Hydroformylation Hydroformylation, also known as oxo synthesis or
... aldehyde and the complex [HCo(CO)3]. It is important that the rate of migatory insertion of the carbonyl into the carbon-metal bond of the alkyl is fast; in systems where the migatory insertion does not occur (such as nickel hydride tristriphenyl phosphite), the reaction of the hydride with the alke ...
... aldehyde and the complex [HCo(CO)3]. It is important that the rate of migatory insertion of the carbonyl into the carbon-metal bond of the alkyl is fast; in systems where the migatory insertion does not occur (such as nickel hydride tristriphenyl phosphite), the reaction of the hydride with the alke ...
No Slide Title
... Enthalphy (DHo): the heat given off or absorbed during a reaction Entropy (DSo): a measure of freedom of motion If DSo is small compared to DHo, DGo ~ DHo ...
... Enthalphy (DHo): the heat given off or absorbed during a reaction Entropy (DSo): a measure of freedom of motion If DSo is small compared to DHo, DGo ~ DHo ...
Answer Key to Assignment #7
... 6. Describe how you would carry out the following transformations. More than one step may be necessary. The aldehyde must be protected first, ...
... 6. Describe how you would carry out the following transformations. More than one step may be necessary. The aldehyde must be protected first, ...
( i ) in enantioselective nhk reaction
... Cr(II) readily inserts into allyl-, alkenyl-, alkynyl-, propargyland aryl halides and sulfonates under aprotic conditions, giving rise to the corresponding organochromium(III) reagents Traces of nickel salts exert a catalytic effect on the formation of the C-Cr(III) bond ...
... Cr(II) readily inserts into allyl-, alkenyl-, alkynyl-, propargyland aryl halides and sulfonates under aprotic conditions, giving rise to the corresponding organochromium(III) reagents Traces of nickel salts exert a catalytic effect on the formation of the C-Cr(III) bond ...
Organic Chemistry - Snow College | It's SNOWing
... • have lower boiling points and are less soluble in water than alcohols of comparable MW ...
... • have lower boiling points and are less soluble in water than alcohols of comparable MW ...
File
... bis(diphenylphosphino)ethane (‘dppe’) in n-butanol leads to the formation of a highlyeffective system for the hydrogenation of alkenes (a) Draw a catalytic cycle for the conversion of ethene and hydrogen to ethane with this system ...
... bis(diphenylphosphino)ethane (‘dppe’) in n-butanol leads to the formation of a highlyeffective system for the hydrogenation of alkenes (a) Draw a catalytic cycle for the conversion of ethene and hydrogen to ethane with this system ...
chapter 8 part 2
... Consider the following reaction Outline a likely mechanism for the solvomercuration step of this ether synthesis Show how you would use solvomercuration-demercuration to prepare tert-butyl methyl ether Why would one use Hg(OCCF3)2 instead of Hg(Oac)2 ...
... Consider the following reaction Outline a likely mechanism for the solvomercuration step of this ether synthesis Show how you would use solvomercuration-demercuration to prepare tert-butyl methyl ether Why would one use Hg(OCCF3)2 instead of Hg(Oac)2 ...
organic quiz 2
... 17) In a reaction between hex-2-ene and hydrochloric acid (aqueous hydrogen chloride), which of the following will be the product(s)? a) 1-chlorohexane only b) 2-chlorohexane only c) 3-chlorohexane only d) both (b) and (c) 18) DNA is a natural polymer composed of a) glucose monomers b) nucleotide mo ...
... 17) In a reaction between hex-2-ene and hydrochloric acid (aqueous hydrogen chloride), which of the following will be the product(s)? a) 1-chlorohexane only b) 2-chlorohexane only c) 3-chlorohexane only d) both (b) and (c) 18) DNA is a natural polymer composed of a) glucose monomers b) nucleotide mo ...
Current Research Click Here
... The mini GC allows one to determine the relative percentages of compounds in a substance. It also allows one to follow the progress of the reaction by monitoring the decrease in concentration using the mini GC. For additional information: http://www.vernier.com/products/sensors/gc2-mini/ . Two vide ...
... The mini GC allows one to determine the relative percentages of compounds in a substance. It also allows one to follow the progress of the reaction by monitoring the decrease in concentration using the mini GC. For additional information: http://www.vernier.com/products/sensors/gc2-mini/ . Two vide ...
reactions of the carbonyl group in aldehydes and ketones
... KETONES L.O.: Outline the mechanism for nucleophilic addition reaction of aldehydes and ketones with hydrides. ...
... KETONES L.O.: Outline the mechanism for nucleophilic addition reaction of aldehydes and ketones with hydrides. ...
Microsoft Word - Final Exam Study Guide
... alcohols/ethers/epoxides, multistep synthesis, protecting groups, redox reactions, reagents for redox reactions, Grignard reaction 1. Mechanisms. These are the very basic types of mechanisms. You should also be able to explain regiochemistry and stereochemistry outcomes, as well as rearrangements, e ...
... alcohols/ethers/epoxides, multistep synthesis, protecting groups, redox reactions, reagents for redox reactions, Grignard reaction 1. Mechanisms. These are the very basic types of mechanisms. You should also be able to explain regiochemistry and stereochemistry outcomes, as well as rearrangements, e ...
Document
... • Imine formation is also a nucleophilic addition. • There is a different end result here, though as elimination of water occurs. • The initial reaction is attack of the amine on the carbonyl to give the alkoxide intermediate as normal. • Following protonation of the alkoxide and loss of the proton ...
... • Imine formation is also a nucleophilic addition. • There is a different end result here, though as elimination of water occurs. • The initial reaction is attack of the amine on the carbonyl to give the alkoxide intermediate as normal. • Following protonation of the alkoxide and loss of the proton ...
Lesson 4 halogenoalkanes
... The following terms are often used when discussing substitution reactions – Y + R3C-X R3C-Y + X ...
... The following terms are often used when discussing substitution reactions – Y + R3C-X R3C-Y + X ...
Name - Clark College
... Please work and place your answers in the spaces provided. Show your work for maximum credit! The last page may be torn off and used for scratch paper. Do not write on the IR/NMR sheet. Bring all material up when you are finished. 1. Provide the reagents for the following 2-step transformation. [6] ...
... Please work and place your answers in the spaces provided. Show your work for maximum credit! The last page may be torn off and used for scratch paper. Do not write on the IR/NMR sheet. Bring all material up when you are finished. 1. Provide the reagents for the following 2-step transformation. [6] ...
Chapter 7 - Alkenes and Alkynes I less substituted alkene due to
... - An addition reaction has a product, an alkane, that results from the addition of hydrogen to an alkene - Alkanes are said to be saturated compounds - Alkenes and alkynes, due to their π bonds, are said to be unsaturated - Catalytic hydrogenation is, for this case, synonymous with addition reaction ...
... - An addition reaction has a product, an alkane, that results from the addition of hydrogen to an alkene - Alkanes are said to be saturated compounds - Alkenes and alkynes, due to their π bonds, are said to be unsaturated - Catalytic hydrogenation is, for this case, synonymous with addition reaction ...
Organometallic Chemistry
... • Every orbital wants to be “used", i.e. contribute to binding an electron pair The strength of the preference for electron-precise structures depends on the position of the element in the periodic table • For early transition metals, 18-e is often unattainable for steric reasons - the required numb ...
... • Every orbital wants to be “used", i.e. contribute to binding an electron pair The strength of the preference for electron-precise structures depends on the position of the element in the periodic table • For early transition metals, 18-e is often unattainable for steric reasons - the required numb ...
Mon Feb 15 lecture
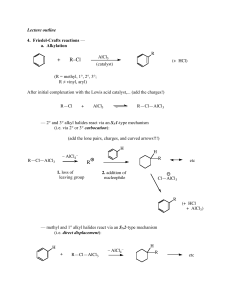
... (R = methyl, 1°, 2°, 3°; R ≠ vinyl, aryl) After initial complexation with the Lewis acid catalyst,... (add the charges!) R—Cl ...
... (R = methyl, 1°, 2°, 3°; R ≠ vinyl, aryl) After initial complexation with the Lewis acid catalyst,... (add the charges!) R—Cl ...
Answers
... carboxylic acid proton to become water. The catalytic amount (0.1 equiv.) of hydroxide left will do the beta hydroxycarbonyl elimination. 5. These two metabolic reactions may occur in either direction. In which direction are they additions? What was added in each case? In which direction are they el ...
... carboxylic acid proton to become water. The catalytic amount (0.1 equiv.) of hydroxide left will do the beta hydroxycarbonyl elimination. 5. These two metabolic reactions may occur in either direction. In which direction are they additions? What was added in each case? In which direction are they el ...
Catalytic Hydrogenation of Alkenes: Relative Stability of
... The thermodynamically most stable alkene or alkene mixture usually results from unimolecular dehydration in the presence of acid. Whenever possible, the most highly substituted system is generated. Trans-substituted alkenes predominate if there is a choice. Treatment of primary alcohols with minera ...
... The thermodynamically most stable alkene or alkene mixture usually results from unimolecular dehydration in the presence of acid. Whenever possible, the most highly substituted system is generated. Trans-substituted alkenes predominate if there is a choice. Treatment of primary alcohols with minera ...
Microsoft Word
... The foundation of synthetic organic chemistry rests on the ability to form and manipulate carbon-carbon bonds. The increasing demand of coupled products in chemical and pharmaceutical industries has prompted the development of several transition metal catalysts, which aim to exert the highest turnov ...
... The foundation of synthetic organic chemistry rests on the ability to form and manipulate carbon-carbon bonds. The increasing demand of coupled products in chemical and pharmaceutical industries has prompted the development of several transition metal catalysts, which aim to exert the highest turnov ...
I (21 points) Complete the following reactions by providing starting
... A. (JOC, 2008, ASAP, Loh) Chemists have been studying the Barbier-Grignard reactions with the goal of affecting the carbon-carbon bond forming reaction in solvents like water. Recent developments include the use of indium metal catalysts that react through single electron transfer mechanisms. Show t ...
... A. (JOC, 2008, ASAP, Loh) Chemists have been studying the Barbier-Grignard reactions with the goal of affecting the carbon-carbon bond forming reaction in solvents like water. Recent developments include the use of indium metal catalysts that react through single electron transfer mechanisms. Show t ...
Chris Sprout”s
... ligands are used in the dialkylzinc addition to aldehydes. I have found that Me2Zn, in the presence of our ligands, adds to 2-naphthaldehyde, 4-Cl-benzaldehyde, and benzaldehyde in 86%, 84%, and 81% enantiomeric excess (ee) respectively. Inspiration for this project stems from the need for high-thro ...
... ligands are used in the dialkylzinc addition to aldehydes. I have found that Me2Zn, in the presence of our ligands, adds to 2-naphthaldehyde, 4-Cl-benzaldehyde, and benzaldehyde in 86%, 84%, and 81% enantiomeric excess (ee) respectively. Inspiration for this project stems from the need for high-thro ...
LOYOLA COLLEGE (AUTONOMOUS), CHENNAI – 600 034
... 18. How are oxidizing agents chemoselective? Explain with suitable examples. 19. Explain the mechanism of Aldol condensation and Wittig reaction. 20. Discuss the mechanism of Baeyer Villiger reaction with suitable example. 21. Discuss on the electroorganic synthesis by reduction with suitable exampl ...
... 18. How are oxidizing agents chemoselective? Explain with suitable examples. 19. Explain the mechanism of Aldol condensation and Wittig reaction. 20. Discuss the mechanism of Baeyer Villiger reaction with suitable example. 21. Discuss on the electroorganic synthesis by reduction with suitable exampl ...
Organometallic Compounds
... Ziegler-Natta catalyst. Ziegler-Natta catalyst consists of TiCl4 and Et3Al with Et2AlCl as co-catalyst. ...
... Ziegler-Natta catalyst. Ziegler-Natta catalyst consists of TiCl4 and Et3Al with Et2AlCl as co-catalyst. ...
Senior Science topics Programme
... understanding of the reactivity of different chemical species with experimental observations to deduce the most likely sequence of elementary steps and thus the mechanism of a particular reaction. Knowledge of reaction mechanisms facilitates scientists to plan for synthesising new compounds from som ...
... understanding of the reactivity of different chemical species with experimental observations to deduce the most likely sequence of elementary steps and thus the mechanism of a particular reaction. Knowledge of reaction mechanisms facilitates scientists to plan for synthesising new compounds from som ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.