Postprint
... until the synthesis by Vaska of his eponymous complex: [IrCl(CO)(PPh3)2] (Figure 1.a), a catalyst of the [L3MX]-type .4 Following parallel development with other platinum group metals, Shrock and Osborn (Figure 1.d) discovered that an [L4Rh]+X- complex could convert in situ to an [L2Rh]+X- active ca ...
... until the synthesis by Vaska of his eponymous complex: [IrCl(CO)(PPh3)2] (Figure 1.a), a catalyst of the [L3MX]-type .4 Following parallel development with other platinum group metals, Shrock and Osborn (Figure 1.d) discovered that an [L4Rh]+X- complex could convert in situ to an [L2Rh]+X- active ca ...
Stereoselective Construction of a β
... furfuryl ethers (E)- and (Z)-8, although the deprotonation site was affected by the base employed in the acyclic system.6 The results of the diastereoselective Wittig rearrangement of cyclic furfuryl ethers 8 are shown in Table 1. Treatment of (E)-8 with excess alkyllithium or LDA in THF afforded is ...
... furfuryl ethers (E)- and (Z)-8, although the deprotonation site was affected by the base employed in the acyclic system.6 The results of the diastereoselective Wittig rearrangement of cyclic furfuryl ethers 8 are shown in Table 1. Treatment of (E)-8 with excess alkyllithium or LDA in THF afforded is ...
coordination of some monodentate and hybrid multident ate
... I wish to express my gratitude to the members of the fluorine group both past and present. In particular, I am grateful to Dr’s Howard Clark, Alison Stuart and David Birdsall. Thanks goes to the project student, Antony Wood, for carrying out the experimental work related to PPh2(2 -CH 3 0 C6H4). I w ...
... I wish to express my gratitude to the members of the fluorine group both past and present. In particular, I am grateful to Dr’s Howard Clark, Alison Stuart and David Birdsall. Thanks goes to the project student, Antony Wood, for carrying out the experimental work related to PPh2(2 -CH 3 0 C6H4). I w ...
Chapter 14: Chemical Kinetics
... For a reaction to occur as a result of a specific collision, the collision must have enough energy to overcome the energy barrier (activation energy). As you saw in Gases (Unit 10) and Intermolecular Forces and the Liquid State (Unit 11), the molecules in a given sample have a Boltzmann distribution ...
... For a reaction to occur as a result of a specific collision, the collision must have enough energy to overcome the energy barrier (activation energy). As you saw in Gases (Unit 10) and Intermolecular Forces and the Liquid State (Unit 11), the molecules in a given sample have a Boltzmann distribution ...
Hydrogenation, Transfer Hydrogenat- ion and Hydrogen Transfer Reactions
... The word “chirality” is derived from the Greek, χειρ (kheir) which means “hand”. Our hands cannot be superimposed onto each other but are mirror images of each other. Chirality can be traced back to the beginning of the 1900s, when the phrase was first introduced by Lord Kelvin,3 whose original stat ...
... The word “chirality” is derived from the Greek, χειρ (kheir) which means “hand”. Our hands cannot be superimposed onto each other but are mirror images of each other. Chirality can be traced back to the beginning of the 1900s, when the phrase was first introduced by Lord Kelvin,3 whose original stat ...
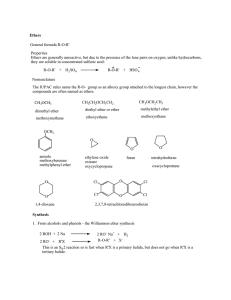
Alkenes
... When there are more than 12 carbons in the ring, trans-cycloalkenes are more stable than cis. The ring is large enough so the cycloalkene behaves much like a noncyclic one. ...
... When there are more than 12 carbons in the ring, trans-cycloalkenes are more stable than cis. The ring is large enough so the cycloalkene behaves much like a noncyclic one. ...
Document
... The carbonyl group is strongly polar but does not produce hydrogen bonding. As a result, the boiling points of aldehydes and ketones are higher than the nonpolar hydrocarbons and the alkyl halides but lower than those of alcohols. Formaldehyde is a gas at room temperature (b.p. = -21 C) but heavier ...
... The carbonyl group is strongly polar but does not produce hydrogen bonding. As a result, the boiling points of aldehydes and ketones are higher than the nonpolar hydrocarbons and the alkyl halides but lower than those of alcohols. Formaldehyde is a gas at room temperature (b.p. = -21 C) but heavier ...
Chapter 20 Amines - FIU Faculty Websites
... The acyl azide is obtained from an acid chloride Rearrangement of the acyl azide occurs with loss of N2, a very stable leaving group In the last step, the isocyanate is hydrolyzed by adding water ...
... The acyl azide is obtained from an acid chloride Rearrangement of the acyl azide occurs with loss of N2, a very stable leaving group In the last step, the isocyanate is hydrolyzed by adding water ...
HMDS+TMCS+Pyridine - Sigma
... excellent solvents for the reagent and the reaction products; they do not accelerate the rate of reaction. Polar solvents such as pyridine, dimethylformamide (DMF), dimethylsulfoxide (DMSO), tetrahydrofuran (THF), and acetonitrile are more often used because they can facilitate the reaction. Pyridin ...
... excellent solvents for the reagent and the reaction products; they do not accelerate the rate of reaction. Polar solvents such as pyridine, dimethylformamide (DMF), dimethylsulfoxide (DMSO), tetrahydrofuran (THF), and acetonitrile are more often used because they can facilitate the reaction. Pyridin ...
chm121 tutorial kit - Covenant University
... (a) an halogenated amide (b) a vicinal dihalide (c) a germinal dihalide (d) a vicinal halohydrin ...
... (a) an halogenated amide (b) a vicinal dihalide (c) a germinal dihalide (d) a vicinal halohydrin ...
New Organoselenium Methodology - ScholarBlogs
... Although the field of organoselenium chemistry dates back to the 19th century, it did not become a part of the mainstream of organic chemistry until the early 1970s. The apparent turning point came when Sharpless reported a mild method for the conversion of epoxides into allylic alcohols, which made ...
... Although the field of organoselenium chemistry dates back to the 19th century, it did not become a part of the mainstream of organic chemistry until the early 1970s. The apparent turning point came when Sharpless reported a mild method for the conversion of epoxides into allylic alcohols, which made ...
Oxidative Alihatic Carbon-Carbon Bond Cleavage Reactions
... of chemical bonds between two carbon atoms. The selective oxidative activation of carbon-carbon bonds is important due to potential applications in the utilization of biomass for fuel production, applications in wastewater treatment and bioremediation, and in developing new reactions for organic syn ...
... of chemical bonds between two carbon atoms. The selective oxidative activation of carbon-carbon bonds is important due to potential applications in the utilization of biomass for fuel production, applications in wastewater treatment and bioremediation, and in developing new reactions for organic syn ...
chemical kinetics type 1.mdi
... Photochemical reactions. Those reactions which take place in the presence of light are called photochemical reactions. Photosynthesis is an example of photochemical reaction. Photosensitization. The process in which a molecule that absorbs light transfers its extra energy to another molecule which m ...
... Photochemical reactions. Those reactions which take place in the presence of light are called photochemical reactions. Photosynthesis is an example of photochemical reaction. Photosensitization. The process in which a molecule that absorbs light transfers its extra energy to another molecule which m ...
File
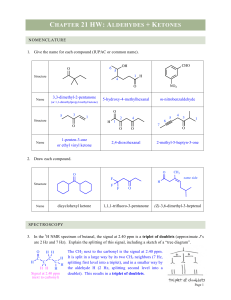
... The CH2 next to the carbonyl is the signal at 2.40 ppm. It is split in a large way by its two CH2 neighbors (7 Hz, splitting first level into a triplet), and in a smaller way by the aldehyde H (2 Hz, splitting second level into a doublet). This results in a triplet of doublets. ...
... The CH2 next to the carbonyl is the signal at 2.40 ppm. It is split in a large way by its two CH2 neighbors (7 Hz, splitting first level into a triplet), and in a smaller way by the aldehyde H (2 Hz, splitting second level into a doublet). This results in a triplet of doublets. ...
synthetic approaches for quinoline and isoquinoline
... S.N. PANDEYA, ALKA TYAGI Department of Pharmaceutical Chemistry, S.I.T.M, LKO, India Received: 10 March 2011, Revised and Accepted: 11 April 2011 ABSTRACT The Quinoline and Isoquinoline nucleus is found to be very important in pharmacy field. In recent years, a lot of synthe ...
... S.N. PANDEYA, ALKA TYAGI Department of Pharmaceutical Chemistry, S.I.T.M, LKO, India Received: 10 March 2011, Revised and Accepted: 11 April 2011 ABSTRACT The Quinoline and Isoquinoline nucleus is found to be very important in pharmacy field. In recent years, a lot of synthe ...
Handout
... CH3CH2CH2CH2-, nbutyl (CH3)2CHCH2-, isobutyl CH3CH2CH(CH3)-, sbutyl (CH3)3C-, t-butyl ...
... CH3CH2CH2CH2-, nbutyl (CH3)2CHCH2-, isobutyl CH3CH2CH(CH3)-, sbutyl (CH3)3C-, t-butyl ...
Marshall Gibson Dalton Manuscript B706663E revised
... because it is soluble and thermally stable in molten lactide, thereby allowing the process to be conducted in the melt phase in the absence of organic solvents. Numerous metal alkoxides are known to initiate lactide polymerisation3-5 and although details of the mechanism by which SnOct2 functions re ...
... because it is soluble and thermally stable in molten lactide, thereby allowing the process to be conducted in the melt phase in the absence of organic solvents. Numerous metal alkoxides are known to initiate lactide polymerisation3-5 and although details of the mechanism by which SnOct2 functions re ...
Chapter 15
... We can generally not isolate the aldehyde intermediate in aqueous solution. If we look at the mechanism you will see why. The first step is the formation of the chromate ester by attack of the alcohol hydroxyl onto the chromium with subsequent loss of water. This is an acid catalyzed esterification ...
... We can generally not isolate the aldehyde intermediate in aqueous solution. If we look at the mechanism you will see why. The first step is the formation of the chromate ester by attack of the alcohol hydroxyl onto the chromium with subsequent loss of water. This is an acid catalyzed esterification ...
New Applications for Sulfur-Based Leaving Groups in Synthesis
... was indeed the radical species originally suggested. Koelsch’s original sample was used for the EPR measurement and was shown to be stable under air after 23 years.2 It would take another three decades before the utility of organic radical chemistry was understood in greater depth. Work by Kharasch ...
... was indeed the radical species originally suggested. Koelsch’s original sample was used for the EPR measurement and was shown to be stable under air after 23 years.2 It would take another three decades before the utility of organic radical chemistry was understood in greater depth. Work by Kharasch ...
Synthesis of DiamidoPyrrolyl Molybdenum Complexes Relevant to Reduction DOI: 10.1021/ic100856n
... transition metal dinitrogen complex, [Ru(NH3)5(N2)]þ,2 inspired syntheses of other transition metal dinitrogen complexes in the hope that an abiological method of reducing dinitrogen under mild conditions could be devised, one that might eventually compete with or replace the Haber-Bosch process.3 W ...
... transition metal dinitrogen complex, [Ru(NH3)5(N2)]þ,2 inspired syntheses of other transition metal dinitrogen complexes in the hope that an abiological method of reducing dinitrogen under mild conditions could be devised, one that might eventually compete with or replace the Haber-Bosch process.3 W ...
Chapter 24. Amines
... They react with acids to form acid–base salts and they react with electrophiles Amines are stronger bases than alcohols, ethers, or water Amines establish an equilibrium with water in which the amine becomes protonated and hydroxide is produced ...
... They react with acids to form acid–base salts and they react with electrophiles Amines are stronger bases than alcohols, ethers, or water Amines establish an equilibrium with water in which the amine becomes protonated and hydroxide is produced ...
Carbonyl Compounds I. Aldehydes and Ketones
... We turn now to discuss a few specific addition reactions of the carbonyl groups of aldehydes and ketones. We shall not attempt to provide an extensive catalog of reactions, but will try to emphasize the principles involved with especially important reactions that are useful in synthesis. Grignard re ...
... We turn now to discuss a few specific addition reactions of the carbonyl groups of aldehydes and ketones. We shall not attempt to provide an extensive catalog of reactions, but will try to emphasize the principles involved with especially important reactions that are useful in synthesis. Grignard re ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.