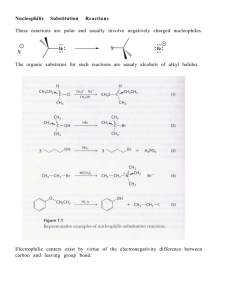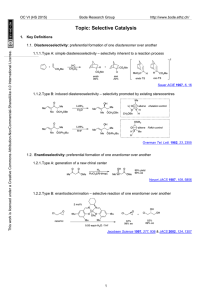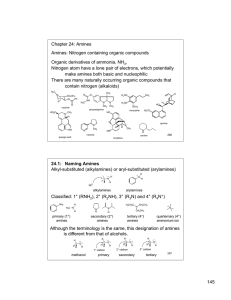
145 Chapter 24: Amines Amines: Nitrogen containing organic
... Symmetrical secondary and tertiary amines are named by adding the prefix di- or tri- to the alkyl group Unsymmetrical secondary and tertiary amines are named as N-substituted primary amines. The largest alkyl group is the parent name, and other alkyl groups are considered N-substituents. H H3CH2C ...
... Symmetrical secondary and tertiary amines are named by adding the prefix di- or tri- to the alkyl group Unsymmetrical secondary and tertiary amines are named as N-substituted primary amines. The largest alkyl group is the parent name, and other alkyl groups are considered N-substituents. H H3CH2C ...
PHENOL - Gneet's
... 1. Alkali fusion of sulphonates Sodium salt of aryl sulphonic acids on fusion with sodium hydroxide at 300350oC yield phenol ...
... 1. Alkali fusion of sulphonates Sodium salt of aryl sulphonic acids on fusion with sodium hydroxide at 300350oC yield phenol ...
15.1 Amines
... transition-metal catalyst. • Reduction of an aldehyde gives a primary alcohol. • Reduction a ketone gives a secondary alcohol. ...
... transition-metal catalyst. • Reduction of an aldehyde gives a primary alcohol. • Reduction a ketone gives a secondary alcohol. ...
review-rough
... 22. This artificial sweetener is thought to be dangerous because it releases methanol as one of its breakdown products. Experts feel it is not in high enough concentration to be dangerous. ...
... 22. This artificial sweetener is thought to be dangerous because it releases methanol as one of its breakdown products. Experts feel it is not in high enough concentration to be dangerous. ...
Chapter: Haloalkanes and Haloarenes
... new forces of attraction set up between haloalkanes and the solvent molecules are of same strength as the forces of attraction being broken. (ii) A mixture of equal amounts of two enantiomers is known as racemic mixture. For example: When a 3o halide undergoes substitution with KOH, the reaction pro ...
... new forces of attraction set up between haloalkanes and the solvent molecules are of same strength as the forces of attraction being broken. (ii) A mixture of equal amounts of two enantiomers is known as racemic mixture. For example: When a 3o halide undergoes substitution with KOH, the reaction pro ...
Cleavage of dinitrogen to yield a (t-BuPOCOP)molybdenum( ) nitridew Chem. Commun Cite this:
... formation of a neutral species in which it is proposed that the proton has added across the Mo–P bond. The catalytic reduction of N2 to NH3 at low temperature (22 1C) and pressure (1 atm) has been a long standing goal, both in terms of understanding how this transformation might be achieved in vario ...
... formation of a neutral species in which it is proposed that the proton has added across the Mo–P bond. The catalytic reduction of N2 to NH3 at low temperature (22 1C) and pressure (1 atm) has been a long standing goal, both in terms of understanding how this transformation might be achieved in vario ...
A manganese catalyst for highly reactive yet chemoselective
... while other chemoselective catalysts 1 and [Ru2(hp)4] are less reactive (12 and 25%, respectively)11,15. This electronic insensitivity is further highlighted by the tolerance of electron-withdrawing nitrogen functionality (22, 73%) introduced via palladium-catalysed intermolecular allylic C–H aminat ...
... while other chemoselective catalysts 1 and [Ru2(hp)4] are less reactive (12 and 25%, respectively)11,15. This electronic insensitivity is further highlighted by the tolerance of electron-withdrawing nitrogen functionality (22, 73%) introduced via palladium-catalysed intermolecular allylic C–H aminat ...
Oxidative Alihatic Carbon-Carbon Bond Cleavage Reactions
... Department: Chemistry and Biochemistry The work presented in this dissertation has focused on the activation and cleavage of chemical bonds between two carbon atoms. The selective oxidative activation of carbon-carbon bonds is important due to potential applications in the utilization of biomass for ...
... Department: Chemistry and Biochemistry The work presented in this dissertation has focused on the activation and cleavage of chemical bonds between two carbon atoms. The selective oxidative activation of carbon-carbon bonds is important due to potential applications in the utilization of biomass for ...
molecules
... and affords 80% cis- and 10% trans-stilbene oxides, respectively. This catalytic system exhibits a good regioselectivity for epoxidation of R-(+)-limonene. The ratio among 1,2- and 8,9-epoxides was found to be 2.3:1. Selective partial alkane hydroxylation is a particularly challenging problem in org ...
... and affords 80% cis- and 10% trans-stilbene oxides, respectively. This catalytic system exhibits a good regioselectivity for epoxidation of R-(+)-limonene. The ratio among 1,2- and 8,9-epoxides was found to be 2.3:1. Selective partial alkane hydroxylation is a particularly challenging problem in org ...
phenols - Gneet`s
... Sodium salt of aryl sulphonic acids on fusion with sodium hydroxide at 300-350oC yield phenol ...
... Sodium salt of aryl sulphonic acids on fusion with sodium hydroxide at 300-350oC yield phenol ...
Alkenes notes
... Symmetrical alkenes only give one product when elecrophiles are added. Unsymmetrical alkenes only give one product if the electrophile is symmetrical (Eg propene Br2). Thus two products are only obtained when both the alkene and the electrophile are unsymmetrical. In such cases the identity of the m ...
... Symmetrical alkenes only give one product when elecrophiles are added. Unsymmetrical alkenes only give one product if the electrophile is symmetrical (Eg propene Br2). Thus two products are only obtained when both the alkene and the electrophile are unsymmetrical. In such cases the identity of the m ...
Alcohols
... • Nucleophilic substitution of OH- on alkyl halide • Hydration of alkenes water in acid solution (not very effective) oxymercuration - demercuration hydroboration - oxidation ...
... • Nucleophilic substitution of OH- on alkyl halide • Hydration of alkenes water in acid solution (not very effective) oxymercuration - demercuration hydroboration - oxidation ...
Triruthenium and triosmium carbonyl clusters containing chiral
... IR spectroscopy.11a A spot TLC analysis of the solution also indicated the presence of small amounts of 1 + 2, [Ru3 (CO)12 ], and a yellow species that decomposed on the TLC plate after a couple of minutes. Heating this solution to reflux temperature led to 1 and 2 as the major products. In a subsequ ...
... IR spectroscopy.11a A spot TLC analysis of the solution also indicated the presence of small amounts of 1 + 2, [Ru3 (CO)12 ], and a yellow species that decomposed on the TLC plate after a couple of minutes. Heating this solution to reflux temperature led to 1 and 2 as the major products. In a subsequ ...
Learning Guide for Chapter 9 - Alkyl Halides I
... Exclusion of aryl halides, vinyl halides, and acid chlorides Summary of Substitution Reactions III. Synthesis Using Substitution Reactions I. Introduction to alkyl halides Types of organic halides What is the purpose of categorizing organic halides into different groups? so that we can predict how t ...
... Exclusion of aryl halides, vinyl halides, and acid chlorides Summary of Substitution Reactions III. Synthesis Using Substitution Reactions I. Introduction to alkyl halides Types of organic halides What is the purpose of categorizing organic halides into different groups? so that we can predict how t ...
Enol esters: Versatile substrates in synthesis of fine and specialty
... acidic conditions with mostly retention of configuration. Both α-acyloxy ketones and their αhydroxy ketone derivatives, are essential in synthesis of alkaloids, sugars, antibiotics, terpenes and pheromones, for they function as stereodirective groups or chiral synthons. Zhu et al. practised the rear ...
... acidic conditions with mostly retention of configuration. Both α-acyloxy ketones and their αhydroxy ketone derivatives, are essential in synthesis of alkaloids, sugars, antibiotics, terpenes and pheromones, for they function as stereodirective groups or chiral synthons. Zhu et al. practised the rear ...
RES15_c2_wp
... This is in preparation for Activity M1: ‘Could phenol be used as an antiseptic in hospitals (or the home) today?’ Students to work in groups. ...
... This is in preparation for Activity M1: ‘Could phenol be used as an antiseptic in hospitals (or the home) today?’ Students to work in groups. ...
Chapter 22 and 23 Study Guide
... notes or help from anyone. You should review homework, quizzes and other assignments for complete test preparation. These questions are only examples. Different chemicals will be on the test. ...
... notes or help from anyone. You should review homework, quizzes and other assignments for complete test preparation. These questions are only examples. Different chemicals will be on the test. ...
Module 5 Reactions with Miscellaneous Reagents
... The reaction is initiated by light or peroxide. Although a number of reagents are available for bromination of allylic C-H bond of alkenes, NBS is most commonly used. The reaction is called Wohl-Zigler bromination. For example, cyclohexene reacts with NBS to give 3-bromocyclohexene in the presence o ...
... The reaction is initiated by light or peroxide. Although a number of reagents are available for bromination of allylic C-H bond of alkenes, NBS is most commonly used. The reaction is called Wohl-Zigler bromination. For example, cyclohexene reacts with NBS to give 3-bromocyclohexene in the presence o ...
4.9 Preparation of Alkyl Halides from Alcohols and Hydrogen Halides
... The last step in the mechanism of the reaction of tert-butyl alcohol with hydrogen chloride is the reaction between an electrophile and a nucleophile. tert-Butyl cation is the electrophile. Chloride ion is the nucleophile. ...
... The last step in the mechanism of the reaction of tert-butyl alcohol with hydrogen chloride is the reaction between an electrophile and a nucleophile. tert-Butyl cation is the electrophile. Chloride ion is the nucleophile. ...
Chapter Seven - U of L Class Index
... Both these reactions operate on the principal of converting OH into a better leaving ...
... Both these reactions operate on the principal of converting OH into a better leaving ...
Sample pages 6 PDF
... salt of the acid that can be isolated by filtration. Generally, the remainder of the workup of the reaction can be made according to three alternative protocols: Workup A: The reaction mixture is optionally washed with an organic solvent like Et2 O or CH2 Cl2 . When the product—even as a sodium carb ...
... salt of the acid that can be isolated by filtration. Generally, the remainder of the workup of the reaction can be made according to three alternative protocols: Workup A: The reaction mixture is optionally washed with an organic solvent like Et2 O or CH2 Cl2 . When the product—even as a sodium carb ...
Redox reactions_organic - Welcome to SALEM
... Alkenes react rapidly and quantitatively with ozone form an unstable compound, known as ozonide ...
... Alkenes react rapidly and quantitatively with ozone form an unstable compound, known as ozonide ...
Changing counterion can switch the preference for selective 1,2
... electronic properties of the components involved. However, this inbuilt selectivity is rarely complete, and is often challenging to control. However, catalytic methods for divergent regioselective cycloaddition reactions have begun to emerge. For example, Maruoka has reported the site-divergent Diel ...
... electronic properties of the components involved. However, this inbuilt selectivity is rarely complete, and is often challenging to control. However, catalytic methods for divergent regioselective cycloaddition reactions have begun to emerge. For example, Maruoka has reported the site-divergent Diel ...
IOSR Journal of Applied Chemistry (IOSR-JAC)
... the carbonyl group. In this case, the nucleophile is the amine. In the first part of the mechanism, the amine reacts with the aldehyde or ketone to give an unstable addition compound called carbinolamine. The carbinolamine loses water by either acid or base catalyzed pathways. Since the carbinolamin ...
... the carbonyl group. In this case, the nucleophile is the amine. In the first part of the mechanism, the amine reacts with the aldehyde or ketone to give an unstable addition compound called carbinolamine. The carbinolamine loses water by either acid or base catalyzed pathways. Since the carbinolamin ...
Class Notes Test 1
... • 3º alcohol, all three attachments different ß Ketone Precursor • 3º alcohol, two (or more) of the attachments identical ß Ester • 2º alcohol ß Aldehyde • 1º alcohol ß Formaldehyde or ethylene oxide ...
... • 3º alcohol, all three attachments different ß Ketone Precursor • 3º alcohol, two (or more) of the attachments identical ß Ester • 2º alcohol ß Aldehyde • 1º alcohol ß Formaldehyde or ethylene oxide ...
Wolff–Kishner reduction

The Wolff–Kishner reduction is a reaction used in organic chemistry to convert carbonyl functionalities into methylene groups. In the context of complex molecule synthesis, it is most frequently employed to remove a carbonyl group after it has served its synthetic purpose of activating an intermediate in a preceding step. As such, there is no obvious retron for this reaction. Originally reported by Nikolai Kischner in 1911 and Ludwig Wolff in 1912, it has been applied to the total synthesis of scopadulcic acid B, aspidospermidine and dysidiolide.In general, the reaction mechanism first involves the in situ generation of a hydrazone by condensation of hydrazine with the ketone or aldehyde substrate. Sometimes it is however advantageous to use a pre-formed hydrazone as substrate (see modifications). The hydrazone is deprotonated by alkoxide base followed by a concerted, rate-determining step in which a diimide anion is formed. Collapse of this alkyldiimde with loss of N2 leads to formation of an alkylanion which can be protonated by solvent to give the desired product.Because the Wolff–Kishner reduction requires highly basic conditions, it is unsuitable for base-sensitive substrates. However, this method can be superior over the related Clemmensen reduction for acid-sensitive compounds such as pyrroles and for high-molecular weight compounds.