Electrophilic Aromatic Substitution and Substituted Benzenes
... followed by nitration, and nitration first, followed by bromination. ...
... followed by nitration, and nitration first, followed by bromination. ...
VITA - Trace: Tennessee Research and Creative Exchange
... that strong σ-donor mono-, bi-, and tridentate ligands can be used to stabilize the metal ligand multiple bond (Figure 1.1A-C, respectively).10 In one example by Peters, it is found that the iron trisphosphinoborate complex (Figure 1.1C) can even activate an organic azide, however, the resulting imi ...
... that strong σ-donor mono-, bi-, and tridentate ligands can be used to stabilize the metal ligand multiple bond (Figure 1.1A-C, respectively).10 In one example by Peters, it is found that the iron trisphosphinoborate complex (Figure 1.1C) can even activate an organic azide, however, the resulting imi ...
4.9 Preparation of Alkyl Halides from Alcohols and Hydrogen Halides
... hybridized carbon is attached to C+, but is not availabe when H is attached to C+. Therefore,alkyl groups stabilize carbocations better than H does. ...
... hybridized carbon is attached to C+, but is not availabe when H is attached to C+. Therefore,alkyl groups stabilize carbocations better than H does. ...
Amidine: Structure, Reactivity and Complexation Behaviour
... interactions between the benzamidines and their respective environments in the two physical states[24]. Structure and Reactivity Most fundamental aspects in chemical and biochemical studies are the concepts of structure, energetic and reactivity as well as their interrelationships. In most chemical ...
... interactions between the benzamidines and their respective environments in the two physical states[24]. Structure and Reactivity Most fundamental aspects in chemical and biochemical studies are the concepts of structure, energetic and reactivity as well as their interrelationships. In most chemical ...
Alcohols and Thiols
... The alcoholic proton is also somewhat acidic. The pKa for alcohols runs between 15 and 18 - certainly more acidic than your average alkyne! The general trend in acidity is 1°>2°>3°, with t-butanol being about the least acidic alcohol you’ll find...this is why t-butoxide is used as a base in many of ...
... The alcoholic proton is also somewhat acidic. The pKa for alcohols runs between 15 and 18 - certainly more acidic than your average alkyne! The general trend in acidity is 1°>2°>3°, with t-butanol being about the least acidic alcohol you’ll find...this is why t-butoxide is used as a base in many of ...
PREPARATION OF ALDEHYDES
... EXAMPLES OF NUCLEOPHILIC ADDITION TO ALDEHYDES & KETONES Addition of HCN (neutral-basic conditions). CN Ө is a very good nucleophile (ionic nucleophile). The use of the actual compound HCN is not experimentally feasible, as it is a lethal gas, bp 26 oC. Addition of the elements of HCN to a C=O grou ...
... EXAMPLES OF NUCLEOPHILIC ADDITION TO ALDEHYDES & KETONES Addition of HCN (neutral-basic conditions). CN Ө is a very good nucleophile (ionic nucleophile). The use of the actual compound HCN is not experimentally feasible, as it is a lethal gas, bp 26 oC. Addition of the elements of HCN to a C=O grou ...
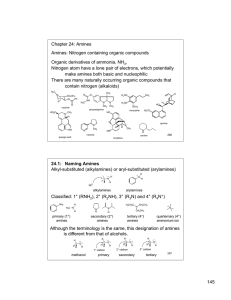
145 Chapter 24: Amines Amines: Nitrogen containing organic
... Synthesis of primary amines from the reaction of alkyl halides or tosylates with “ammonia equivalents” Azide ion is a very strong nucleophile and react with 1° or 2° alkyl halides or tosylates via an SN2 reaction. The resulting azide can be reduced to a 1° amine. H N N N ...
... Synthesis of primary amines from the reaction of alkyl halides or tosylates with “ammonia equivalents” Azide ion is a very strong nucleophile and react with 1° or 2° alkyl halides or tosylates via an SN2 reaction. The resulting azide can be reduced to a 1° amine. H N N N ...
Ethers and Epoxides
... Et-O-Et + H-Br EtBr + EtOH Et-O-Et + 2H-Br 2EtBr The alcohol produced reacts to generate a second molecule of alkyl halide. Phenyl ethers are slightly different, and cleave to give alkyl halides and ...
... Et-O-Et + H-Br EtBr + EtOH Et-O-Et + 2H-Br 2EtBr The alcohol produced reacts to generate a second molecule of alkyl halide. Phenyl ethers are slightly different, and cleave to give alkyl halides and ...
10 Haloalkanes and Haloarenes
... Direct halogenation of an alkane is a chain reaction and follows free radical mechanism. ii. This method of preparation gives the mixture of mono, di, tri and tetra halogen derivatives of an alkane and it is difficult to separate each component in pure form. eg. Preparation of methyl chloride by dir ...
... Direct halogenation of an alkane is a chain reaction and follows free radical mechanism. ii. This method of preparation gives the mixture of mono, di, tri and tetra halogen derivatives of an alkane and it is difficult to separate each component in pure form. eg. Preparation of methyl chloride by dir ...
1 THE BARTON-McCOMBIE REACTION STUART W. McCOMBIE 28
... natural products. They may also be employed to introduce deuterium or tritium in a site-specific manner. Reductive methods that involve ionic or highly polarized reagents or intermediates can be limited in their applicability: for example, competing reaction pathways including cationic rearrangement ...
... natural products. They may also be employed to introduce deuterium or tritium in a site-specific manner. Reductive methods that involve ionic or highly polarized reagents or intermediates can be limited in their applicability: for example, competing reaction pathways including cationic rearrangement ...
One-Electron Transformations of Paramagnetic Cobalt
... Generally, the Co(II) systems studied have been low-spin, rigidly square planar systems (porphyrins, salen-type ligands) as these compounds are considered suitable models for the corrin framework in Vitamin B12,24,25 although the addition of RX to Co(CN)53- shows that this reaction can be generalize ...
... Generally, the Co(II) systems studied have been low-spin, rigidly square planar systems (porphyrins, salen-type ligands) as these compounds are considered suitable models for the corrin framework in Vitamin B12,24,25 although the addition of RX to Co(CN)53- shows that this reaction can be generalize ...
Document
... First, he was fascinated by the chemistry of organocerium reagents in addition to easily enolizable electrophiles. Then he discovered that cerium was not a chelating metal and he stated the rules for the stereoselective reduction of functionalised ketones in the presence of a Lewis acid promoter. Re ...
... First, he was fascinated by the chemistry of organocerium reagents in addition to easily enolizable electrophiles. Then he discovered that cerium was not a chelating metal and he stated the rules for the stereoselective reduction of functionalised ketones in the presence of a Lewis acid promoter. Re ...
Direct organocatalytic enantioselective a-aminomethylation
... Trost and co-workers12 developed di-nuclear zinc organometallic complexes as catalyst for the direct catalytic enantioselective Mannich-type reactions between hydroxyarylketones and preformed imines. In addition, Jørgensen and co-workers have developed elegant direct asymmetric Mannich reactions inv ...
... Trost and co-workers12 developed di-nuclear zinc organometallic complexes as catalyst for the direct catalytic enantioselective Mannich-type reactions between hydroxyarylketones and preformed imines. In addition, Jørgensen and co-workers have developed elegant direct asymmetric Mannich reactions inv ...
molecules
... temperature. Mn(Br8TPPS) is a sulfonated hindered metalloporphyrin with electron withdrawing substituents at the -positions of the pyrroles. The electronegative groups on the pyrrole moieties enhance the reactivity of the metal oxo species and immobilization of the metalloporphyrin on ionexchange r ...
... temperature. Mn(Br8TPPS) is a sulfonated hindered metalloporphyrin with electron withdrawing substituents at the -positions of the pyrroles. The electronegative groups on the pyrrole moieties enhance the reactivity of the metal oxo species and immobilization of the metalloporphyrin on ionexchange r ...
Unsaturated hydrocarbons Alkenes
... When hydrogen halide is added to a symmetric alkene such as; 2-Butene [RCH=CHR], there is only one possible product because the two double-double bonded carbons are equivalent. ...
... When hydrogen halide is added to a symmetric alkene such as; 2-Butene [RCH=CHR], there is only one possible product because the two double-double bonded carbons are equivalent. ...
$doc.title
... They are important solvents and synthesis intermediates Phenols contain an OH group connected to a carbon in a benzene ring Methanol, CH3OH, called methyl alcohol, is a common solvent, a fuel addi1ve, ...
... They are important solvents and synthesis intermediates Phenols contain an OH group connected to a carbon in a benzene ring Methanol, CH3OH, called methyl alcohol, is a common solvent, a fuel addi1ve, ...
Chapter 5 - DORAS
... This coupling of Grignard reagents with alkyl, vinyl or aryl halides under Ni -catalysis provided an economic transformation, but the reaction itself is limited to halide partners that do not react with organomagnesium compounds. This first reported coupling reaction proved so successful that it is ...
... This coupling of Grignard reagents with alkyl, vinyl or aryl halides under Ni -catalysis provided an economic transformation, but the reaction itself is limited to halide partners that do not react with organomagnesium compounds. This first reported coupling reaction proved so successful that it is ...
Application of Novel Phosphine Ligands in Palladium
... rate-determining for the whole reaction. The third part of catalyzed reactions constitutes the transformations promoted by biomolecules, namely enzymes. Thus, nature can be considered as the world’s leading catalyst designer providing the most selective, active and complex catalysts. Biocatalysis is ...
... rate-determining for the whole reaction. The third part of catalyzed reactions constitutes the transformations promoted by biomolecules, namely enzymes. Thus, nature can be considered as the world’s leading catalyst designer providing the most selective, active and complex catalysts. Biocatalysis is ...
Organic Chemistry
... wavelength and molar extinction coefficient (Table 1 and Figure 2). However, the emission spectra of compound 1 appeared at a slightly longer wavelength with higher quantum efficiency than 2. This might attribute to the steric hindrance of the three 2-ethylhexyl groups that enhances the solubility o ...
... wavelength and molar extinction coefficient (Table 1 and Figure 2). However, the emission spectra of compound 1 appeared at a slightly longer wavelength with higher quantum efficiency than 2. This might attribute to the steric hindrance of the three 2-ethylhexyl groups that enhances the solubility o ...
4.8 Preparation of Alkyl Halides from Alcohols and Hydrogen
... electrons in this σ bond can be shared by positively charged carbon because the s orbital can overlap with the empty 2p orbital of positively charged carbon ...
... electrons in this σ bond can be shared by positively charged carbon because the s orbital can overlap with the empty 2p orbital of positively charged carbon ...
PDF - Nanyang Technological University
... a cooperative catalytic system to develop the enantioselective reaction between 1 a and 2 a. The reaction catalyzed by (S)proline/CuBr2 in DMF gave the desired product in 63 % yield and in a 1:1 diastereomeric ratio (d.r.), with virtually no enantioselectivity (4 %/3 % ee) (Table 1, entry 4). The re ...
... a cooperative catalytic system to develop the enantioselective reaction between 1 a and 2 a. The reaction catalyzed by (S)proline/CuBr2 in DMF gave the desired product in 63 % yield and in a 1:1 diastereomeric ratio (d.r.), with virtually no enantioselectivity (4 %/3 % ee) (Table 1, entry 4). The re ...
PowerPoint 演示文稿
... The stronger the nucleophilicity of a reagent, the more rapid the reaction. • Nucleophilicity roughly parallels basicity when comparing nucleophiles that have the same attacking atom. RO ...
... The stronger the nucleophilicity of a reagent, the more rapid the reaction. • Nucleophilicity roughly parallels basicity when comparing nucleophiles that have the same attacking atom. RO ...
HALO-ORGANICS – Fully functional fluorine 1
... An alternative which ensures only one isomer – the trans form – is isolated involves the carbonyl addition of a zinc carbenoid formed from the intermediate (Figure 2). 3 The adduct is acetylated and then reduced with zinc dust, resulting in the desired cyclopropyl ester. The alternative cis isomer ...
... An alternative which ensures only one isomer – the trans form – is isolated involves the carbonyl addition of a zinc carbenoid formed from the intermediate (Figure 2). 3 The adduct is acetylated and then reduced with zinc dust, resulting in the desired cyclopropyl ester. The alternative cis isomer ...
ETHER
... oxygen atom involves hydrogen and one involves carbon. Such compounds are called ethers. For example, dimethyl ether is isomeric with ethanol, and methyl ethyl ether is isomeric with propanol. Crown Ethers: These cyclic polyethers can form chemical complexes with metal cations (such as lithium, sodi ...
... oxygen atom involves hydrogen and one involves carbon. Such compounds are called ethers. For example, dimethyl ether is isomeric with ethanol, and methyl ethyl ether is isomeric with propanol. Crown Ethers: These cyclic polyethers can form chemical complexes with metal cations (such as lithium, sodi ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.