Alkenes - Gadjah Mada University
... CH3CH=CHCH3 + H2 CH3CH2CH2CH3 Hydrogenation requires high temperatures and pressures as well as the presence of a catalyst (e.g. Ni). ...
... CH3CH=CHCH3 + H2 CH3CH2CH2CH3 Hydrogenation requires high temperatures and pressures as well as the presence of a catalyst (e.g. Ni). ...
amines
... alkylamines, the ending –amine is added to the name of the alkyl group that bears the nitrogen. When named as alkanamines, the alkyl group is named as an alkane and the -e ...
... alkylamines, the ending –amine is added to the name of the alkyl group that bears the nitrogen. When named as alkanamines, the alkyl group is named as an alkane and the -e ...
Organic Chemistry Fifth Edition
... Phase-transfer agents promote the solubility of ionic substances in nonpolar solvents. They transfer the ionic substance from an aqueous phase to a non-aqueous one. Phase-transfer agents increase the rates of reactions involving anions. The anion is relatively unsolvated and very reactive in nonpola ...
... Phase-transfer agents promote the solubility of ionic substances in nonpolar solvents. They transfer the ionic substance from an aqueous phase to a non-aqueous one. Phase-transfer agents increase the rates of reactions involving anions. The anion is relatively unsolvated and very reactive in nonpola ...
homogeneous catalysis
... This book has grown out of a graduate-level course on homogeneous catalysis that one of us taught at Northwestern University several times in the recent past. It deals with an interdisciplinary area of chemistry that offers challenging research problems. Industrial applications of homogeneous cataly ...
... This book has grown out of a graduate-level course on homogeneous catalysis that one of us taught at Northwestern University several times in the recent past. It deals with an interdisciplinary area of chemistry that offers challenging research problems. Industrial applications of homogeneous cataly ...
IOSR Journal of Applied Chemistry (IOSR-JAC)
... catalyzed dehydration. Iminium salt (R2C=N+R2) at the other extremes are very rapidly hydrolyzed by water and have to be prepared under rigorously anhydrous conditions. The facility of iminium salt hydrolysis has been put to use in a synthesis of secondary amines from primary amines which involves c ...
... catalyzed dehydration. Iminium salt (R2C=N+R2) at the other extremes are very rapidly hydrolyzed by water and have to be prepared under rigorously anhydrous conditions. The facility of iminium salt hydrolysis has been put to use in a synthesis of secondary amines from primary amines which involves c ...
top organomet chem-2006-19-207 pauson
... The idea that alkenes possessing electron-withdrawing groups are not adequate substrates for Pauson–Khand reactions has in recent years turned out not to be precise. Carretero has reported several examples involving electrondeficient alkenes 3), such as α,β-unsaturated ketones, esters, nitriles, sulf ...
... The idea that alkenes possessing electron-withdrawing groups are not adequate substrates for Pauson–Khand reactions has in recent years turned out not to be precise. Carretero has reported several examples involving electrondeficient alkenes 3), such as α,β-unsaturated ketones, esters, nitriles, sulf ...
Chapter 11
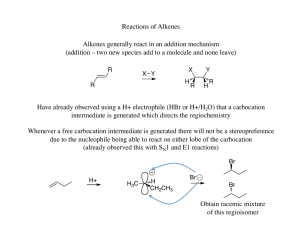
... The halonium ion thus directs both the regiochemistry (oxygen adds to the carbon that can best stabilize the partial positive charge) and the stereochemistry (due to the three membered ring the oxygen must add anti to the the bromine already present) ...
... The halonium ion thus directs both the regiochemistry (oxygen adds to the carbon that can best stabilize the partial positive charge) and the stereochemistry (due to the three membered ring the oxygen must add anti to the the bromine already present) ...
Chem 33 Lab - Santa Clara University
... laboratory until such time as any deficiencies have been addressed. 4. Use of cell phones, radios, iPods, and the like is not permitted in the laboratory. 5. Most organic solvents are flammable and should never be heated with an open flame. Hot plates or heating mantles are available for this purpos ...
... laboratory until such time as any deficiencies have been addressed. 4. Use of cell phones, radios, iPods, and the like is not permitted in the laboratory. 5. Most organic solvents are flammable and should never be heated with an open flame. Hot plates or heating mantles are available for this purpos ...
06_10_13.html
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
... The more stable the carbocation, the faster it is formed, and the faster the reaction rate. ...
RheniumCatalyzed Deoxydehydration of Diols and Polyols
... While the majority of oil, coal, and gas is used for energy production, the realization of an economy completely independent of fossil resources also requires biomass-based substitutes for polymers, medicine, pesticides, and so forth.[1] The evergrowing world population makes it questionable to use ...
... While the majority of oil, coal, and gas is used for energy production, the realization of an economy completely independent of fossil resources also requires biomass-based substitutes for polymers, medicine, pesticides, and so forth.[1] The evergrowing world population makes it questionable to use ...
Gas-Phase Reactions of Fe (CH2O)+ and Fe (CH2S)+ with Small
... bonding differences between Fe(CH2O)+ and Fe(CH2S)+. To do this, product ion structures were probed by collision-induced dissociation, specific ion-molecule reactions, and use of labeled compounds, and experimental bond energies were obtained by using ion-molecule bracketing and competitive collisio ...
... bonding differences between Fe(CH2O)+ and Fe(CH2S)+. To do this, product ion structures were probed by collision-induced dissociation, specific ion-molecule reactions, and use of labeled compounds, and experimental bond energies were obtained by using ion-molecule bracketing and competitive collisio ...
Modern Synthetic Methods for Copper-Mediated C(aryl
... Chan and co-workers[18] first described a collection of Nand O-arylation transformations under novel reaction conditions as early as in June 1997, but only part of the work was disclosed at this meeting. However in the first publication[14] it was already demonstrated that this method with a boronic ...
... Chan and co-workers[18] first described a collection of Nand O-arylation transformations under novel reaction conditions as early as in June 1997, but only part of the work was disclosed at this meeting. However in the first publication[14] it was already demonstrated that this method with a boronic ...
kinetic characterisation of catalysts for methanol synthesis
... catalyst called KATALCO which is the standard of Cu/ZnO/Al2O3. A new generation of catalysts also based on the copper catalyst is named APICO. Haldor Topsoe company produces CuO/ZnO/Al2O3 called MK-121 where the composition of the catalyst is: CuO (wt%) - 55; ZnO (wt%) 21-25; Al2O3 (wt%) 8-10; catal ...
... catalyst called KATALCO which is the standard of Cu/ZnO/Al2O3. A new generation of catalysts also based on the copper catalyst is named APICO. Haldor Topsoe company produces CuO/ZnO/Al2O3 called MK-121 where the composition of the catalyst is: CuO (wt%) - 55; ZnO (wt%) 21-25; Al2O3 (wt%) 8-10; catal ...
Chemistry 162 Workbook 10.6
... Namin g Alkenes, A lkynes, & Aromatics Please complete the following table either by providing the name or the structure of the compound. Don’t worry about cis and trans nomenclature but please use ortho, meta & ...
... Namin g Alkenes, A lkynes, & Aromatics Please complete the following table either by providing the name or the structure of the compound. Don’t worry about cis and trans nomenclature but please use ortho, meta & ...
The integration of flow reactors into synthetic organic chemistry
... laboratory practices have also become standardized to make the best use of these tools and associated pieces of equipment. A standard sequence for a reaction today and over a century ago would still be easily recognizable to both bench chemists (Figure 1). From a simple analysis of the individual pr ...
... laboratory practices have also become standardized to make the best use of these tools and associated pieces of equipment. A standard sequence for a reaction today and over a century ago would still be easily recognizable to both bench chemists (Figure 1). From a simple analysis of the individual pr ...
aldehydes and ketones
... In aldehydes, the carbonyl group is linked to either two hydrogen atom or one hydrogen atom and one carbon containing group such as alkyl, aryl or aralkyl group Examples ...
... In aldehydes, the carbonyl group is linked to either two hydrogen atom or one hydrogen atom and one carbon containing group such as alkyl, aryl or aralkyl group Examples ...
Chapter 16
... The alkoxide will reform a carbonyl (strong bond) with the good leaving group present ...
... The alkoxide will reform a carbonyl (strong bond) with the good leaving group present ...
Document
... increases because of increased surface area. • Alkenes are soluble in organic solvents and insoluble in water. • The C—C single bond between an alkyl group and one of the double bond carbons of an alkene is slightly polar because the sp3 hybridized alkyl carbon donates electron density to the sp2 hy ...
... increases because of increased surface area. • Alkenes are soluble in organic solvents and insoluble in water. • The C—C single bond between an alkyl group and one of the double bond carbons of an alkene is slightly polar because the sp3 hybridized alkyl carbon donates electron density to the sp2 hy ...
Four new mechanisms to learn: SN2 vs E2 and SN1 vs E1
... SN1 and E1 reactions are multistep reactions and also compete with one another. Both of these reactions begin with the same rate-limiting step of carbocation formation from an R-X compound. Carbocations (R+) are very reactive electron deficient carbon intermediates that typically follow one of three ...
... SN1 and E1 reactions are multistep reactions and also compete with one another. Both of these reactions begin with the same rate-limiting step of carbocation formation from an R-X compound. Carbocations (R+) are very reactive electron deficient carbon intermediates that typically follow one of three ...
Module II Reduction Reactions
... Lithium borohydride is prepared by reaction of NaBH4 with lithium bromide. It is more powerful reducing agent than NaBH4, reducing not only aldehydes, ketones, but also epoxides, esters and lactones (Scheme 6). This is due to the fact that the Li+ cation is stronger Lewis acid than Na+ cation. There ...
... Lithium borohydride is prepared by reaction of NaBH4 with lithium bromide. It is more powerful reducing agent than NaBH4, reducing not only aldehydes, ketones, but also epoxides, esters and lactones (Scheme 6). This is due to the fact that the Li+ cation is stronger Lewis acid than Na+ cation. There ...
Pseudoasymmetry as a Means for Distinguishing Meso
... chirality is a useful complement to the use of pseudoasymmetry since chemical-shift nonequivalence points unequivocally to the dl isomer, while the creation of a pseudoasymmetric center provides unambiguous evidence of the meso isomer. Thus, the combination can be used to make unambiguous assignment ...
... chirality is a useful complement to the use of pseudoasymmetry since chemical-shift nonequivalence points unequivocally to the dl isomer, while the creation of a pseudoasymmetric center provides unambiguous evidence of the meso isomer. Thus, the combination can be used to make unambiguous assignment ...
Chapter 22: Phenols. Alcohols contain an OH group bonded to an
... 22.3: Physical Properties (please read). Like other alcohols the OH group of phenols can participate in hydrogen bonding with other phenol molecules and to water. 22.4: Acidity of Phenols. Phenols are more acidic than ...
... 22.3: Physical Properties (please read). Like other alcohols the OH group of phenols can participate in hydrogen bonding with other phenol molecules and to water. 22.4: Acidity of Phenols. Phenols are more acidic than ...
Boron and Metal Catalyzed CC and CH Bond Formation
... Research efforts focused on the use of boron and metals to form new carboncarbon and carbon-hydrogen bonds are summarized in this dissertation. Several novel reactions have been developed. These include: the deoxygenation of benzylic alcohols using chloroboranes, alkenylation of benzylic alcohols us ...
... Research efforts focused on the use of boron and metals to form new carboncarbon and carbon-hydrogen bonds are summarized in this dissertation. Several novel reactions have been developed. These include: the deoxygenation of benzylic alcohols using chloroboranes, alkenylation of benzylic alcohols us ...
120 Chapter 24: Phenols. Alcohols contain an OH group bonded to
... and stabilizes the negative charge through inductive effects ...
... and stabilizes the negative charge through inductive effects ...
Synthesis and Characterisation of N
... I hereby certify that I am the sole author of this thesis and that no part of this thesis has been published or submitted for publication. I certify that, to the best of my knowledge, my thesis does not infringe upon anyone’s copyright nor violate any proprietary rights and that any ideas, technique ...
... I hereby certify that I am the sole author of this thesis and that no part of this thesis has been published or submitted for publication. I certify that, to the best of my knowledge, my thesis does not infringe upon anyone’s copyright nor violate any proprietary rights and that any ideas, technique ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.