Synthesis of Imidine Hydrochloride and Some
... propioimidate (7)(25): At first prepare the free imidate by shaken the (0.01 mole) imidate hydrochloride in ether with a strong solution of potassium carbonate at 0 C. The ethereal solution was shaken with (0.01 mole) glycine ethyl ester hydrochloride in 2 ml of water for 1 hr. The ether layer was ...
... propioimidate (7)(25): At first prepare the free imidate by shaken the (0.01 mole) imidate hydrochloride in ether with a strong solution of potassium carbonate at 0 C. The ethereal solution was shaken with (0.01 mole) glycine ethyl ester hydrochloride in 2 ml of water for 1 hr. The ether layer was ...
friedel-craft reaction: a review - Advance Institute of Biotech
... chloride and hydrogen chloride via benzyl alcohol as an intermediate, called chloromethylation reaction. It is also known that the reaction of benzaldehyde and ...
... chloride and hydrogen chloride via benzyl alcohol as an intermediate, called chloromethylation reaction. It is also known that the reaction of benzaldehyde and ...
Alkynes
... But typical of synthetic problems side reaction occurs to some extent and must be taken into account. ...
... But typical of synthetic problems side reaction occurs to some extent and must be taken into account. ...
Enhanced diastereoselectivity of an ene hydroperoxidation reaction
... 0040-4039/01/$ - see front matter © 2001 Elsevier Science Ltd. All rights reserved. PII: S 0 0 4 0 - 4 0 3 9 ( 0 1 ) 0 1 1 7 0 - 4 ...
... 0040-4039/01/$ - see front matter © 2001 Elsevier Science Ltd. All rights reserved. PII: S 0 0 4 0 - 4 0 3 9 ( 0 1 ) 0 1 1 7 0 - 4 ...
CI 12.4 - Sackville School
... CI 12.2 Ethene is unsaturated; it has a double C=C bond. It undergoes electrophilic addition reactions in which another molecule is added on. A saturated compound is produced. a. Ethene with bromine (g) or (l) or dissolved in an organic solvent. Ethene will decolourise orange bromine; it reacts rapi ...
... CI 12.2 Ethene is unsaturated; it has a double C=C bond. It undergoes electrophilic addition reactions in which another molecule is added on. A saturated compound is produced. a. Ethene with bromine (g) or (l) or dissolved in an organic solvent. Ethene will decolourise orange bromine; it reacts rapi ...
ethers - WordPress.com
... Reactions of Ethers: Acidic Cleavage • Ethers are generally unreactive • Strong acid will cleave an ether at elevated temperature • HI, HBr produce an alkyl halide from less hindered component by SN2 (tertiary ethers undergo SN1) ...
... Reactions of Ethers: Acidic Cleavage • Ethers are generally unreactive • Strong acid will cleave an ether at elevated temperature • HI, HBr produce an alkyl halide from less hindered component by SN2 (tertiary ethers undergo SN1) ...
ANSWERS: Types of Reactions - Chemical Minds
... 6) Addition – occurs in alkenes because they have double bonds. Ethene is an alkene so will undergo addition reactions. The chlorine (molecule) will add (across the double bond.) CH2 = CH2 + Cl2 → CH2Cl – CH2Cl Substitution occurs in alkanes (because they have single bonds). Ethane is an alkane, so ...
... 6) Addition – occurs in alkenes because they have double bonds. Ethene is an alkene so will undergo addition reactions. The chlorine (molecule) will add (across the double bond.) CH2 = CH2 + Cl2 → CH2Cl – CH2Cl Substitution occurs in alkanes (because they have single bonds). Ethane is an alkane, so ...
An Efficient Method for Selective Deprotection of Trimethylsilyl
... An Efficient Method for Selective Deprotection of Trimethylsilyl Ethers and Tetrahydropyranyl Ethers under Solvent-free Conditions A. R. Hajipour,†,‡,* Hamid R. Bagheri,‡ and Arnold E. Ruoho† Dept. of Pharmacology, University of Wisconsin, Medical School, 1300 University Avenue, Madison, WI, 53706-1 ...
... An Efficient Method for Selective Deprotection of Trimethylsilyl Ethers and Tetrahydropyranyl Ethers under Solvent-free Conditions A. R. Hajipour,†,‡,* Hamid R. Bagheri,‡ and Arnold E. Ruoho† Dept. of Pharmacology, University of Wisconsin, Medical School, 1300 University Avenue, Madison, WI, 53706-1 ...
Pyrrolidine-2-carboxylic Acid (l
... Abhimanyu S. Paraskar obtained his Masters degree in chemistry from Amravati University, Maharastra, India. He is currently working on his PhD thesis under the supervision of Dr. Arumugum Sudalai at National Chemical Laboratory, Pune, India. ...
... Abhimanyu S. Paraskar obtained his Masters degree in chemistry from Amravati University, Maharastra, India. He is currently working on his PhD thesis under the supervision of Dr. Arumugum Sudalai at National Chemical Laboratory, Pune, India. ...
Chemistry 250A -- Exam #3 Answer Key -
... and the leaving group are periplanar to each other, which in a six-membered ring requires that they be in a trans-diaxial conformation. In menthyl chloride there is only one beta hydrogen in this orientation and therefore elimination proceeds to give only the product shown. By contrast, the second r ...
... and the leaving group are periplanar to each other, which in a six-membered ring requires that they be in a trans-diaxial conformation. In menthyl chloride there is only one beta hydrogen in this orientation and therefore elimination proceeds to give only the product shown. By contrast, the second r ...
A Biocatalytic Henry Reaction-The Hydroxynitrile Lyase from Hevea
... absolute configuration of the product was determined to be S,[5] which is in agreement with the known stereopreference of HbHNL in cyanohydrin reactions. Although the nitroaldol reaction has been known for more than a century,[6] stereoselective protocols started to evolve only a few decades ago. In ...
... absolute configuration of the product was determined to be S,[5] which is in agreement with the known stereopreference of HbHNL in cyanohydrin reactions. Although the nitroaldol reaction has been known for more than a century,[6] stereoselective protocols started to evolve only a few decades ago. In ...
Direct ester condensation catalyzed by bulky diarylammonium
... steric bulkiness of the mesityl and pentalimitations of the present method is the need for less-polar solvents: fluorophenyl groups in the catalyst suppressed the dehydrative it is difficult to perform ester condensation of hydrophilic subelimination of secondary alcohols to produce alkenes. When th ...
... steric bulkiness of the mesityl and pentalimitations of the present method is the need for less-polar solvents: fluorophenyl groups in the catalyst suppressed the dehydrative it is difficult to perform ester condensation of hydrophilic subelimination of secondary alcohols to produce alkenes. When th ...
Silica supported zinc chloride catalyzed acetylation of amines
... In the area of green chemistry, the development of heterogeneous catalysts has played an important role, since they offer several advantages such as clean reaction conditions, easy separation of the catalyst from the reaction medium, greater selectivity and generally give higher yield of products an ...
... In the area of green chemistry, the development of heterogeneous catalysts has played an important role, since they offer several advantages such as clean reaction conditions, easy separation of the catalyst from the reaction medium, greater selectivity and generally give higher yield of products an ...
Microsoft Word
... Thesis entitled "Asymmetric Dihydroxylation and Wittig-Horner approach to the Synthesis of Bioactive Molecules and Heterogeneous Catalysis for Organic Transformations" is divided into five chapters. Chapter 1: describes a brief introduction to the Sharpless Asymmetric dihydroxylation (SAD) and its a ...
... Thesis entitled "Asymmetric Dihydroxylation and Wittig-Horner approach to the Synthesis of Bioactive Molecules and Heterogeneous Catalysis for Organic Transformations" is divided into five chapters. Chapter 1: describes a brief introduction to the Sharpless Asymmetric dihydroxylation (SAD) and its a ...
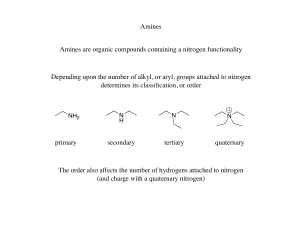
Chapter 20 Amines - FIU Faculty Websites
... • 10 Amines are named using either systematic or common names. • To assign a systematic name, find the longest continuous chain bonded to the amine nitrogen, and change the –e ending of the parent alkane to the suffix –amine. Then use the usual rules of nomenclature to number the chain and name the ...
... • 10 Amines are named using either systematic or common names. • To assign a systematic name, find the longest continuous chain bonded to the amine nitrogen, and change the –e ending of the parent alkane to the suffix –amine. Then use the usual rules of nomenclature to number the chain and name the ...
NCEA Level 3 Chemistry (91391) 2013
... Carboxylic acid (butanoic acid) is obtained by reacting a mixture of butan-1ol with acidified potassium dichromate solution (under reflux conditions) until all of the reactant has been converted to butanoic acid. Observations: orange Cr2O72– to green /, purple MnO4– to colourless / aldehyde condense ...
... Carboxylic acid (butanoic acid) is obtained by reacting a mixture of butan-1ol with acidified potassium dichromate solution (under reflux conditions) until all of the reactant has been converted to butanoic acid. Observations: orange Cr2O72– to green /, purple MnO4– to colourless / aldehyde condense ...
167KB - NZQA
... Carboxylic acid (butanoic acid) is obtained by reacting a mixture of butan-1ol with acidified potassium dichromate solution (under reflux conditions) until all of the reactant has been converted to butanoic acid. Observations: orange Cr2O72– to green /, purple MnO4– to colourless / aldehyde condense ...
... Carboxylic acid (butanoic acid) is obtained by reacting a mixture of butan-1ol with acidified potassium dichromate solution (under reflux conditions) until all of the reactant has been converted to butanoic acid. Observations: orange Cr2O72– to green /, purple MnO4– to colourless / aldehyde condense ...
Amines and amides
... This reaction involves breaking the C-N bond at the carbonyl carbon. a. acid hydrolysis: forms “ammonium” ion and carboxylic acid b. alkaline hydrolysis: forms carboxylate ion and amine Condensation Polymers As we have seen –COOH (and COCl) react with amines to form 2ry amides A Condensation reactio ...
... This reaction involves breaking the C-N bond at the carbonyl carbon. a. acid hydrolysis: forms “ammonium” ion and carboxylic acid b. alkaline hydrolysis: forms carboxylate ion and amine Condensation Polymers As we have seen –COOH (and COCl) react with amines to form 2ry amides A Condensation reactio ...
EXPERIMENT 4 Objectives Principles
... From the reaction above, cyclohexene is the only alkene that can be formed under these conditions. Cyclohexene and water are removed via azeotropic distillation to drive the equilibrium to product. Traces of acid in crude product are removed by treatment with sodium carbonate solution. A final wash ...
... From the reaction above, cyclohexene is the only alkene that can be formed under these conditions. Cyclohexene and water are removed via azeotropic distillation to drive the equilibrium to product. Traces of acid in crude product are removed by treatment with sodium carbonate solution. A final wash ...
Exam 2
... 1. (10 pts) Circle what is true about Substitution and elimination reactions. a. Strong Bases (B") have conjugate acids (BH) that have small pKa values (<5) ...
... 1. (10 pts) Circle what is true about Substitution and elimination reactions. a. Strong Bases (B") have conjugate acids (BH) that have small pKa values (<5) ...
A Direct Access to 3-(2-Oxoalkyl)indoles via
... 413a as a sole or major product when ZnCl2 or SnCl4 was used as catalysts (entries 1 ,2, and 5, Table 1). FeCl3 yielded dimeric product 513b,c and diacetylated product 6 (entry 3, Table 1) in low yields. The use of TiCl4 led to the formation of a mixture of unidentified products (entry 4, Table 1). ...
... 413a as a sole or major product when ZnCl2 or SnCl4 was used as catalysts (entries 1 ,2, and 5, Table 1). FeCl3 yielded dimeric product 513b,c and diacetylated product 6 (entry 3, Table 1) in low yields. The use of TiCl4 led to the formation of a mixture of unidentified products (entry 4, Table 1). ...
PART 3 Principles and Applications of Organometallics in Catalysis
... The effect of a catalyst is to change the rate of conversion of a substrate into products, but they do not change the position of an equilibrium. The thermodynamics of the reaction concerned have to be favourable at the outset; catalysts can't perform the miracle of pushing a reaction up the thermod ...
... The effect of a catalyst is to change the rate of conversion of a substrate into products, but they do not change the position of an equilibrium. The thermodynamics of the reaction concerned have to be favourable at the outset; catalysts can't perform the miracle of pushing a reaction up the thermod ...
Hofmann–Löffler reaction

The Hofmann–Löffler reaction (also referred to as Hofmann–Löffler–Freytag reaction, Löffler–Freytag reaction, Löffler–Hofmann reaction, as well as Löffler's method) is an organic reaction in which a cyclic amine 2 (pyrrolidine or, in some cases, piperidine) is generated by thermal or photochemical decomposition of N-halogenated amine 1 in the presence of a strong acid (concentrated sulfuric acid or concentrated CF3CO2H). The Hofmann–Löffler–Freytag reaction proceeds via an intramolecular hydrogen atom transfer to a nitrogen-centered radical and is an example of a remote intramolecular free radical C–H functionalization.