Chemistry 1250 - Sp17 Solutions for Midterm 1
... throughout. These are often referred to as solutions. Gases dissolve in each other to form solutions and gases dissolve in liquids to form solutions. Heterogeneous mixtures have physical and chemical properties that are NOT uniform throughout the sample. Heterogeneous mixtures may contain elements a ...
... throughout. These are often referred to as solutions. Gases dissolve in each other to form solutions and gases dissolve in liquids to form solutions. Heterogeneous mixtures have physical and chemical properties that are NOT uniform throughout the sample. Heterogeneous mixtures may contain elements a ...
Quiz contsts questions chemistry
... 56 cm3 of oxygen combine with 112 cm3 of hydrogen to form water : When 56 cm3 of H2 is passed over heated capric oxide, the latter loses 0.04 g of its weight. All measurements are done under similar conditions of temperature and pressure (at. wt., H=1, O=16). Which of the following law is obeyed by ...
... 56 cm3 of oxygen combine with 112 cm3 of hydrogen to form water : When 56 cm3 of H2 is passed over heated capric oxide, the latter loses 0.04 g of its weight. All measurements are done under similar conditions of temperature and pressure (at. wt., H=1, O=16). Which of the following law is obeyed by ...
M.Sc. Part-I Chemistry - North Maharashtra University
... 2) A long sleeved, knee length laboratory coat is recommended. Long pants and closed toed shoes must be worn for individual safety. Loose clothing, open style shoes and sandals are prohibited. Long hair must be tied up. Each student will have to get his / her own necessary protection items. 3) Prior ...
... 2) A long sleeved, knee length laboratory coat is recommended. Long pants and closed toed shoes must be worn for individual safety. Loose clothing, open style shoes and sandals are prohibited. Long hair must be tied up. Each student will have to get his / her own necessary protection items. 3) Prior ...
Stoichiometry
... What the mass of H2 must be supplied to make 500. kg of NH3 in a system that has an 88.8% yield? Percent yield = 100% x actual yield/theore?cal yield Theore?cal yield = 100% x actual yield/percent yield = 100% x 500. kg of NH3 / 88.8% = 563 kg of NH3 563,000 gm NH3 x 1 mole NH3 x 3 ...
... What the mass of H2 must be supplied to make 500. kg of NH3 in a system that has an 88.8% yield? Percent yield = 100% x actual yield/theore?cal yield Theore?cal yield = 100% x actual yield/percent yield = 100% x 500. kg of NH3 / 88.8% = 563 kg of NH3 563,000 gm NH3 x 1 mole NH3 x 3 ...
BASIC CONCEPTS OF CHEMISTRY
... the nature of the process, resulting in a system of this state . The functions of the state are: 1) the internal energy of the system (U); 2) the enthalpy ( heat content ) of ( H) ; 3 ) the entropy (a measure of disorder) of the system (S); 4) the Gibbs’ free energy (G); 5) the Helmholtz’s free ener ...
... the nature of the process, resulting in a system of this state . The functions of the state are: 1) the internal energy of the system (U); 2) the enthalpy ( heat content ) of ( H) ; 3 ) the entropy (a measure of disorder) of the system (S); 4) the Gibbs’ free energy (G); 5) the Helmholtz’s free ener ...
Catalysts Containing Depleted Uranium Compounds
... are easy for recycling, whereas reaction products can be readily purified. It has been revealed that complexes of U4+ è U6+ are bright coloured, readily forming acilated ions, not forming complexes with ketones and aromatic esters. A possible mechanism for acylation reaction involving uranium salts ...
... are easy for recycling, whereas reaction products can be readily purified. It has been revealed that complexes of U4+ è U6+ are bright coloured, readily forming acilated ions, not forming complexes with ketones and aromatic esters. A possible mechanism for acylation reaction involving uranium salts ...
Final Exam
... ____ 15. If the energy of 1.00 mole of photons is 441 kJ, what is the wavelength of the light? a. 271 nm b. 451 nm c. 680 nm d. 732 nm e. 891 nm ____ 16. What type of orbital is designated n = 4, = 3, m = -3? a. 4s b. 4p c. 4d d. 4f e. none ____ 17. Which of the following orbitals might have m = ...
... ____ 15. If the energy of 1.00 mole of photons is 441 kJ, what is the wavelength of the light? a. 271 nm b. 451 nm c. 680 nm d. 732 nm e. 891 nm ____ 16. What type of orbital is designated n = 4, = 3, m = -3? a. 4s b. 4p c. 4d d. 4f e. none ____ 17. Which of the following orbitals might have m = ...
odd - WWW2
... formed by the most electropositive metals. These may contain the dicarbide(2 ) ion, C22 , or the true carbide ion C4 . Both types of ionic carbides react with water to produce the appropriate hydrocarbon. Covalent carbides are formed by nonmetals, specifically boron and silicon, more electronegative ...
... formed by the most electropositive metals. These may contain the dicarbide(2 ) ion, C22 , or the true carbide ion C4 . Both types of ionic carbides react with water to produce the appropriate hydrocarbon. Covalent carbides are formed by nonmetals, specifically boron and silicon, more electronegative ...
COMPOUNDS OF CARBON CONTAINING NITROGEN
... intermediates in drug synthesis. The quaternary ammonium salts derived from long chain aliphatic tertiary amines are widely used as detergents. Aromatic amines e.g. aniline and its derivatives, are used for the production of dyes, drugs and photographic developers. 1,4-Diaminobenzene is the main ing ...
... intermediates in drug synthesis. The quaternary ammonium salts derived from long chain aliphatic tertiary amines are widely used as detergents. Aromatic amines e.g. aniline and its derivatives, are used for the production of dyes, drugs and photographic developers. 1,4-Diaminobenzene is the main ing ...
CSEC Chemistry Revision Guide Answers.indd
... 5. The magnesium atoms are packed tightly together in rows to form a metal lattice and their valence electrons become delocalised. This forms positive magnesium cations and a sea of mobile electrons. The metal lattice is held together by the electrostatic forces of attraction between the delocalised ...
... 5. The magnesium atoms are packed tightly together in rows to form a metal lattice and their valence electrons become delocalised. This forms positive magnesium cations and a sea of mobile electrons. The metal lattice is held together by the electrostatic forces of attraction between the delocalised ...
RedOx notes:
... Which elements have specific rules? Which element(s) do(es) not have rules? Use rule 8 or 9 from above to calculate these. ...
... Which elements have specific rules? Which element(s) do(es) not have rules? Use rule 8 or 9 from above to calculate these. ...
8 theoretical problems 2 practical problems
... excess acidic phenylhydrazine. Compound C reacts with nitric acid to give an optically inactive compound D. The Kiliani-Fischer approach is used to establish the configurational relationship between D-glyceraldehyde and C. The intermediate aldotetrose which leads to C does not give a meso compound w ...
... excess acidic phenylhydrazine. Compound C reacts with nitric acid to give an optically inactive compound D. The Kiliani-Fischer approach is used to establish the configurational relationship between D-glyceraldehyde and C. The intermediate aldotetrose which leads to C does not give a meso compound w ...
Appendices and Glossary
... In this appendix, we will look at some of the quantitative relationships associated with the mass of elements and compounds. This subset of chemistry is called stoichiometry, a word derived from the Greek word “stoikheion”, meaning element. Before we begin, we need to say a few words about the appro ...
... In this appendix, we will look at some of the quantitative relationships associated with the mass of elements and compounds. This subset of chemistry is called stoichiometry, a word derived from the Greek word “stoikheion”, meaning element. Before we begin, we need to say a few words about the appro ...
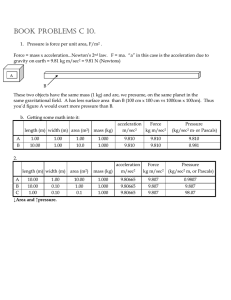
book problems c 10.
... To use this approach, it is necessary to have accurate values of atomic weights, often obtained by measuring the mass of atomic ions. For example, an ion trap, employing extremely uniform and stable magnetic and electric fields should allow such measurements to be made to better than 1 part in 1010. ...
... To use this approach, it is necessary to have accurate values of atomic weights, often obtained by measuring the mass of atomic ions. For example, an ion trap, employing extremely uniform and stable magnetic and electric fields should allow such measurements to be made to better than 1 part in 1010. ...
Concept Development Studies in Chemistry
... In contrast, the compound we call iron rust bears little resemblance to elemental iron: iron rust does not exhibit elemental iron's color, density, hardness, magnetism, etc. Since the properties of the elements are not maintained by the compound, then the compound must not be a simple mixture of the ...
... In contrast, the compound we call iron rust bears little resemblance to elemental iron: iron rust does not exhibit elemental iron's color, density, hardness, magnetism, etc. Since the properties of the elements are not maintained by the compound, then the compound must not be a simple mixture of the ...
Mass Relationships in Chemical Reactions
... Determining Empirical Formulas When the element % composition is known: 1. Assume 100g sample and change element percents to grams 2. Convert each to moles by dividing by molar mass of each atom ...
... Determining Empirical Formulas When the element % composition is known: 1. Assume 100g sample and change element percents to grams 2. Convert each to moles by dividing by molar mass of each atom ...
chapter 3 Questions
... This reaction generates a large amount of heat and many gaseous products. It is the sudden formation of these gases, together with their rapid expansion, that produces the explosion. Calculate the percent yield in this reaction if the amount of O2 generated from 2.00 102 g of nitroglycerin is foun ...
... This reaction generates a large amount of heat and many gaseous products. It is the sudden formation of these gases, together with their rapid expansion, that produces the explosion. Calculate the percent yield in this reaction if the amount of O2 generated from 2.00 102 g of nitroglycerin is foun ...
1ST CHAPTER Long-questions-basic-concept
... The term atom is derived from the Greek word “atoms” meaning indivisible. The smallest particle of an element which may or may not have independent existence is called an atom. For example ,the atoms of He,Ne and A r exist independently while the atoms of hydrogen ,nitrogen and oxygen do not have in ...
... The term atom is derived from the Greek word “atoms” meaning indivisible. The smallest particle of an element which may or may not have independent existence is called an atom. For example ,the atoms of He,Ne and A r exist independently while the atoms of hydrogen ,nitrogen and oxygen do not have in ...
4 ORGANIC CHEMISTRY: STRUCTURE AND NOMENCLATURE
... effect of your diet on the level of cholesterol in your blood, which brings up the questions, “What is cholesterol?” and “Why do so many people worry about it?” Answers to each of these questions fall within the realm of a field known as organic chemistry. For more than 200 years, chemists have divi ...
... effect of your diet on the level of cholesterol in your blood, which brings up the questions, “What is cholesterol?” and “Why do so many people worry about it?” Answers to each of these questions fall within the realm of a field known as organic chemistry. For more than 200 years, chemists have divi ...
RedOx notes:
... released heat is doing, keep in mind that iron melts at 1,530 oC. The amount of themite used in this reaction is suitable for a large lecture hall. The amount may be scaled down for smaller rooms while still providing a spectacular demonstration. There are numerous methods listed in the literature f ...
... released heat is doing, keep in mind that iron melts at 1,530 oC. The amount of themite used in this reaction is suitable for a large lecture hall. The amount may be scaled down for smaller rooms while still providing a spectacular demonstration. There are numerous methods listed in the literature f ...
2 CHEMICAL ARITHMATICS W MODULE - 1
... different elements in a molecule of a compound. There is another kind of formula, the empirical formul of a compound, which gives only relative number of atoms of different elements. These numbers are expressed as the simplest ratio. For example, empirical formula of glucose, which consists of carbo ...
... different elements in a molecule of a compound. There is another kind of formula, the empirical formul of a compound, which gives only relative number of atoms of different elements. These numbers are expressed as the simplest ratio. For example, empirical formula of glucose, which consists of carbo ...
Chapter 23 + Practice Problems - Bloomsburg Area School District
... have a large amount of saturated fatty acids, fats are solids at room temperature. Oils have more unsaturated fatty acids than fats, and are liquids. Like other animals, humans make fat, which is stored in adipose tissue until it is needed as an energy source. Fat has about twice as much energy per ...
... have a large amount of saturated fatty acids, fats are solids at room temperature. Oils have more unsaturated fatty acids than fats, and are liquids. Like other animals, humans make fat, which is stored in adipose tissue until it is needed as an energy source. Fat has about twice as much energy per ...
Oxidation-Reduction Reactions - An Introduction to Chemistry
... atoms is the general process for the formation of any binary ionic compound from its elements. For example, when sodium chloride is formed from the reaction of metallic sodium with gaseous chlorine, each sodium atom loses an electron, and each chlorine atom gains one. ...
... atoms is the general process for the formation of any binary ionic compound from its elements. For example, when sodium chloride is formed from the reaction of metallic sodium with gaseous chlorine, each sodium atom loses an electron, and each chlorine atom gains one. ...
Brilliant Preparatory Section, Sitamarhi
... viii. All the reactants and products should be written as molecules including the elements like hydrogen, oxygen, nitrogen, fluorine chlorine, bromine and iodine as H2, O2, N2, F2, Cl2, Br2 and I2. ...
... viii. All the reactants and products should be written as molecules including the elements like hydrogen, oxygen, nitrogen, fluorine chlorine, bromine and iodine as H2, O2, N2, F2, Cl2, Br2 and I2. ...