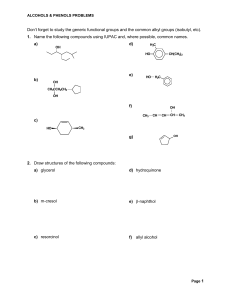
Don`t forget to study the generic functional groups and the common
... Also draw the structures of any carbonyl compounds that can be reduced by a LiAlH4 to prepare these compounds a) ...
... Also draw the structures of any carbonyl compounds that can be reduced by a LiAlH4 to prepare these compounds a) ...
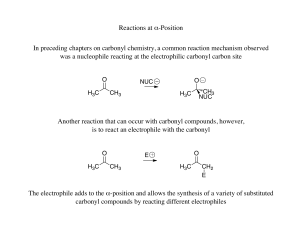
Reactions at α-Position In preceding chapters on carbonyl chemistry
... Alkylation of aldehydes can sometimes be problematic because the aldehyde carbonyl is more reactive than a ketone, therefore the enolate formed can react with the carbonyl ...
... Alkylation of aldehydes can sometimes be problematic because the aldehyde carbonyl is more reactive than a ketone, therefore the enolate formed can react with the carbonyl ...
Proofs to - Research Explorer
... The character of the iron to C-alpha bond in a series of alkylidenes and heteroatom (E) stabilised carbenes has been the focus of a series of theory investigations on complexes of the type [Fe{C(ER’x)R}L2Cp]+ [36,37]. There is a clear correlation between the extent of heteroatom stabilisation and t ...
... The character of the iron to C-alpha bond in a series of alkylidenes and heteroatom (E) stabilised carbenes has been the focus of a series of theory investigations on complexes of the type [Fe{C(ER’x)R}L2Cp]+ [36,37]. There is a clear correlation between the extent of heteroatom stabilisation and t ...
Carbonyl Condensation Reactions
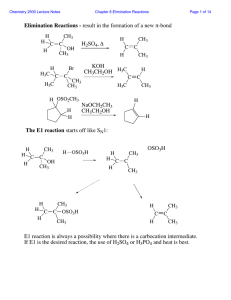
... mechanisms of elimination, E1 and E2, which were discussed in Chapter 8. The E1cB mechanism involves two steps, and proceeds by way of an anionic intermediate. Regular alcohols dehydrate only in the presence of acid but not base, because hydroxide is a poor leaving group. When the hydroxy group is β ...
... mechanisms of elimination, E1 and E2, which were discussed in Chapter 8. The E1cB mechanism involves two steps, and proceeds by way of an anionic intermediate. Regular alcohols dehydrate only in the presence of acid but not base, because hydroxide is a poor leaving group. When the hydroxy group is β ...
Efficient hydrogenation of organic carbonates, carbamates and
... recently, rare examples of the significantly more difficult hydrogenation of esters4,5 and amides6,7 have also been reported. However, the hydrogenation of organic carbonates and carbamates remains a major challenge. Indeed, as far as we know, catalytic hydrogenation of these important families of com ...
... recently, rare examples of the significantly more difficult hydrogenation of esters4,5 and amides6,7 have also been reported. However, the hydrogenation of organic carbonates and carbamates remains a major challenge. Indeed, as far as we know, catalytic hydrogenation of these important families of com ...
RUMPLE-DISSERTATION-2014 - SMARTech Home
... Eckert. The opportunity to work with such skilled scientists and kind mentors is a rare one, and I am extremely glad I had the opportunity to learn from them. I have always been in awe of their brilliance (I’m pretty sure they have each forgotten more than I’ll ever learn, and they still know so muc ...
... Eckert. The opportunity to work with such skilled scientists and kind mentors is a rare one, and I am extremely glad I had the opportunity to learn from them. I have always been in awe of their brilliance (I’m pretty sure they have each forgotten more than I’ll ever learn, and they still know so muc ...
Catalytic Asymmetric Induction. Highly Enantioselective Addition of
... N M R spectra compared to liquid-state spectra makes the interpretation of the spectra cumbersome due to peak overlap. In some cases, such as porphyrins or phthalocyanines, these problems arising in the I3C spectra have been overcome by observing 15N instead of 13C.2-4 This requires considerable syn ...
... N M R spectra compared to liquid-state spectra makes the interpretation of the spectra cumbersome due to peak overlap. In some cases, such as porphyrins or phthalocyanines, these problems arising in the I3C spectra have been overcome by observing 15N instead of 13C.2-4 This requires considerable syn ...
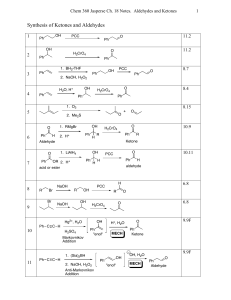
Synthesis of Ketones and Aldehydes
... Classification of Mechanisms Associated With Ketone/Aldehyde Reactions. • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict wh ...
... Classification of Mechanisms Associated With Ketone/Aldehyde Reactions. • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict wh ...
Chapter 17 Amines
... Secondary amines: both aryl amines and alkyl amines react with nitrous acid to yield N-nitrosoamines(亚硝胺) N O NHCH3 + NaNO2 + 2 HCl ...
... Secondary amines: both aryl amines and alkyl amines react with nitrous acid to yield N-nitrosoamines(亚硝胺) N O NHCH3 + NaNO2 + 2 HCl ...
The Organometallic Chemistry Of Transition Metals
... the interaction between inorganic metal species and organic molecules. In the related metal–organic compound area, in contrast, the organic fragment is bound only by metal–heteroatom bonds [e.g., Ti(OMe)4 ]. The organometallic field has provided a series of important conceptual insights, surprising ...
... the interaction between inorganic metal species and organic molecules. In the related metal–organic compound area, in contrast, the organic fragment is bound only by metal–heteroatom bonds [e.g., Ti(OMe)4 ]. The organometallic field has provided a series of important conceptual insights, surprising ...
Synthesis of Ketones and Aldehydes
... Classification of Mechanisms Associated With Ketone/Aldehyde Reactions. • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict wh ...
... Classification of Mechanisms Associated With Ketone/Aldehyde Reactions. • There may seem to be a dizzying number of mechanisms this chapter. But all of them simplify into some combination of acid- or base-catalyzed addition reaction, elimination reaction and/or substitution reaction. • To predict wh ...
Novel Transition Metal-Catalysed Syntheses of Carboxylic Acid
... been used extensively in alkene metathesis and this reactivity has been exploited in sequential reactions, particularly with respect to sequential metathesis/hydrogenation. The first reaction of this type was reported by McLain and co-workers who showed that an ethylene/methacrylate copolymer formed ...
... been used extensively in alkene metathesis and this reactivity has been exploited in sequential reactions, particularly with respect to sequential metathesis/hydrogenation. The first reaction of this type was reported by McLain and co-workers who showed that an ethylene/methacrylate copolymer formed ...
APPROACHES TO CARBOHYDRATE-BASED CHEMICAL LIBRARIES: THE
... isolated from animal sources, plant extracts, and microbial fermentations. The leads are then laboriously refined into drug candidates through a process of systematic optimization. Sequential modifications of the lead compounds are individually synthesized and tested for activity, with beneficial ch ...
... isolated from animal sources, plant extracts, and microbial fermentations. The leads are then laboriously refined into drug candidates through a process of systematic optimization. Sequential modifications of the lead compounds are individually synthesized and tested for activity, with beneficial ch ...
Document
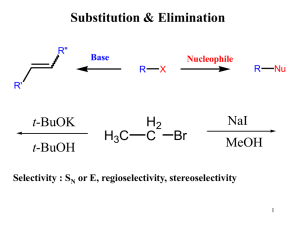
... • Because alkyl tosylates have good leaving groups, they undergo both nucleophilic substitution and elimination, exactly as alkyl halides do. ...
... • Because alkyl tosylates have good leaving groups, they undergo both nucleophilic substitution and elimination, exactly as alkyl halides do. ...
Iridium(III) and Rhodium(III) compounds of dipyridyl-N
... acetonitrile showed fragmentation of molecular ion peaks at m/z = 470 and 435 due to ([(η5 -C5 Me5 )Rh {(C5 H4 N)2 C=N-Me}Cl])+ ([2]PF6 -PF6 )+ and ([(η5 C5 Me5 )Rh{(C5 H4 N)2 C=N-Me}])+ ([2]PF6 -PF6 -Cl)+ (see Supplementary Information). In addition, the spectrum also showed peak at m/z 421 due to ...
... acetonitrile showed fragmentation of molecular ion peaks at m/z = 470 and 435 due to ([(η5 -C5 Me5 )Rh {(C5 H4 N)2 C=N-Me}Cl])+ ([2]PF6 -PF6 )+ and ([(η5 C5 Me5 )Rh{(C5 H4 N)2 C=N-Me}])+ ([2]PF6 -PF6 -Cl)+ (see Supplementary Information). In addition, the spectrum also showed peak at m/z 421 due to ...
The Grob Fragmentation
... olefinic species if following the fragmentation route (similar to E1 or SN1) However the carbonium ion can further react via elimination, substitution, or ring-closure. The rate-determining step is the ionization to the carbonium ion. The tendency to ionize is greater when a tertiary and thus stable ...
... olefinic species if following the fragmentation route (similar to E1 or SN1) However the carbonium ion can further react via elimination, substitution, or ring-closure. The rate-determining step is the ionization to the carbonium ion. The tendency to ionize is greater when a tertiary and thus stable ...
Chapter 1 Structure and Bonding
... Hyperconjugation makes substituted alkenes more stable by stabilizing p-orbitals cis alkenes are less stable than trans alkenes because of steric crowding cis cycloalkenes are more stable than trans for the small rings ...
... Hyperconjugation makes substituted alkenes more stable by stabilizing p-orbitals cis alkenes are less stable than trans alkenes because of steric crowding cis cycloalkenes are more stable than trans for the small rings ...
Visible Light Photoredox Catalysis with Transition
... occur under extremely mild conditions, with most reactions proceeding at room temperature without the need for highly reactive radical initiators. The irradiation source is typically a commercial household light bulb, a significant advantage over the specialized equipment required for processes emplo ...
... occur under extremely mild conditions, with most reactions proceeding at room temperature without the need for highly reactive radical initiators. The irradiation source is typically a commercial household light bulb, a significant advantage over the specialized equipment required for processes emplo ...
lec-3- 211( Elim+ Re..
... Oxidation is the beginning of the deterioration process. Think of how a slice of apple turns brown when exposed to air. Oxidation leads to the formation of free radicals which are unstable molecules in the body that have one unpaired electron. They can cause oxidation and damage to the cells. This ...
... Oxidation is the beginning of the deterioration process. Think of how a slice of apple turns brown when exposed to air. Oxidation leads to the formation of free radicals which are unstable molecules in the body that have one unpaired electron. They can cause oxidation and damage to the cells. This ...
Beyond Conventional N-Heterocyclic Carbenes
... product distributionsand, thus, the site of metallationsis strongly anion-dependent. Large anions such as BF4- typically are only weak partners for hydrogen bonding and effect small changes in charge distribution. Consequently, such anions favor an oxidative addition pathway, leading to carbene C4 b ...
... product distributionsand, thus, the site of metallationsis strongly anion-dependent. Large anions such as BF4- typically are only weak partners for hydrogen bonding and effect small changes in charge distribution. Consequently, such anions favor an oxidative addition pathway, leading to carbene C4 b ...
Carbon–hydrogen bond activation of chloroalkanes by a rhodium
... also leads to a C–H bond activation product, 5. A single hydride resonance is observed just as for 4 at d )14.92 (d, J ¼ 25 Hz). However, examination of the Tp0 region of the spectrum shows evidence for two products. It was recognized that terminal activation of 3-chloropentane should yield two dias ...
... also leads to a C–H bond activation product, 5. A single hydride resonance is observed just as for 4 at d )14.92 (d, J ¼ 25 Hz). However, examination of the Tp0 region of the spectrum shows evidence for two products. It was recognized that terminal activation of 3-chloropentane should yield two dias ...
Improved Synthesis of Seven-Coordinate Molybdenum( I I) and
... syntheses7* of [M(CNR),I2+ and M(CNR),X2 (X = CF3C02-, Cl-) involve cleavage of the quadruply bonded M o ~ ~ + core by alkyl isocyanide. This paper describes high-yield preparations of the desired compounds through oxidation of M(CNR),(CO),, (1 Ix 5 3; R = alkyl, aryl; M = Mo, W) with mild oxidants ...
... syntheses7* of [M(CNR),I2+ and M(CNR),X2 (X = CF3C02-, Cl-) involve cleavage of the quadruply bonded M o ~ ~ + core by alkyl isocyanide. This paper describes high-yield preparations of the desired compounds through oxidation of M(CNR),(CO),, (1 Ix 5 3; R = alkyl, aryl; M = Mo, W) with mild oxidants ...
technical report 91 -32
... It is planned to dispose of nuclear waste in deep geologic formations in order to isolate it from the environment. The waste matrix itself, the steel containers, the backfill material and the surrounding rocks act 'as barriers to hinder migration of the radioactive elements to the biosphere. Intrusi ...
... It is planned to dispose of nuclear waste in deep geologic formations in order to isolate it from the environment. The waste matrix itself, the steel containers, the backfill material and the surrounding rocks act 'as barriers to hinder migration of the radioactive elements to the biosphere. Intrusi ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.