CC 2 097-110..7686hdisk chapter .. Page97
... yellow–orange colored solution that slowly faded (within about an hour). Introduction of oxygen accelerated the bleaching. Analysis of the solution after aeration gave a positive test for peroxides indicating that oxygen was mostly likely reduced to hydrogen peroxide in the course of bleaching the t ...
... yellow–orange colored solution that slowly faded (within about an hour). Introduction of oxygen accelerated the bleaching. Analysis of the solution after aeration gave a positive test for peroxides indicating that oxygen was mostly likely reduced to hydrogen peroxide in the course of bleaching the t ...
Addition/elimination under acidic conditions
... Nucleophilic acyl substitutions can occur with weak nucleophiles under basic conditions, but only if the carboxylic acid derivative is very reactive Nulceophilic acyl substitutions can occur with weak nucleophiles under acidic conditions Carboxylic acids can be esterified with alcohols under a ...
... Nucleophilic acyl substitutions can occur with weak nucleophiles under basic conditions, but only if the carboxylic acid derivative is very reactive Nulceophilic acyl substitutions can occur with weak nucleophiles under acidic conditions Carboxylic acids can be esterified with alcohols under a ...
Document
... 21.7: Preparation of Amines by Alkylation of Ammonia Ammonia and other alkylamines are good nucleophiles and react with 1° and 2° alkyl halides or tosylates via an SN2 reaction yielding alkyl amines. ...
... 21.7: Preparation of Amines by Alkylation of Ammonia Ammonia and other alkylamines are good nucleophiles and react with 1° and 2° alkyl halides or tosylates via an SN2 reaction yielding alkyl amines. ...
( +)-Limonene Oxidation with Selenium Dioxide
... studied by several workers,2a-d and the products identified involved oxidation a t all allylic positions except carbon-3 (menthol series). Most of these oxidation products are constituents of natural products such as citrus essential oils,2e oi which (+)-limonene is the major constituent. As part of ...
... studied by several workers,2a-d and the products identified involved oxidation a t all allylic positions except carbon-3 (menthol series). Most of these oxidation products are constituents of natural products such as citrus essential oils,2e oi which (+)-limonene is the major constituent. As part of ...
Lecture Resource ()
... The goal is to convert the OH group into a better leaving group such that the acyl chloride can be prepared ...
... The goal is to convert the OH group into a better leaving group such that the acyl chloride can be prepared ...
BHR - A Brief History - Process Intensification Network
... Three main issues with the running of these trials ...
... Three main issues with the running of these trials ...
論 文 の 内 容 の 要 旨 論文題目 Synthesis and Property of Low
... nucleophilic attack of the boryl anion to main group element’s center or small molecules was rather less studied. In this chapter, the reaction of nucleophilic boryl anion with borane was studied. The reaction of boryllithium with borane-THF complex afforded the first boryl-substituted borohydride, ...
... nucleophilic attack of the boryl anion to main group element’s center or small molecules was rather less studied. In this chapter, the reaction of nucleophilic boryl anion with borane was studied. The reaction of boryllithium with borane-THF complex afforded the first boryl-substituted borohydride, ...
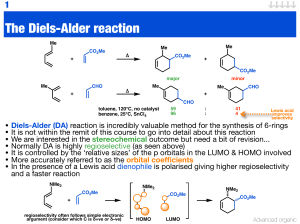
The Diels-Alder reaction
... • Diels-Alder (DA) reaction is incredibly valuable method for the synthesis of 6-rings • It is not within the remit of this course to go into detail about this reaction • We are interested in the stereochemical outcome but need a bit of revision... • Normally DA is highly regioselective (as seen abo ...
... • Diels-Alder (DA) reaction is incredibly valuable method for the synthesis of 6-rings • It is not within the remit of this course to go into detail about this reaction • We are interested in the stereochemical outcome but need a bit of revision... • Normally DA is highly regioselective (as seen abo ...
Origin of the Diastereoselection in the Indium
... of (E)-1a and 2a did not produce 3a at all, which manifested that the allylic sulfone unit served as a good leaving group in the allyl Grignard reagent. The Zn- or Sn-mediated Barbier reaction of (E)-1a and 2a under the above optimized condition (the 4:1 mixed solvent of H2O and THF at reflux for 2. ...
... of (E)-1a and 2a did not produce 3a at all, which manifested that the allylic sulfone unit served as a good leaving group in the allyl Grignard reagent. The Zn- or Sn-mediated Barbier reaction of (E)-1a and 2a under the above optimized condition (the 4:1 mixed solvent of H2O and THF at reflux for 2. ...
Chapter 16 – Amines and Amides
... when pure (as a result of bonding to each other). They are also generally quite water-soluble. Unlike amines, amides show no measurable basicity. Two factors account for this. Most importantly, the lone pair of electrons on nitrogen interacts with the carbonyl π-bond much like the π-bonds in benzene ...
... when pure (as a result of bonding to each other). They are also generally quite water-soluble. Unlike amines, amides show no measurable basicity. Two factors account for this. Most importantly, the lone pair of electrons on nitrogen interacts with the carbonyl π-bond much like the π-bonds in benzene ...
Chem 3.5 #6 Amines
... Name each of the following amines and classify them as primary, secondary or tertiary. (a) ...
... Name each of the following amines and classify them as primary, secondary or tertiary. (a) ...
EXPERIMENT 5: Oxidation of Alcohols: Solid
... relationship has led to the development of a convenient qualitative test for distinguishing primary and secondary alcohols (and aldehydes) from tertiary alcohols (and ketones). The qualitative test involves the addition of a solution of CrO3 in sulfuric acid (Jones' Reagent) to a solution of the com ...
... relationship has led to the development of a convenient qualitative test for distinguishing primary and secondary alcohols (and aldehydes) from tertiary alcohols (and ketones). The qualitative test involves the addition of a solution of CrO3 in sulfuric acid (Jones' Reagent) to a solution of the com ...
Conjugate (1,4
... • New small molecule organic catalysts are now achieving remarkable results • Enone is activated by formation of the charged iminium species • The catalyst also blocks one face of the enone allowing selective attack 123.702 Organic Chemistry ...
... • New small molecule organic catalysts are now achieving remarkable results • Enone is activated by formation of the charged iminium species • The catalyst also blocks one face of the enone allowing selective attack 123.702 Organic Chemistry ...
2287 Summary
... Brominated Ketones.-The procedure for the preparation was that of Schmidt,6 according to which the ketone is brominated in glacial acetic acid. a-Bromo-n-va1erophenone.-As this substance has not previously been described in the literature, it may be mentioned that it is a straw-colored liquid boilin ...
... Brominated Ketones.-The procedure for the preparation was that of Schmidt,6 according to which the ketone is brominated in glacial acetic acid. a-Bromo-n-va1erophenone.-As this substance has not previously been described in the literature, it may be mentioned that it is a straw-colored liquid boilin ...
A Model for Catalytically Active Zinc(I1) Ion in Liver
... C.-I.; Akeson, A. J . Mol. Biol. 1976, 102, 27. (c) Branden, C.-I. In The Pyridine Nucleotide-Dependent Dehydrogenases; Sund, H., Ed.; W. de Gruiter: Berlin, 1977; pp 325-338. (d) Dowarschack, R. T.; Plapp, B. V. Biochemistry 1977, 16, 2716. (e) Drysdale, B.-E.; Hollis, D. P. Arch. Biochem. Biophys. ...
... C.-I.; Akeson, A. J . Mol. Biol. 1976, 102, 27. (c) Branden, C.-I. In The Pyridine Nucleotide-Dependent Dehydrogenases; Sund, H., Ed.; W. de Gruiter: Berlin, 1977; pp 325-338. (d) Dowarschack, R. T.; Plapp, B. V. Biochemistry 1977, 16, 2716. (e) Drysdale, B.-E.; Hollis, D. P. Arch. Biochem. Biophys. ...
alcohols-II-12-ques
... RCH2OH + PDC [(C5H5NH+)2 Cr2O72–] A) The alcohol is oxidized to an acid, and the Cr(VI) is reduced. B) The alcohol is oxidized to an aldehyde, and the Cr(VI) is reduced. C) The alcohol is reduced to an aldehyde, and the Cr(III) is oxidized. D) The alcohol is oxidized to a ketone, and the Cr(VI) is ...
... RCH2OH + PDC [(C5H5NH+)2 Cr2O72–] A) The alcohol is oxidized to an acid, and the Cr(VI) is reduced. B) The alcohol is oxidized to an aldehyde, and the Cr(VI) is reduced. C) The alcohol is reduced to an aldehyde, and the Cr(III) is oxidized. D) The alcohol is oxidized to a ketone, and the Cr(VI) is ...
Esters are reduced by hydride reagents to give alcohols or aldehydes.
... Esters form enolates that can be alkylated. Treatment of esters with a strong base at low temperatures produces ester enolates (acidic -hydrogens). These enolates react like ketone enolates, undergoing alkylations. ...
... Esters form enolates that can be alkylated. Treatment of esters with a strong base at low temperatures produces ester enolates (acidic -hydrogens). These enolates react like ketone enolates, undergoing alkylations. ...
Mechanism
... One of the main drawbacks of the Henry Reaction is the potential for side reactions throughout the course of the reaction. Aside from the reversibility of the reaction (Retro-Henry) which could prevent the reaction from proceeding, the β-nitro alcohol has the potential to undergo dehydration, and fo ...
... One of the main drawbacks of the Henry Reaction is the potential for side reactions throughout the course of the reaction. Aside from the reversibility of the reaction (Retro-Henry) which could prevent the reaction from proceeding, the β-nitro alcohol has the potential to undergo dehydration, and fo ...
A New Method for Halodecarboxylation of Acids Using Lead(IV
... The mixture is swept with nitrogen and heated to 80" with stirring. With LiCl and CaC12, though initially heterogeneous, the reaction is complete within a few minutes. The very insoluble KC1 and NaCl require longer heating. Acetonitrile and hexane-benzene mixtures can also be employed as solvents, a ...
... The mixture is swept with nitrogen and heated to 80" with stirring. With LiCl and CaC12, though initially heterogeneous, the reaction is complete within a few minutes. The very insoluble KC1 and NaCl require longer heating. Acetonitrile and hexane-benzene mixtures can also be employed as solvents, a ...
Rapid Microwave Synthesis, Characterization and Reactivity
... IR spectra were collected at room temperature (20 scans/sample, 8 cm−1 resolution) using a Shimadzu FTIR 8400S instrument with a Pike MIRacle ATR sampling accessory. Raman spectra were collected at room temperature using a Horiba LabRAM HR confocal microscope system (Horiba Itd., Kyoto, Japan) with ...
... IR spectra were collected at room temperature (20 scans/sample, 8 cm−1 resolution) using a Shimadzu FTIR 8400S instrument with a Pike MIRacle ATR sampling accessory. Raman spectra were collected at room temperature using a Horiba LabRAM HR confocal microscope system (Horiba Itd., Kyoto, Japan) with ...
Instructor notes
... a possible mechanism for this Hg(II)/H2SO4 system is shown. Because Hg(II) does not have an accessible oxidative addition pathway, an electrophilic C-H activation route is believed to occur. The methylmercury(II) intermediate has been detected by NMR spectroscopy. Attack by SO42- could lead to nucle ...
... a possible mechanism for this Hg(II)/H2SO4 system is shown. Because Hg(II) does not have an accessible oxidative addition pathway, an electrophilic C-H activation route is believed to occur. The methylmercury(II) intermediate has been detected by NMR spectroscopy. Attack by SO42- could lead to nucle ...
haloalkanes - Knockhardy
... alternative method involves the initial breaking of the C-X bond to form a carbocation, or carbonium ion, (a unimolecular process - SN1 mechanism), which is then attacked by the nucleophile. SN1 is favoured for tertiary haloalkanes where there is steric hindrance to the attack and a more stable tert ...
... alternative method involves the initial breaking of the C-X bond to form a carbocation, or carbonium ion, (a unimolecular process - SN1 mechanism), which is then attacked by the nucleophile. SN1 is favoured for tertiary haloalkanes where there is steric hindrance to the attack and a more stable tert ...
Oxidation of Cyclohexanol to Cyclohexanone Notes
... Cyclohexanol is an irritant. Avoid contact with skin, eyes, and clothing. Glacial acetic acid is a dehydrating agent, an irritant, and corrosive which means it causes burns. Handle it with care. Dispense it in a fume hood and avoid contact with skin, eyes, and clothing. Sodium hypochlorite solution ...
... Cyclohexanol is an irritant. Avoid contact with skin, eyes, and clothing. Glacial acetic acid is a dehydrating agent, an irritant, and corrosive which means it causes burns. Handle it with care. Dispense it in a fume hood and avoid contact with skin, eyes, and clothing. Sodium hypochlorite solution ...
Acid-Catalyzed Dehydration of Alcohols
... as a layer on the surface of the acid-alcohol mixture (the density of the alkene is less than one); the gas can then be collected over water or the liquid layer can be removed by simple distillation to give the final alkene product. In alcohols where there are more than two kinks of -hydrogens, the ...
... as a layer on the surface of the acid-alcohol mixture (the density of the alkene is less than one); the gas can then be collected over water or the liquid layer can be removed by simple distillation to give the final alkene product. In alcohols where there are more than two kinks of -hydrogens, the ...
Hofmann–Löffler reaction

The Hofmann–Löffler reaction (also referred to as Hofmann–Löffler–Freytag reaction, Löffler–Freytag reaction, Löffler–Hofmann reaction, as well as Löffler's method) is an organic reaction in which a cyclic amine 2 (pyrrolidine or, in some cases, piperidine) is generated by thermal or photochemical decomposition of N-halogenated amine 1 in the presence of a strong acid (concentrated sulfuric acid or concentrated CF3CO2H). The Hofmann–Löffler–Freytag reaction proceeds via an intramolecular hydrogen atom transfer to a nitrogen-centered radical and is an example of a remote intramolecular free radical C–H functionalization.