or ethyl-substituted aromatics (e.g., trimethylbenzene, methyl-ethylbenzene) and light gases (e.g., CO, CO2, C1–C3 hydrocarbons). The primary condensation products were detected on H-MFI at these higher temperatures onl ...
... occur much faster than the initial alkanal turnovers and lead to diverse methyl- or ethyl-substituted aromatics (e.g., trimethylbenzene, methyl-ethylbenzene) and light gases (e.g., CO, CO2, C1–C3 hydrocarbons). The primary condensation products were detected on H-MFI at these higher temperatures onl ...
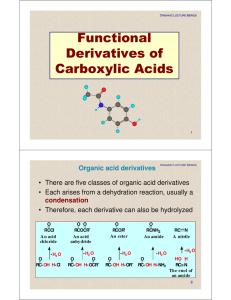
Functional Derivatives of Carboxylic Acids
... • Step 3: Make a new bond between a nucleophile and an electrophile. The ketone reacts with a second mole of Grignard reagent to give a second tetrahedral carbonyl addition intermediate. ...
... • Step 3: Make a new bond between a nucleophile and an electrophile. The ketone reacts with a second mole of Grignard reagent to give a second tetrahedral carbonyl addition intermediate. ...
Dehydration of ROH
... depends primarily on the degree of stabilization and solvation of the alkoxide ion. • The negatively charged oxygens of methoxide and ethoxide are about as accessible as the oxygen of hydroxide ion for solvation; these alcohol are about as acidic as water. • As the bulk of the alkyl group increases, ...
... depends primarily on the degree of stabilization and solvation of the alkoxide ion. • The negatively charged oxygens of methoxide and ethoxide are about as accessible as the oxygen of hydroxide ion for solvation; these alcohol are about as acidic as water. • As the bulk of the alkyl group increases, ...
Chapter 10
... Since 2˚ carbocations are more stable than 1˚, 2-chloropropane is the only product obtained ...
... Since 2˚ carbocations are more stable than 1˚, 2-chloropropane is the only product obtained ...
Ch04-04-alkenes-2
... Exergonic reaction: early transition state resembles reactants (I). Endergonic reaction: late transition state resembles products (II). ...
... Exergonic reaction: early transition state resembles reactants (I). Endergonic reaction: late transition state resembles products (II). ...
Group D
... CuCl dissolved in ammonia will give a rusty brown precipitate with terminal alkynes like but-1yne but shows no visible reaction with butadiene. (A similar reaction is that with aq.Ag(NH3)2+ but this gives a white precipitate with but-1-yne and nothing with buta-1,3-diene.) ...
... CuCl dissolved in ammonia will give a rusty brown precipitate with terminal alkynes like but-1yne but shows no visible reaction with butadiene. (A similar reaction is that with aq.Ag(NH3)2+ but this gives a white precipitate with but-1-yne and nothing with buta-1,3-diene.) ...
Chemistry 209 - Experiment 3, Spring 2003
... Place approx. 0.2 mL (4-5 drops) of the liquid or approx. 0.1 g (1 small spatula tip) of the solid to be tested, in a test tube and add a total of approx. 3 mL of the solvent (either distilled water or hexane) in several portions. Cover the tube with a cork or rubber sleeve stopper and shake it vigo ...
... Place approx. 0.2 mL (4-5 drops) of the liquid or approx. 0.1 g (1 small spatula tip) of the solid to be tested, in a test tube and add a total of approx. 3 mL of the solvent (either distilled water or hexane) in several portions. Cover the tube with a cork or rubber sleeve stopper and shake it vigo ...
Manganese-Catalyzed Epoxidations of Alkenes in
... as phase transfer agents.52-61 Typically, these are formed in situ, but catalytically active complexes such as (R4N)3{PO4(W(O)(O2)2)4} have been isolated and even characterized crystallographically. 62 Noyori showed that terminal aliphatic alkenes could be epoxidized by this system at 90 °C without ...
... as phase transfer agents.52-61 Typically, these are formed in situ, but catalytically active complexes such as (R4N)3{PO4(W(O)(O2)2)4} have been isolated and even characterized crystallographically. 62 Noyori showed that terminal aliphatic alkenes could be epoxidized by this system at 90 °C without ...
Nitrogen Fixation by Transition Metals: A Review
... On the other hand, micro bacteria fix molecular nitrogen using nitrogenase enzymes, which under ambient temperature and pressures. Three types of nitrogenases are known, which are Molybdenum (Mo), Vanadium (V) & Iron (Fe) nitrogenases. Micro bacteria mediate nitrogen fixation with the help of these ...
... On the other hand, micro bacteria fix molecular nitrogen using nitrogenase enzymes, which under ambient temperature and pressures. Three types of nitrogenases are known, which are Molybdenum (Mo), Vanadium (V) & Iron (Fe) nitrogenases. Micro bacteria mediate nitrogen fixation with the help of these ...
synthesis, chemistry and optical resol
... arrangement of the two rings was found to block additions to the buried double bond, resulting in dramatic reactivity differences between the trans (13, n = 12) and cis (12, n = 12) i~omers.l-~ We now wish to record the synthesis of [20.10]-, [22.10]-, and [26.10]betweenanenes (13a-c) by a new route ...
... arrangement of the two rings was found to block additions to the buried double bond, resulting in dramatic reactivity differences between the trans (13, n = 12) and cis (12, n = 12) i~omers.l-~ We now wish to record the synthesis of [20.10]-, [22.10]-, and [26.10]betweenanenes (13a-c) by a new route ...
Copper-catalysed selective hydroamination reactions of alkynes Please share
... The proposed mechanisms for the formation of enamines and alkylamines (shown in Fig. 3a) both commence with syn-selective Cu–H addition to the alkyne substrate to give vinylcopper species 11. In the absence of a proton source (alcohol), direct interception by electrophilic amine 2 (likely via oxidat ...
... The proposed mechanisms for the formation of enamines and alkylamines (shown in Fig. 3a) both commence with syn-selective Cu–H addition to the alkyne substrate to give vinylcopper species 11. In the absence of a proton source (alcohol), direct interception by electrophilic amine 2 (likely via oxidat ...
T_AllylCF3paperBM[5]
... Thus, the anhydrous FeCl3 was found to be very effective Lewis acid to achieve the transformations of CF3-substituted allyl alcohols 1 to the corresponding dimers 3 at rt for just 1 hour. It is worth mentioning that these reactions need at least 50 mole % of FeCl3, rather than catalytic one. The use ...
... Thus, the anhydrous FeCl3 was found to be very effective Lewis acid to achieve the transformations of CF3-substituted allyl alcohols 1 to the corresponding dimers 3 at rt for just 1 hour. It is worth mentioning that these reactions need at least 50 mole % of FeCl3, rather than catalytic one. The use ...
Organic Chemistry Fifth Edition
... When both acyl groups are the same, name the acid and add the word anhydride. When the groups are different, list the names of the corresponding acids in alphabetical order and add the word anhydride. ...
... When both acyl groups are the same, name the acid and add the word anhydride. When the groups are different, list the names of the corresponding acids in alphabetical order and add the word anhydride. ...
organic compound containing nitrogen
... This reaction is called Hoffmann elimination, is quite analogous to the dehydrohalogenation of an alkyl halide. Most commonly reaction is E2 : Hydroxide ion abstracts a proton from carbon; a molecule of tertiary amine is expelled. ...
... This reaction is called Hoffmann elimination, is quite analogous to the dehydrohalogenation of an alkyl halide. Most commonly reaction is E2 : Hydroxide ion abstracts a proton from carbon; a molecule of tertiary amine is expelled. ...
Alcohols phenols ethers
... or R-OH where R is any alkyl group .These are named by the following systems. (i) common system: In the common system, monohydric alcohols are called alkyl alcohols their names are derived by adding the word alcohol to the name of the alkyl group present in the molecule. (ii) carbinol system: In thi ...
... or R-OH where R is any alkyl group .These are named by the following systems. (i) common system: In the common system, monohydric alcohols are called alkyl alcohols their names are derived by adding the word alcohol to the name of the alkyl group present in the molecule. (ii) carbinol system: In thi ...
Amines
... Amides, RCONR'2, can be reduced to the amine, RCH2NR'2 by conversion of the C=O to -CH2Amides can be reduced by LiAlH4 but NOT the less reactive NaBH4 Typical reagents : LiAlH4 / ether solvent, followed by aqueous work-up. Note that this reaction is different to that of other C=O compounds which red ...
... Amides, RCONR'2, can be reduced to the amine, RCH2NR'2 by conversion of the C=O to -CH2Amides can be reduced by LiAlH4 but NOT the less reactive NaBH4 Typical reagents : LiAlH4 / ether solvent, followed by aqueous work-up. Note that this reaction is different to that of other C=O compounds which red ...
Mechanism of Action of Sodium Hypochlorite - FORP
... with a detergent) on S. aureus, E. faecalis, P. aeruginosa, B. subtilis, C. albicans and a mixture of these microorganisms. The purpose was to determine the minimum inhibitory concentration of the tested solutions with a serial dilution in the proportion of 1:10, and antimicrobial activity with dire ...
... with a detergent) on S. aureus, E. faecalis, P. aeruginosa, B. subtilis, C. albicans and a mixture of these microorganisms. The purpose was to determine the minimum inhibitory concentration of the tested solutions with a serial dilution in the proportion of 1:10, and antimicrobial activity with dire ...
lecture 11 catalysis_hydrogenation of alkenes
... Once CoH(CN)53− has been formed, a H• atom is transferred to the substrate in the second step, a reaction that does not require a vacant site at the metal, but does require the resulting organic radical to be moderately stable—hence the fact that the Iguchi catalyst will reduce only activated alken ...
... Once CoH(CN)53− has been formed, a H• atom is transferred to the substrate in the second step, a reaction that does not require a vacant site at the metal, but does require the resulting organic radical to be moderately stable—hence the fact that the Iguchi catalyst will reduce only activated alken ...
Carbonyl Compounds notes
... CH3COO-Na+(aq) + HCl(aq) CH3COOH(aq) + NaCl(l) sodium ethanoate ethanoic acid ...
... CH3COO-Na+(aq) + HCl(aq) CH3COOH(aq) + NaCl(l) sodium ethanoate ethanoic acid ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.














![T_AllylCF3paperBM[5]](http://s1.studyres.com/store/data/003584459_1-3decab572f7fca68901a941affab18ea-300x300.png)







