Programma Inglese XXXII Scuola Corbella
... Non-heme iron oxidation catalysis: from bioinspiration to practical application ...
... Non-heme iron oxidation catalysis: from bioinspiration to practical application ...
Viju B - IS MU
... prochiral substrate, that induce a selective formation of one of the enantiomeric products during a stereoselective reaction. When the reaction stereochemistry is achieved, the auxiliary is removed. The benzoin group has already been used as a photoremovable protecting group2 for various functionali ...
... prochiral substrate, that induce a selective formation of one of the enantiomeric products during a stereoselective reaction. When the reaction stereochemistry is achieved, the auxiliary is removed. The benzoin group has already been used as a photoremovable protecting group2 for various functionali ...
Asymmetric Organocatalysis
... The Maruoka group used their highly enantioselective, structurally rigid, chiral spiro catalysts of type 29 in the synthesis of L-Dopa ester (S)-40 and an analog thereof. Initial asymmetric alkylation in the presence of 1 mol% (R,R)-29 gave the intermediate (S)-20q in 81% yield and 98% ee (Scheme 3 ...
... The Maruoka group used their highly enantioselective, structurally rigid, chiral spiro catalysts of type 29 in the synthesis of L-Dopa ester (S)-40 and an analog thereof. Initial asymmetric alkylation in the presence of 1 mol% (R,R)-29 gave the intermediate (S)-20q in 81% yield and 98% ee (Scheme 3 ...
Combi chemistry
... -> Although combinatorial chemistry has only really been taken up by industry since the 1990s, its roots can be seen as far back as the 1960s when are searcher at Rockefeller University, Bruce Merrifield, started investigating the solid-state synthesis of peptides -> Bruce Merrifield won the Nobel p ...
... -> Although combinatorial chemistry has only really been taken up by industry since the 1990s, its roots can be seen as far back as the 1960s when are searcher at Rockefeller University, Bruce Merrifield, started investigating the solid-state synthesis of peptides -> Bruce Merrifield won the Nobel p ...
F017006 - Fluorous Technologies
... F-PMB-OH is the fluorous equivalent of p-methoxybenzyl alcohol (PMB-OH) used in protecting alcohols in multi-step organic synthesis. Protection of an alcohol with F-PMB-OH and deprotection are achieved under traditional reaction conditions, with the advantage that products containing the F-PMB group ...
... F-PMB-OH is the fluorous equivalent of p-methoxybenzyl alcohol (PMB-OH) used in protecting alcohols in multi-step organic synthesis. Protection of an alcohol with F-PMB-OH and deprotection are achieved under traditional reaction conditions, with the advantage that products containing the F-PMB group ...
Organic Synthesis
... to infer reactions from there. Although that’s the current convention, for this document, we’ll use explicit compounds, at least for a while. How was that retrosynthesis done? Notice that the restriction on starting materials requires that at least one C-C bond be made. It could be more than one, bu ...
... to infer reactions from there. Although that’s the current convention, for this document, we’ll use explicit compounds, at least for a while. How was that retrosynthesis done? Notice that the restriction on starting materials requires that at least one C-C bond be made. It could be more than one, bu ...
Document
... oxidized by this reagent, the silver ions are reduced to metallic silver, which forms a black precipitate and, if the test tube is clean, a silver mirror on the test tube. (E. Ferric Chloride Test for Phenols) When a phenol is treated with a solution of ferric chloride, colored complex ions are rapi ...
... oxidized by this reagent, the silver ions are reduced to metallic silver, which forms a black precipitate and, if the test tube is clean, a silver mirror on the test tube. (E. Ferric Chloride Test for Phenols) When a phenol is treated with a solution of ferric chloride, colored complex ions are rapi ...
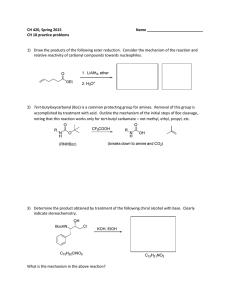
CH 420, Spring 2015 Name ___________________________ CH 18 practice problems
... 1) Draw the products of the following ester reduction. Consider the mechanism of the reaction and relative reactivity of carbonyl compounds towards nucleophiles. ...
... 1) Draw the products of the following ester reduction. Consider the mechanism of the reaction and relative reactivity of carbonyl compounds towards nucleophiles. ...
Synthesis, Isolation and Purification of an Ester
... the general outline for the procedure (refluxing, extraction, and distillation) and ask them to calculate the amounts of ethyl alcohol and glacial (17.4 M) acetic acid needed to prepare 15 g of ethyl acetate. Discuss the following prerequisite knowledge: (1) Synthesis of an ester is generally revers ...
... the general outline for the procedure (refluxing, extraction, and distillation) and ask them to calculate the amounts of ethyl alcohol and glacial (17.4 M) acetic acid needed to prepare 15 g of ethyl acetate. Discuss the following prerequisite knowledge: (1) Synthesis of an ester is generally revers ...
Macromolecule Wkst
... Part E. Each of the following structures below shows a fatty acid molecule. On the line, identify the fatty acid as saturated, unsaturated, or polyunsaturated. ...
... Part E. Each of the following structures below shows a fatty acid molecule. On the line, identify the fatty acid as saturated, unsaturated, or polyunsaturated. ...
Problem Set: Empirical and Molecular Formulas
... 4. Using the same reaction for the production of ammonia as in #3, determine the percent yield when 400.0 kg of H2 are added to an excess of N2, and 1040. kg of NH3 are produced. ...
... 4. Using the same reaction for the production of ammonia as in #3, determine the percent yield when 400.0 kg of H2 are added to an excess of N2, and 1040. kg of NH3 are produced. ...
Microsoft Word - Open Access Repository of Indian Theses
... components. Consequently a variety of approaches have been developed for their asymmetric and racemic synthesis. The chapter starts with the introduction and description of the various literature methods for the synthesis of various functionalized -butyrolactones. The approach towards the synthesis ...
... components. Consequently a variety of approaches have been developed for their asymmetric and racemic synthesis. The chapter starts with the introduction and description of the various literature methods for the synthesis of various functionalized -butyrolactones. The approach towards the synthesis ...
ABSTRACT SYNTHESIS AND STUDY OF ELECTRO
... such as low cost, easy processing, and great opportunities for structural modification. These materials are now being considered as active components in electronic and optoelectronic devices such as light emitting diodes for display applications, thin film transistors for low-cost and ultra-dense lo ...
... such as low cost, easy processing, and great opportunities for structural modification. These materials are now being considered as active components in electronic and optoelectronic devices such as light emitting diodes for display applications, thin film transistors for low-cost and ultra-dense lo ...
投影片 1
... a positive charge, whereas the other E class group resides on a carbon with a negative charge. ...
... a positive charge, whereas the other E class group resides on a carbon with a negative charge. ...
18 Important and sometimes forgotten) organic transformations
... •Phosphines can also be used •DMAP and DBU are better in some cases ...
... •Phosphines can also be used •DMAP and DBU are better in some cases ...
$doc.title
... http://www.chem.wisc.edu/areas/clc (Resource page) Reactions of Alcohols #8: Reaction of a 1° Alcohol with Hydrogen Halides ...
... http://www.chem.wisc.edu/areas/clc (Resource page) Reactions of Alcohols #8: Reaction of a 1° Alcohol with Hydrogen Halides ...
슬라이드 1
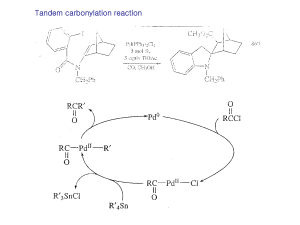
... Nickel carbonyl is an extremely toxic compound, and a number of other nickel reagents with generally similar reactivity can be used in its place. ...
... Nickel carbonyl is an extremely toxic compound, and a number of other nickel reagents with generally similar reactivity can be used in its place. ...
SAMPLE PROBLEM
... Another feasible stereoselective retrosynthesis involves net addition of two nitrogen atoms across the double bond which should generate structure 8 from 9.1 The diastereoselectivity is controlled at this point but there will be a racemic mixture (two enantiomers in the reaction vessel) once the syn ...
... Another feasible stereoselective retrosynthesis involves net addition of two nitrogen atoms across the double bond which should generate structure 8 from 9.1 The diastereoselectivity is controlled at this point but there will be a racemic mixture (two enantiomers in the reaction vessel) once the syn ...
Discodermolide

(+)-Discodermolide is a polyketide natural product found to stabilize microtubule. (+)-discodermolide was isolated by Gunasekera and his co-workers at the Harbor Branch Oceanographic Institute from the deep-sea sponge Discodermia dissoluta in 1990. (+)-Discodermolide was found to be a potent inhibitor of tumor cell growth in several MDR cancer cell lines. (+)-discodermolide also shows some unique characters, including a linear backbone structure, immunosuppressive properties both in vitro and in vivo, potent induction of an accelerated senescence phenotype, and synergistic antiproliferative activity in combination with paclitaxel. Discodermolide was recognized as one of the most potent natural promoters of tubulin assembly. A large number of efforts toward the total synthesis of (+)-discodermolide were directed by its interesting biological activities and extreme scarcity of natural sources (0.002% w/w from frozen marine sponge). The compound supply necessary for complete clinical trials cannot be met by harvesting, isolation, and purification. As of 2005, attempts at synthesis or semi-synthesis by fermentation have proven unsuccessful. As a result, all discodermolide used in preclinical studies and clinical trials has come from large-scale total synthesis.