Reaction of Organometallic Reagents with Aldehydes and Ketones.
... • The difference between the two reactions is what then happens to the intermediate. • Aldehydes and ketones cannot undergo substitution because they do not have a good leaving group bonded to the newly formed sp3 hybridized carbon. ...
... • The difference between the two reactions is what then happens to the intermediate. • Aldehydes and ketones cannot undergo substitution because they do not have a good leaving group bonded to the newly formed sp3 hybridized carbon. ...
ch 1: organic chemistry
... alkanes- single C-C bonds, if all C’s have H’s attached, molecules are called saturated hydrocarbons alkenes- have one or more C=C double bonds alkynes- have one or more CΞC triple bonds alkenes & alkynes are called unsaturated hydrocarbons because they have fewer than the maximum possible n ...
... alkanes- single C-C bonds, if all C’s have H’s attached, molecules are called saturated hydrocarbons alkenes- have one or more C=C double bonds alkynes- have one or more CΞC triple bonds alkenes & alkynes are called unsaturated hydrocarbons because they have fewer than the maximum possible n ...
Topic 8 specification content - A
... I can explain that chlorine atoms are formed in the upper atmosphere when ultraviolet radiation causes C–Cl bonds in chlorofluorocarbons (CFCs) to break, and that these chlorine atoms catalyse the decomposition of ozone and contribute to the hole in the ...
... I can explain that chlorine atoms are formed in the upper atmosphere when ultraviolet radiation causes C–Cl bonds in chlorofluorocarbons (CFCs) to break, and that these chlorine atoms catalyse the decomposition of ozone and contribute to the hole in the ...
Development of New Synthetic Routes to Organoboronates by Catalytic Allylic Substitution and
... analogues, may result in scrambling of the E/Z geometry of the double bond and thereby loss of stereochemical information. Allylboronates (such as 1b-e) on the other hand are much less prone to this type of rearrangement and thereby posses higher configurational stability.47 ...
... analogues, may result in scrambling of the E/Z geometry of the double bond and thereby loss of stereochemical information. Allylboronates (such as 1b-e) on the other hand are much less prone to this type of rearrangement and thereby posses higher configurational stability.47 ...
- Wiley Online Library
... Especially interesting with regard to the regiochemistry is the deacetylation of tri-0-acetyl-D-glucal 5c. The lipaseP-catalyzed ester cleavage in weakly buffered solution without pH-control affords 4,6-di-O-acetyl-~-glucal6c in 90% yield. In the case of the tri-0-acetyl-D-galactal 5d, on the other ...
... Especially interesting with regard to the regiochemistry is the deacetylation of tri-0-acetyl-D-glucal 5c. The lipaseP-catalyzed ester cleavage in weakly buffered solution without pH-control affords 4,6-di-O-acetyl-~-glucal6c in 90% yield. In the case of the tri-0-acetyl-D-galactal 5d, on the other ...
CN>Chapter 22CT>Carbonyl Alpha
... as being in the α position Electrophilic substitution occurs at this position through either an enol or enolate ion ...
... as being in the α position Electrophilic substitution occurs at this position through either an enol or enolate ion ...
Nucleophilic Substitution Reactions
... electronegative than carbon, and so exserts a stronger pull on the shared electrons in the carbon-halogen bond. ■ As a result, the halogen gains a partial negative charge and the carbon gains a partial positive charge, and it is said to be electron deficient. ...
... electronegative than carbon, and so exserts a stronger pull on the shared electrons in the carbon-halogen bond. ■ As a result, the halogen gains a partial negative charge and the carbon gains a partial positive charge, and it is said to be electron deficient. ...
Faculteit der Natuurwetenschappen, Wiskunde en Informatica
... diastereomeric ratios are obtained; from 55:45 to 93:7. It should be noted that using bulkier Grignard reagents in the addition leads to higher diastereomeric ratios. This is most likely because bulkier substituents are more sterically hindered and causes the Grignard reagent to only attack the side ...
... diastereomeric ratios are obtained; from 55:45 to 93:7. It should be noted that using bulkier Grignard reagents in the addition leads to higher diastereomeric ratios. This is most likely because bulkier substituents are more sterically hindered and causes the Grignard reagent to only attack the side ...
Mild Conversion of Alcohols to Alkyl Halides Using Halide
... The mechanism is believed to involve protonation of the OH group followed by nucleophilic displacement by halide anions. The proton is presumably in association with either the halide or the conjugate bases of the Brønsted acids used. The rate is accelerated largely because of the ionic liquid media ...
... The mechanism is believed to involve protonation of the OH group followed by nucleophilic displacement by halide anions. The proton is presumably in association with either the halide or the conjugate bases of the Brønsted acids used. The rate is accelerated largely because of the ionic liquid media ...
lecture 11 catalysis_hydrogenation of alkenes
... Once CoH(CN)53− has been formed, a H• atom is transferred to the substrate in the second step, a reaction that does not require a vacant site at the metal, but does require the resulting organic radical to be moderately stable—hence the fact that the Iguchi catalyst will reduce only activated alken ...
... Once CoH(CN)53− has been formed, a H• atom is transferred to the substrate in the second step, a reaction that does not require a vacant site at the metal, but does require the resulting organic radical to be moderately stable—hence the fact that the Iguchi catalyst will reduce only activated alken ...
Reactions of Aromatic Compounds
... Arene (Ar-H) is the generic term for an aromatic hydrocarbon ...
... Arene (Ar-H) is the generic term for an aromatic hydrocarbon ...
Chapter 24. Amines
... has an odd-numbered molecular weight and a corresponding parent ion Alkylamines cleave at the C–C bond nearest the nitrogen to yield an alkyl radical and a nitrogencontaining cation ...
... has an odd-numbered molecular weight and a corresponding parent ion Alkylamines cleave at the C–C bond nearest the nitrogen to yield an alkyl radical and a nitrogencontaining cation ...
Chapter 24. Amines - Houston Community College System
... has an odd-numbered molecular weight and a corresponding parent ion Alkylamines cleave at the C–C bond nearest the nitrogen to yield an alkyl radical and a nitrogencontaining cation ...
... has an odd-numbered molecular weight and a corresponding parent ion Alkylamines cleave at the C–C bond nearest the nitrogen to yield an alkyl radical and a nitrogencontaining cation ...
lecture 5 ligand substitution
... trans influence and trans effect • In square planar complexes, some ligands direct substitution to a position trans to themselves. • When reaction is controlled by factors influencing the ground state energy of the complex – trans influence • Reaction is controlled by factors affecting the transiti ...
... trans influence and trans effect • In square planar complexes, some ligands direct substitution to a position trans to themselves. • When reaction is controlled by factors influencing the ground state energy of the complex – trans influence • Reaction is controlled by factors affecting the transiti ...
ppt
... • There will be two possible Wittig routes to an alkene. • Analyze the structure retrosynthetically, i.e., work the synthesis out backworks • Disconnect (break the bond of the target that can be formed by a known reaction) the doubly bonded carbons. One becomes the aldehyde or ketone, the other the ...
... • There will be two possible Wittig routes to an alkene. • Analyze the structure retrosynthetically, i.e., work the synthesis out backworks • Disconnect (break the bond of the target that can be formed by a known reaction) the doubly bonded carbons. One becomes the aldehyde or ketone, the other the ...
Synthesis of Platinum Group Metal Complexes for
... • Synthesis of of Pt(II) and Pd(II) complexes as Lewis acids, containing new C-, N- and P- based ligands, to be used in homogeneous catalysis such as epoxidation and isomerization of olefins, hydration of alkynes, Baeyer-Villiger oxidation of ketones and the catalytic formation of C-C and/or C-X (X ...
... • Synthesis of of Pt(II) and Pd(II) complexes as Lewis acids, containing new C-, N- and P- based ligands, to be used in homogeneous catalysis such as epoxidation and isomerization of olefins, hydration of alkynes, Baeyer-Villiger oxidation of ketones and the catalytic formation of C-C and/or C-X (X ...
CHEMISTRY 1000
... hydrogen to a carbonyl (using NaBH4 or LiAlH4 as the source of nucleophilic hydrogen). This was a chemoselective reaction – in other words, the reducing agent only reduced one functional group (the carbonyl) and left others alone (e.g. alkenes). If we want to reduce an alkene or alkyne, we need to u ...
... hydrogen to a carbonyl (using NaBH4 or LiAlH4 as the source of nucleophilic hydrogen). This was a chemoselective reaction – in other words, the reducing agent only reduced one functional group (the carbonyl) and left others alone (e.g. alkenes). If we want to reduce an alkene or alkyne, we need to u ...
Peer-reviewed Article PDF
... copper metal complex. The catalytic activity is dependent on the type of oxidant. The oxidation with t-BuOOH gave a higher yield (expressed as a percentage) than H2O2, because t-BuOOH is a more-efficient oxidant, due to weaker O-O bonds, with respect to H2O2. This trend is similar to that which was ...
... copper metal complex. The catalytic activity is dependent on the type of oxidant. The oxidation with t-BuOOH gave a higher yield (expressed as a percentage) than H2O2, because t-BuOOH is a more-efficient oxidant, due to weaker O-O bonds, with respect to H2O2. This trend is similar to that which was ...
127 - Chimica
... (CO),] (compound 4), identified spectroscopically (IR and 'H NMR), which was previously synthesized'" by photochemical hydrogenation of [Re2(CO)lo].The new method parallels that recently discovered8for the transformation of [Re4H6(CO)12]2into the unsaturated [Re4H5(CO),,]-. As in that case, the proc ...
... (CO),] (compound 4), identified spectroscopically (IR and 'H NMR), which was previously synthesized'" by photochemical hydrogenation of [Re2(CO)lo].The new method parallels that recently discovered8for the transformation of [Re4H6(CO)12]2into the unsaturated [Re4H5(CO),,]-. As in that case, the proc ...
top-curr-chem-1990-155-1
... Scheme 1 In 1977, an article from the authors' laboratories [9] reported an TiChmediated coupling reaction of l-alkoxy-l-siloxy-cyclopropane with aldehydes (Scheme 1), in which the intermediate formation of a titanium homoenolate (path b) was postulated instead of a then-more-likely Friedel-Crafts-l ...
... Scheme 1 In 1977, an article from the authors' laboratories [9] reported an TiChmediated coupling reaction of l-alkoxy-l-siloxy-cyclopropane with aldehydes (Scheme 1), in which the intermediate formation of a titanium homoenolate (path b) was postulated instead of a then-more-likely Friedel-Crafts-l ...
ENGLISH VERSION Exam Organic Chemistry 2
... Acetylcholine may otherwise be degraded in as well acidic as basic solution. Describe with detailed mechanisms how the hydrolysis of acetylcholine occurs in acid and base, respectively (3p each), and design a possible mechanism for how the enzyme acetylcholinesterase can catalyze the hydrolysis at n ...
... Acetylcholine may otherwise be degraded in as well acidic as basic solution. Describe with detailed mechanisms how the hydrolysis of acetylcholine occurs in acid and base, respectively (3p each), and design a possible mechanism for how the enzyme acetylcholinesterase can catalyze the hydrolysis at n ...
Chapter 17: Aldehydes and Ketones: Nucleophilic Addition to the
... • There will be two possible Wittig routes to an alkene. • Analyze the structure retrosynthetically, i.e., work the synthesis out backwards. • Disconnect (break the bond of the target that can be formed by a known reaction) the doubly bonded carbons. One becomes the aldehyde or ketone, the other th ...
... • There will be two possible Wittig routes to an alkene. • Analyze the structure retrosynthetically, i.e., work the synthesis out backwards. • Disconnect (break the bond of the target that can be formed by a known reaction) the doubly bonded carbons. One becomes the aldehyde or ketone, the other th ...
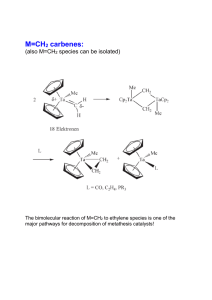
Steric protection of alkylidene is not needed:
... The bimolecular reaction of M=CH2 to ethylene species is one of the major pathways for decomposition of metathesis catalysts! ...
... The bimolecular reaction of M=CH2 to ethylene species is one of the major pathways for decomposition of metathesis catalysts! ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.