
Chapter 21: Carboxylic Acid Derivatives and Nucleophilic Acyl
... Relative reactivity – substituent effects ...
... Relative reactivity – substituent effects ...
Chapter 18 – Carbonyl Compounds II (Last Chapter we mostly talk
... nucleophile. This reaction was first discovered in the 1800’s by Hugo Schiff a German Chemist, and the product of this reaction is often referred to as a ...
... nucleophile. This reaction was first discovered in the 1800’s by Hugo Schiff a German Chemist, and the product of this reaction is often referred to as a ...
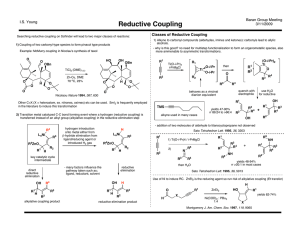
Reductive Couplings
... Development of NHC Ligands (allow for efficient intermoleuclar RC with triethylsilanes) ...
... Development of NHC Ligands (allow for efficient intermoleuclar RC with triethylsilanes) ...
KHSO4-SiO2-MeOH – An efficient selective solid
... Ester hydrolysis is an important fundamental reaction for deprotection of alcohols generally carried out either by chemical or biochemical methods. Chemical methods involve hydrolytic1 or nonhydrolytic2 methods using homogeneous or heterogeneous reagents. Because of the advantages of recycling and e ...
... Ester hydrolysis is an important fundamental reaction for deprotection of alcohols generally carried out either by chemical or biochemical methods. Chemical methods involve hydrolytic1 or nonhydrolytic2 methods using homogeneous or heterogeneous reagents. Because of the advantages of recycling and e ...
functional group
... Nomenclature assembles names like trees: start with a parent structure (e.g., an alkane), then add prefixes and a functional group (FG) suffix. ...
... Nomenclature assembles names like trees: start with a parent structure (e.g., an alkane), then add prefixes and a functional group (FG) suffix. ...
Grignard-syn-12-ques
... A Grignard reagent, RMgX, is a very strong base and a good nucleophile; water/moisture MUST be avoided in its reaction. ...
... A Grignard reagent, RMgX, is a very strong base and a good nucleophile; water/moisture MUST be avoided in its reaction. ...
ethers - WordPress.com
... • Simple ethers are named by identifying the two organic substituents and adding the word ether • If other functional groups are present, the ether part is considered an alkoxy substituent • R–O–R ~ tetrahedral bond angle (112° in dimethyl ether) • Oxygen is sp3-hybridized • Oxygen atom gives e ...
... • Simple ethers are named by identifying the two organic substituents and adding the word ether • If other functional groups are present, the ether part is considered an alkoxy substituent • R–O–R ~ tetrahedral bond angle (112° in dimethyl ether) • Oxygen is sp3-hybridized • Oxygen atom gives e ...
幻灯片 1
... • The E2 reaction (for elimination, bimolecular) occurs when an alkyl halide is treated with a strong base, such as hydroxide ion or alkoxide ion (RO-). It is the most commonly occurring pathway for elimination and can be formulated as shown below: ...
... • The E2 reaction (for elimination, bimolecular) occurs when an alkyl halide is treated with a strong base, such as hydroxide ion or alkoxide ion (RO-). It is the most commonly occurring pathway for elimination and can be formulated as shown below: ...
lecture 6 oxidative addition
... • In the SN2 pathway, adopted for polarized A‐B substrates such as alkyl halides, the metal electron pair of LnM directly attacks the A–B σ* orbital by an in‐line attack at the least electronegative atom (where σ* is largest) formally to give LnM2+ , A−, and B− fragments (ionic model). ...
... • In the SN2 pathway, adopted for polarized A‐B substrates such as alkyl halides, the metal electron pair of LnM directly attacks the A–B σ* orbital by an in‐line attack at the least electronegative atom (where σ* is largest) formally to give LnM2+ , A−, and B− fragments (ionic model). ...
The 16 and 18 Electron Rule in Organometallic Chemistry and
... 2 Types of Organometallic Reactions Reactions of ligands which do not directly involve the transition metal are excluded from this classification.Some examples of such reactions are methanolysis of Ni(PF& to give Ni(PF,) ,[PF,-n(OMe)n], Friedel-Crafts acylation of ferrocene, and nucleophilic attack ...
... 2 Types of Organometallic Reactions Reactions of ligands which do not directly involve the transition metal are excluded from this classification.Some examples of such reactions are methanolysis of Ni(PF& to give Ni(PF,) ,[PF,-n(OMe)n], Friedel-Crafts acylation of ferrocene, and nucleophilic attack ...
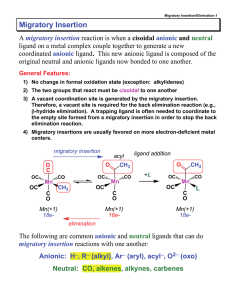
Migratory Insertion - vrg
... best Lewis acid - can coordinate to electron-rich CO ligands and drain off some e- density ...
... best Lewis acid - can coordinate to electron-rich CO ligands and drain off some e- density ...
Chapter 18 Ketones and Aldehydes 1) Which of the following
... 84) Provide the preferred reagent pair to synthesize 3-ethylpent-2-ene via a Wittig reaction. 85) Show how the following reaction may be accomplished by filling in the correct reagents. ...
... 84) Provide the preferred reagent pair to synthesize 3-ethylpent-2-ene via a Wittig reaction. 85) Show how the following reaction may be accomplished by filling in the correct reagents. ...
New L-Serine Derivative Ligands as Cocatalysts for Diels
... It is noteworthy that ligand 8 completely deactivated the metal catalyst (entries 1, 2, and 3), with the strong catalytic effect of AlCl3 and FeCl3 (entries 2 and 4, Table 1) being totally suppressed. This suggests that the cation coordinates to the basic amine group of 6 and the resulting complex i ...
... It is noteworthy that ligand 8 completely deactivated the metal catalyst (entries 1, 2, and 3), with the strong catalytic effect of AlCl3 and FeCl3 (entries 2 and 4, Table 1) being totally suppressed. This suggests that the cation coordinates to the basic amine group of 6 and the resulting complex i ...
Solid-Phase Organic Synthesis: Creation of Carbon
... aldehydes and ketones. Subsequently, treatments with a variety of phosphorane ylides or phosphonate anions provided alkenes. Stabilized phosphoranes quantitatively transformed aliphatic and aromatic aldehydes (no starting materials remaining) and gave E-olefins in good yields, purities, and stereose ...
... aldehydes and ketones. Subsequently, treatments with a variety of phosphorane ylides or phosphonate anions provided alkenes. Stabilized phosphoranes quantitatively transformed aliphatic and aromatic aldehydes (no starting materials remaining) and gave E-olefins in good yields, purities, and stereose ...
Reactions of Alkenes and Alkynes
... Ozone is generated by passing a stream of oxygen through a highvoltage electrical discharge Ozone adds rapidly to C=C bond at low temperature to give molozonide which spontaneously rearranges to ozonide Ozonide is treated with reducing agent to convert it to carbonyl ...
... Ozone is generated by passing a stream of oxygen through a highvoltage electrical discharge Ozone adds rapidly to C=C bond at low temperature to give molozonide which spontaneously rearranges to ozonide Ozonide is treated with reducing agent to convert it to carbonyl ...
[1] Ans1.Dows-proc - Sacred Heart School Moga,Best ICSE School
... pair of electrons. Since N is less electronegative than oxygen, therefore lone pair of electrons on the nitrogen atom is more easily available for bond formation. In other hand, nucleophillic attack occurs through N and hence silver nitrite predominantly gives nitro compounds. Q9. Explain, why the t ...
... pair of electrons. Since N is less electronegative than oxygen, therefore lone pair of electrons on the nitrogen atom is more easily available for bond formation. In other hand, nucleophillic attack occurs through N and hence silver nitrite predominantly gives nitro compounds. Q9. Explain, why the t ...
HIGHLIGHTS OF NUCLEOPHILIC SUBSTITUTION REACTIONS
... than it does on whether the reaction will follow an Sn1 or an Sn2 mechanism. The most important thing to remember in this regard is that good leaving groups are weak bases. a) All halogens, except for fluorine, are good leaving groups b) Groups that leave as resonance stabilized ions are also weak b ...
... than it does on whether the reaction will follow an Sn1 or an Sn2 mechanism. The most important thing to remember in this regard is that good leaving groups are weak bases. a) All halogens, except for fluorine, are good leaving groups b) Groups that leave as resonance stabilized ions are also weak b ...
Chapter 1 - chemistry
... sometimes called wood alcohol because it used to be prepared by the distillation of wood. Wood alcohol is extremely toxic. As little as 10 mL has been reported to cause permanent blindness and as little as 30 mL to can cause death. ...
... sometimes called wood alcohol because it used to be prepared by the distillation of wood. Wood alcohol is extremely toxic. As little as 10 mL has been reported to cause permanent blindness and as little as 30 mL to can cause death. ...
fulltext $(function(){PrimeFaces.cw("Tooltip","widget_formSmash_items_resultList_20_j_idt799_0_j_idt801",{id:"formSmash:items:resultList:20:j_idt799:0:j_idt801",widgetVar:"widget_formSmash_items_resultList_20_j_idt799_0_j_idt801",showEffect:"fade",hideEffect:"fade",target:"formSmash:items:resultList:20:j_idt799:0:fullText"});});
... calculations have provided a better understanding of the reaction pathways in some catalytic systems.17 Some catalysts have shown very high reactivities.10a, 12b, 18 However, this reaction typically requires high temperature (>100 oC) and there are only a few reported examples of the reaction at low ...
... calculations have provided a better understanding of the reaction pathways in some catalytic systems.17 Some catalysts have shown very high reactivities.10a, 12b, 18 However, this reaction typically requires high temperature (>100 oC) and there are only a few reported examples of the reaction at low ...
Practical and selective aerobic oxidation of alcohols to
... By performing reactions in a flow reactor, the product is obtained as a solution in toluene, which can be isolated simply by evaporating the solvent. However, more commonly, aldehydes and ketones are transformed into further compounds. As a workup procedure is not necessary, this can improve the ato ...
... By performing reactions in a flow reactor, the product is obtained as a solution in toluene, which can be isolated simply by evaporating the solvent. However, more commonly, aldehydes and ketones are transformed into further compounds. As a workup procedure is not necessary, this can improve the ato ...
Chapter 24. Amines
... • Can be more basic or less basic than aniline • Electron-donating substituents (such as CH3, NH2, OCH3) increase the basicity of the corresponding arylamine • Electron-withdrawing substituents (such as Cl, NO2, CN) decrease arylamine basicity ...
... • Can be more basic or less basic than aniline • Electron-donating substituents (such as CH3, NH2, OCH3) increase the basicity of the corresponding arylamine • Electron-withdrawing substituents (such as Cl, NO2, CN) decrease arylamine basicity ...
Chapter 24. Amines
... • Can be more basic or less basic than aniline • Electron-donating substituents (such as CH3, NH2, OCH3) increase the basicity of the corresponding arylamine • Electron-withdrawing substituents (such as Cl, NO2, CN) decrease arylamine basicity ...
... • Can be more basic or less basic than aniline • Electron-donating substituents (such as CH3, NH2, OCH3) increase the basicity of the corresponding arylamine • Electron-withdrawing substituents (such as Cl, NO2, CN) decrease arylamine basicity ...
AROMATIC COMPOUNDS
... alkylbenzene containing a straight-chain group, because the carbocation will rearrange: ...
... alkylbenzene containing a straight-chain group, because the carbocation will rearrange: ...
Reaction of Organometallic Reagents with Aldehydes and Ketones.
... • The difference between the two reactions is what then happens to the intermediate. • Aldehydes and ketones cannot undergo substitution because they do not have a good leaving group bonded to the newly formed sp3 hybridized carbon. ...
... • The difference between the two reactions is what then happens to the intermediate. • Aldehydes and ketones cannot undergo substitution because they do not have a good leaving group bonded to the newly formed sp3 hybridized carbon. ...
Stille reaction

The Stille reaction, or the Migita-Kosugi-Stille coupling, is a chemical reaction widely used in organic synthesis which involves the coupling of an organotin compound (also known as organostannanes) with a variety of organic electrophiles via palladium-catalyzed coupling reaction.The R1 group attached to the trialkyltin is normally sp2-hybridized, including alkenes, and aryl groups; however, conditions have been devised to incorporate both sp3-hybridized groups, such as allylic and benzylic substituents, and sp-hybridized alkynes. These organostannanes are also stable to both air and moisture, and many of these reagents are either commercially available or can be synthesized from literature precedent. However, these tin reagents tend to be highly toxic. X is typically a halide, such as Cl, Br, I, yet pseudohalides such as triflates and sulfonates and phosphates can also be used.The groundwork for the Stille reaction was laid by Colin Eaborn, Toshihiko Migita, and Masanori Kosugi in 1976 and 1977, who explored numerous palladium catalyzed couplings involving organotin reagents. John Stille and David Milstein developed a much milder and more broadly applicable procedure in 1978. Stille’s work on this area might have earned him a share of the 2010 Nobel Prize, which was awarded to Richard Heck, Ei-ichi Negishi, and Akira Suzuki for their work on the Heck, Negishi, and Suzuki coupling reactions. However, Stille died in the plane crash of United Airlines Flight 232 in 1989.Several reviews have been published on the Stille reaction.













![[1] Ans1.Dows-proc - Sacred Heart School Moga,Best ICSE School](http://s1.studyres.com/store/data/015878975_1-55791b331e05591620375059b6f74bac-300x300.png)







