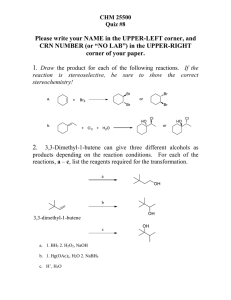
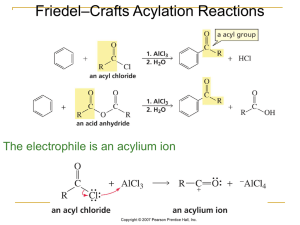
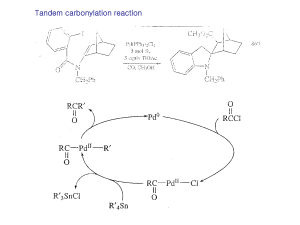
Organometallic Reactions and Catalysis
... – The carbene reacts with an alkene to form a metallocyclobutane intermediate. The intermediate can either revert to reactants or form new products. – Schrock metathesis catalysts are most effective and the most studied (available commercially). ...
... – The carbene reacts with an alkene to form a metallocyclobutane intermediate. The intermediate can either revert to reactants or form new products. – Schrock metathesis catalysts are most effective and the most studied (available commercially). ...
Exam 2 Review A
... You should be familiar with the detailed mechanisms of the SN1 and SN2 reactions. Rate determining steps are important to consider, as are the transition states associated with these steps. Compare and contrast the SN1 and SN2 reactions with respect to kinetics, nature of the electrophile [structure ...
... You should be familiar with the detailed mechanisms of the SN1 and SN2 reactions. Rate determining steps are important to consider, as are the transition states associated with these steps. Compare and contrast the SN1 and SN2 reactions with respect to kinetics, nature of the electrophile [structure ...
DESIGN OF CHIRAL IMINO- AND AMINOPYRIDINE LIGANDS
... Doctoral Thesis by Victor Hernández Olmos The synthesis of new molecules that can work as chiral ligands in metal catalyzed enantioselective reactions is currently an area of great interest.1,2 These ligands must be capable of coordinating with the metal ion, generating around it a highly asymmetric ...
... Doctoral Thesis by Victor Hernández Olmos The synthesis of new molecules that can work as chiral ligands in metal catalyzed enantioselective reactions is currently an area of great interest.1,2 These ligands must be capable of coordinating with the metal ion, generating around it a highly asymmetric ...
COUPLING REACTIONS IN ORGANIC SYNTHESIS
... For example, adding more reactant to the reaction shifts equilibrium to the right. More product is made, and some of the extra reactant is used up, so that the system can come back to its natural equilibrium. If products are somehow removed from the system, the reaction will also shift to the right, ...
... For example, adding more reactant to the reaction shifts equilibrium to the right. More product is made, and some of the extra reactant is used up, so that the system can come back to its natural equilibrium. If products are somehow removed from the system, the reaction will also shift to the right, ...
Enantiospecific skeleton expanding cross
... pathway for the modification and expansion of α-hydroxy carbonyls, which are themselves readily available from the chiral pool. Grignard reagents possessing a range of potential substituents are reacted under mild conditions with a triflate modified carbonyl at the α-carbon to bind a wide range of s ...
... pathway for the modification and expansion of α-hydroxy carbonyls, which are themselves readily available from the chiral pool. Grignard reagents possessing a range of potential substituents are reacted under mild conditions with a triflate modified carbonyl at the α-carbon to bind a wide range of s ...
Exam 2 Review A
... You should be familiar with the detailed mechanisms of the SN1 and SN2 reactions. Rate determining steps are important to consider, as are the transition states associated with these steps. Compare and contrast the SN1 and SN2 reactions with respect to kinetics, nature of the electrophile [structure ...
... You should be familiar with the detailed mechanisms of the SN1 and SN2 reactions. Rate determining steps are important to consider, as are the transition states associated with these steps. Compare and contrast the SN1 and SN2 reactions with respect to kinetics, nature of the electrophile [structure ...
Exam 2 Review A
... You should be familiar with the detailed mechanisms of the SN1 and SN2 reactions. Rate determining steps are important to consider, as are the transition states associated with these steps. Compare and contrast the SN1 and SN2 reactions with respect to kinetics, nature of the electrophile [structure ...
... You should be familiar with the detailed mechanisms of the SN1 and SN2 reactions. Rate determining steps are important to consider, as are the transition states associated with these steps. Compare and contrast the SN1 and SN2 reactions with respect to kinetics, nature of the electrophile [structure ...
Assignment 2 Group A and B
... 9) Which of the following alcohols can be prepared by the reaction of methyl formate with excess Grignard reagent? A) 1-pentanol B) 2-pentanol C) 3-pentanol D) 2-methyl-2-pentanol E) 3-methyl-3-pentanol 10) What reagent(s) would you use to accomplish the following conversion? ...
... 9) Which of the following alcohols can be prepared by the reaction of methyl formate with excess Grignard reagent? A) 1-pentanol B) 2-pentanol C) 3-pentanol D) 2-methyl-2-pentanol E) 3-methyl-3-pentanol 10) What reagent(s) would you use to accomplish the following conversion? ...
UNIVERSITAT ROVIRA I VIRGILI
... reactions, with an extra-ethene insertion in the growing polymer chain up to 23.2 % on addition of p-toluensulfonic acid, while the complexes containing the more rigid phosphine sulfonated ligand are orders of magnitude more active yielding ...
... reactions, with an extra-ethene insertion in the growing polymer chain up to 23.2 % on addition of p-toluensulfonic acid, while the complexes containing the more rigid phosphine sulfonated ligand are orders of magnitude more active yielding ...
Organic Tutorial 1st Year MT03
... Peter Sykes,“A Guidebook to Mechanism in Organic Chemistry”, and Eames & Peach “Stereochemistry at a Glance”. Notes and Questions a) Summary on not more than 6 sides. This should outline the possible mechanisms and the evidence on which they are based, in particular the evidence for inversion during ...
... Peter Sykes,“A Guidebook to Mechanism in Organic Chemistry”, and Eames & Peach “Stereochemistry at a Glance”. Notes and Questions a) Summary on not more than 6 sides. This should outline the possible mechanisms and the evidence on which they are based, in particular the evidence for inversion during ...
Heck Reactions
... catalyst). A general ratio for high activity system is 2:1. Pd(II) precatalysts include Pd(OAc)2, PdCl2(CH3CN), Pd(PPh3)2Cl2, and Pd[(allyl)Cl]2. These complexes are air stable and reduced by phosphines, water, and amines. In most cases, 5-20 mol% catalyst is used, even though more stable catalysts ...
... catalyst). A general ratio for high activity system is 2:1. Pd(II) precatalysts include Pd(OAc)2, PdCl2(CH3CN), Pd(PPh3)2Cl2, and Pd[(allyl)Cl]2. These complexes are air stable and reduced by phosphines, water, and amines. In most cases, 5-20 mol% catalyst is used, even though more stable catalysts ...
Oxidative Addition
... 1) A vacant 2e site is always required on the metal. We can either start with a 16e complex or a 2e site must be opened up in an 18e complex by the loss of a ligand producing a 16e intermediate species. 2) The starting metal complex of a given oxidation state must also have a stable oxidation state ...
... 1) A vacant 2e site is always required on the metal. We can either start with a 16e complex or a 2e site must be opened up in an 18e complex by the loss of a ligand producing a 16e intermediate species. 2) The starting metal complex of a given oxidation state must also have a stable oxidation state ...
Combustion, Addition and Elimination Objective Combustion Example
... It may be possible to have more than one product from a reaction. Example: but-1-ene + hydrogen fluoride. ...
... It may be possible to have more than one product from a reaction. Example: but-1-ene + hydrogen fluoride. ...
Slides for Chapter 1-4 - Department of Chemistry and Physics
... Alkyl Halides React with Nucleophiles and Bases Alkyl halides are polarized at the carbon-halide bond, ...
... Alkyl Halides React with Nucleophiles and Bases Alkyl halides are polarized at the carbon-halide bond, ...
InorgCh14.1
... i. Free 13CO + complex gives no labeled acyl product This rules out mechanism #1, CO insertion ii. Reverse reaction gives 100% cis 13CO and R Both mechanism #2 and mechanism #3 are consistent with this result ...
... i. Free 13CO + complex gives no labeled acyl product This rules out mechanism #1, CO insertion ii. Reverse reaction gives 100% cis 13CO and R Both mechanism #2 and mechanism #3 are consistent with this result ...
Hung Anh Duong Project Synopsis
... pharmaceuticals and materials. A popular method to gain access to this type of compound is via the Nobel Prize winning metal-catalyzed cross-coupling reactions. While being the most-scaled technology for biaryl coupling, the palladium-catalyzed Suzuki-Miyaura reactions are not without drawbacks. Pal ...
... pharmaceuticals and materials. A popular method to gain access to this type of compound is via the Nobel Prize winning metal-catalyzed cross-coupling reactions. While being the most-scaled technology for biaryl coupling, the palladium-catalyzed Suzuki-Miyaura reactions are not without drawbacks. Pal ...
슬라이드 1
... complexes can be obtained from Pd(II) salts and allyl acetates and other compounds with potential leaving groups in an allylic poistion. The p-allyl complexes can be isolated as halide-bridged dimers. ...
... complexes can be obtained from Pd(II) salts and allyl acetates and other compounds with potential leaving groups in an allylic poistion. The p-allyl complexes can be isolated as halide-bridged dimers. ...
Slide 1
... One needs to consider an alternative if there is another functional group present in the compound ...
... One needs to consider an alternative if there is another functional group present in the compound ...
Transition Metal Catalyzed Coupling Reactions
... nucleophiles available for cross-coupling processes as alternatives to Grignards reagents. ...
... nucleophiles available for cross-coupling processes as alternatives to Grignards reagents. ...
$doc.title
... http://www.chem.wisc.edu/areas/clc (Resource page) Reactions of Alcohols #8: Reaction of a 1° Alcohol with Hydrogen Halides ...
... http://www.chem.wisc.edu/areas/clc (Resource page) Reactions of Alcohols #8: Reaction of a 1° Alcohol with Hydrogen Halides ...
슬라이드 1
... complexes can be obtained from Pd(II) salts and allyl acetates and other compounds with potential leaving groups in an allylic poistion. The p-allyl complexes can be isolated as halide-bridged dimers. ...
... complexes can be obtained from Pd(II) salts and allyl acetates and other compounds with potential leaving groups in an allylic poistion. The p-allyl complexes can be isolated as halide-bridged dimers. ...
슬라이드 1
... A soluble bis-phosphine complexes, Ni(dppe)2Cl2, is a particularly effective catalyst. The main distinction between this reaction and Pd-catalyzed cross coupling is that the nickel reaction can be more readily applied to saturated alkyl groups because of a reduced tendency for b-elimination. ...
... A soluble bis-phosphine complexes, Ni(dppe)2Cl2, is a particularly effective catalyst. The main distinction between this reaction and Pd-catalyzed cross coupling is that the nickel reaction can be more readily applied to saturated alkyl groups because of a reduced tendency for b-elimination. ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.