Chapter 8 Alkenes and Alkynes II
... Boron becomes attached to less substituted carbon of double bond l Bulkier boron group approaches the less hindered carbon more easily l This orientation also allows a δ+ charge in the transition state to ...
... Boron becomes attached to less substituted carbon of double bond l Bulkier boron group approaches the less hindered carbon more easily l This orientation also allows a δ+ charge in the transition state to ...
Exp 4_Properties of Alcohols
... ion from hydrochloric acid (HCl), forming an alkyl chloride, as shown in the following equation. Zinc chloride (ZnCl2) is a catalyst for this reaction, which is called the Lucas test. The resultant alkyl chloride is insoluble in water and separates from the Lucas reagent (ZnCl2 in concentrated HCl), ...
... ion from hydrochloric acid (HCl), forming an alkyl chloride, as shown in the following equation. Zinc chloride (ZnCl2) is a catalyst for this reaction, which is called the Lucas test. The resultant alkyl chloride is insoluble in water and separates from the Lucas reagent (ZnCl2 in concentrated HCl), ...
X-ray Structure and Reactivity of (η4
... transfer hydrogenation of acetophenone with 2-propanol. The optical purity of (S)-1-phenylethanol (>99% ee, 0.2 M) in toluene was changed to 45% ee (65% ee with 2 mol % of 1) after heating with 4 mol % of 7 at 70 °C for 5 h, and 1-phenylethanol was obtained in 95% yield (96% yield with 2 mol % of 1) ...
... transfer hydrogenation of acetophenone with 2-propanol. The optical purity of (S)-1-phenylethanol (>99% ee, 0.2 M) in toluene was changed to 45% ee (65% ee with 2 mol % of 1) after heating with 4 mol % of 7 at 70 °C for 5 h, and 1-phenylethanol was obtained in 95% yield (96% yield with 2 mol % of 1) ...
In the bachelor thesis of Esther Schippers, research is
... therefore also be soluble in aqueous solvents. To achieve this, for instance an ionic charge can be brought in the molecule. Ions are often good soluble in aqueous solutions. 3. The scaffold should have a part of the molecule that can easily be synthesized with another functional group. When differe ...
... therefore also be soluble in aqueous solvents. To achieve this, for instance an ionic charge can be brought in the molecule. Ions are often good soluble in aqueous solutions. 3. The scaffold should have a part of the molecule that can easily be synthesized with another functional group. When differe ...
CBS Reduction
... History of CBS reduction and selectivity • In 1981 Itsuno et al reduced achiral ketones to chiral alcohols using alkoxy-amine-borane complexes in enantioselectivity and in high yield. • In 1987 E.J. Corey, with his co-workers, used oxazaborolidines to rapidlly reduce ketones in the presence of BH3- ...
... History of CBS reduction and selectivity • In 1981 Itsuno et al reduced achiral ketones to chiral alcohols using alkoxy-amine-borane complexes in enantioselectivity and in high yield. • In 1987 E.J. Corey, with his co-workers, used oxazaborolidines to rapidlly reduce ketones in the presence of BH3- ...
Methodology for the olefination of aldehydes and ketones via the Meyer-Schuster reaction
... serve as the active reagents are prepared combining triphenylphosphine first with a primary or secondary alkyl halide and subsequently with an appropriate base. Although a strong base is typically used (eg. alkyllithium), if the salt is sufficiently acidic, then a mild base, such as sodium bicarbona ...
... serve as the active reagents are prepared combining triphenylphosphine first with a primary or secondary alkyl halide and subsequently with an appropriate base. Although a strong base is typically used (eg. alkyllithium), if the salt is sufficiently acidic, then a mild base, such as sodium bicarbona ...
AS 2, Module 2
... 12 Hex-1-ene reacts rapidly with bromine in a solvent such as 1,1,1-trichloroethane (TCE). The reaction is carried out in a glass beaker and is exothermic. Using experimental data it is possible to determine the enthalpy of bromination of hex-1-ene and to determine the C Br bond enthalpy. ...
... 12 Hex-1-ene reacts rapidly with bromine in a solvent such as 1,1,1-trichloroethane (TCE). The reaction is carried out in a glass beaker and is exothermic. Using experimental data it is possible to determine the enthalpy of bromination of hex-1-ene and to determine the C Br bond enthalpy. ...
Chemistry 262 Quiz 2 Winter 2017 The following
... BONUS: AN ALTERNATIVE PATHWAY TO DIRECT REARRANGEMENT IS REPETITIVE ELIMINATION AND ADDITION. SHOW HOW THIS COULD EXPLAIN THE FORMATION OF 2-METHYL-2-PENTENE FROM 4-METHYL-2-PENTANOL IN AQUEOUS H2SO4: ...
... BONUS: AN ALTERNATIVE PATHWAY TO DIRECT REARRANGEMENT IS REPETITIVE ELIMINATION AND ADDITION. SHOW HOW THIS COULD EXPLAIN THE FORMATION OF 2-METHYL-2-PENTENE FROM 4-METHYL-2-PENTANOL IN AQUEOUS H2SO4: ...
Organic molecules with functional groups containing oxygen
... the son of a sail maker. He did his PhD in Lyons, working with Philippe Barbier who suggested that he study organomagnesium compounds. He published his thesis in 1900 and over the succeeding 10 years he studied the applications of organomagnesium reagents in synthesis. He was so successful that he w ...
... the son of a sail maker. He did his PhD in Lyons, working with Philippe Barbier who suggested that he study organomagnesium compounds. He published his thesis in 1900 and over the succeeding 10 years he studied the applications of organomagnesium reagents in synthesis. He was so successful that he w ...
Organolithium reagent
... completion if the acidic compound is 2 pKA units stronger than the lithium species, although in practice a larger pKA difference is required for useful rates of deprotonation of weakly acidic C-H acids. As alkyl groups are weakly electron donating, the basicity of the organolithium compound increase ...
... completion if the acidic compound is 2 pKA units stronger than the lithium species, although in practice a larger pKA difference is required for useful rates of deprotonation of weakly acidic C-H acids. As alkyl groups are weakly electron donating, the basicity of the organolithium compound increase ...
Synthesis of p-hydroxy alkyl benzoates
... purity esters can be produced with azeotropic distillation to simultaneously remove water and alcohol from the esters using aromatic and aliphatic hydrocarbons as entrainers. There are distinct advantages to using azeotropic distillation, including energy savings, increased recovery and ability to s ...
... purity esters can be produced with azeotropic distillation to simultaneously remove water and alcohol from the esters using aromatic and aliphatic hydrocarbons as entrainers. There are distinct advantages to using azeotropic distillation, including energy savings, increased recovery and ability to s ...
Results
... (H2O)2 (806, 839), (H2O)3 (871, 888), valamint CH2CH2 (681, 681), CH3CHCH2 (752, 748), (CH3)2CCH2 (796, 806) Solvatation: water cluster models. In these reactions the proton affinity of the medium is an important factor (the water in the medium does not participate in the reaction but has a catalyti ...
... (H2O)2 (806, 839), (H2O)3 (871, 888), valamint CH2CH2 (681, 681), CH3CHCH2 (752, 748), (CH3)2CCH2 (796, 806) Solvatation: water cluster models. In these reactions the proton affinity of the medium is an important factor (the water in the medium does not participate in the reaction but has a catalyti ...
File - the prayas tutorial
... Q. 6. Grignard reagents should be prepared under anhydrous conditions. Why ? Ans. Because Grignard reagents have a very strong affinity for H+ ions. In presence of water, they abstract H+ ions from water and form alkanes. To prevent this, they should be prepared under anhydrous conditions. Q. 7. Hal ...
... Q. 6. Grignard reagents should be prepared under anhydrous conditions. Why ? Ans. Because Grignard reagents have a very strong affinity for H+ ions. In presence of water, they abstract H+ ions from water and form alkanes. To prevent this, they should be prepared under anhydrous conditions. Q. 7. Hal ...
International Indian School Dammam
... (a) Write electrode reactions when a lead storage battery is discharging (b) Galvanized iron pipe does not rust even if the zinc coating is broken. Why? (c) Calculate the Λ0 of NH4OH from the following data: Λ 0 of NaOH = 257.7 Scm2mol-1 Λ 0 of NaCl = 140.6 Scm2mol-1 Λ 0 of NH4 Cl =129.8 Scm2mol-1 ( ...
... (a) Write electrode reactions when a lead storage battery is discharging (b) Galvanized iron pipe does not rust even if the zinc coating is broken. Why? (c) Calculate the Λ0 of NH4OH from the following data: Λ 0 of NaOH = 257.7 Scm2mol-1 Λ 0 of NaCl = 140.6 Scm2mol-1 Λ 0 of NH4 Cl =129.8 Scm2mol-1 ( ...
Microsoft Word
... substituents of the chiral sulfur atom: a lone pair of electrons, an oxygen atom and two different carbon ligands. It is well known that sulfoxides participate as neighbouring groups in a number of reactions. Sulfoxide group participation in halohydrin formation from cyclic and simple acyclic olefin ...
... substituents of the chiral sulfur atom: a lone pair of electrons, an oxygen atom and two different carbon ligands. It is well known that sulfoxides participate as neighbouring groups in a number of reactions. Sulfoxide group participation in halohydrin formation from cyclic and simple acyclic olefin ...
Molecules with Nitrogen and Their Reactions
... Condensation reactions involve the joining of two small molecules to form one larger molecule with the loss of a small molecule (such as water). ...
... Condensation reactions involve the joining of two small molecules to form one larger molecule with the loss of a small molecule (such as water). ...
NUCLEOPHILIC SUBSTITUTION & ELIMINATION ON Csp 3
... SN2 – putting it all (almost) together: - The substrate: 1o C (or, not so good, 2º C) - The nucleophile: good - The leaving group: low pKa of conjugate acid - The solvent: polar, aprotic (next slides) The reaction flow & the transition state: ...
... SN2 – putting it all (almost) together: - The substrate: 1o C (or, not so good, 2º C) - The nucleophile: good - The leaving group: low pKa of conjugate acid - The solvent: polar, aprotic (next slides) The reaction flow & the transition state: ...
ppt
... The aldehyde proton will couple to the protons on the -carbon with a typical coupling constant of J 2 Hz A carbonyl will slightly deshield the protons on the -carbon; typical chemical shift range is 2.0 - 2.5 ppm ...
... The aldehyde proton will couple to the protons on the -carbon with a typical coupling constant of J 2 Hz A carbonyl will slightly deshield the protons on the -carbon; typical chemical shift range is 2.0 - 2.5 ppm ...
RULE
... Reaction of alkenes with HBr: Step 1 - Slow addition of electrophilic proton to nucleophilic alkene (shown via “attack” by Nu: electrons) forms a carbocation intermediate Step 2 - rapid reaction of positively charged carbocation intermediate (E+) with negatively charged bromide ion (Nu:) generates ...
... Reaction of alkenes with HBr: Step 1 - Slow addition of electrophilic proton to nucleophilic alkene (shown via “attack” by Nu: electrons) forms a carbocation intermediate Step 2 - rapid reaction of positively charged carbocation intermediate (E+) with negatively charged bromide ion (Nu:) generates ...
proline catalyzed direct asymmetric aldol and mannich reactions
... revealed no appreciably better catalysts. Direct proline catalysis was considered to be particularly favorable because, as the authors noted, the reactions have several advantages over normal enolate Copyright © 2005 by Mirth Hoyt ...
... revealed no appreciably better catalysts. Direct proline catalysis was considered to be particularly favorable because, as the authors noted, the reactions have several advantages over normal enolate Copyright © 2005 by Mirth Hoyt ...
Slide 1
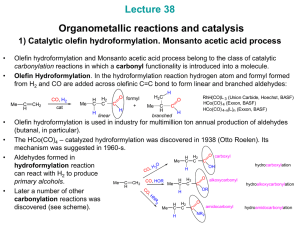
... Olefin hydroformylation and Monsanto acetic acid process belong to the class of catalytic carbonylation reactions in which a carbonyl functionality is introduced into a molecule. Olefin Hydroformylation. In the hydroformylation reaction hydrogen atom and formyl formed from H2 and CO are added across ...
... Olefin hydroformylation and Monsanto acetic acid process belong to the class of catalytic carbonylation reactions in which a carbonyl functionality is introduced into a molecule. Olefin Hydroformylation. In the hydroformylation reaction hydrogen atom and formyl formed from H2 and CO are added across ...
EXPERIMENT 5 (Organic Chemistry II) Pahlavan/Cherif
... The dehydration of alcohols to give alkenes is an important transformation and is an example of elimination reaction. Strong mineral acids such as sulfuric and phosphoric acid catalyze the reaction. Dehydration of an alcohol can follow either the E2 or the E1 mechanism. However, in each case, acid i ...
... The dehydration of alcohols to give alkenes is an important transformation and is an example of elimination reaction. Strong mineral acids such as sulfuric and phosphoric acid catalyze the reaction. Dehydration of an alcohol can follow either the E2 or the E1 mechanism. However, in each case, acid i ...
Dehydration of Alcohols - Dehydration of Cyclohexanol
... The dehydration of alcohols to give alkenes is an important transformation and is an example of elimination reaction. Strong mineral acids such as sulfuric and phosphoric acid catalyze the reaction. Dehydration of an alcohol can follow either the E2 or the E1 mechanism. However, in each case, acid i ...
... The dehydration of alcohols to give alkenes is an important transformation and is an example of elimination reaction. Strong mineral acids such as sulfuric and phosphoric acid catalyze the reaction. Dehydration of an alcohol can follow either the E2 or the E1 mechanism. However, in each case, acid i ...
Organic Synthesis Part 2
... sulfide (BH3.Me2S). Since boron is in the same period as aluminium, there are similarities in the mode of action to DIBAL-H (though not in terms of absolute reactivity). Note also the speed of reaction with carboxylic acids, since they rapidly form triacyloxyboranes. Reaction with alkenes and alkyne ...
... sulfide (BH3.Me2S). Since boron is in the same period as aluminium, there are similarities in the mode of action to DIBAL-H (though not in terms of absolute reactivity). Note also the speed of reaction with carboxylic acids, since they rapidly form triacyloxyboranes. Reaction with alkenes and alkyne ...
From carb acid till end ch 4
... 7. No tripod and gauze. The flask can be directly heated if the Bunsen is held and played across the flask so as not to overheat it. However, if the Bunsen is static the flask will overheat leading to bumping and charring. The gauze spreads the heat and reduces the probability of overheating. A few ...
... 7. No tripod and gauze. The flask can be directly heated if the Bunsen is held and played across the flask so as not to overheat it. However, if the Bunsen is static the flask will overheat leading to bumping and charring. The gauze spreads the heat and reduces the probability of overheating. A few ...
Hofmann–Löffler reaction

The Hofmann–Löffler reaction (also referred to as Hofmann–Löffler–Freytag reaction, Löffler–Freytag reaction, Löffler–Hofmann reaction, as well as Löffler's method) is an organic reaction in which a cyclic amine 2 (pyrrolidine or, in some cases, piperidine) is generated by thermal or photochemical decomposition of N-halogenated amine 1 in the presence of a strong acid (concentrated sulfuric acid or concentrated CF3CO2H). The Hofmann–Löffler–Freytag reaction proceeds via an intramolecular hydrogen atom transfer to a nitrogen-centered radical and is an example of a remote intramolecular free radical C–H functionalization.