Chapter 1 Introduction to Forensic Chemistry
... Precipitation reactions begin with reagents dissolved in solution and produce a solid as a product. Combustion reactions involve the reaction of a substance with oxygen (often with applied heat); carbon-containing compounds then produce carbon dioxide and hydrogen-containing compounds then produce w ...
... Precipitation reactions begin with reagents dissolved in solution and produce a solid as a product. Combustion reactions involve the reaction of a substance with oxygen (often with applied heat); carbon-containing compounds then produce carbon dioxide and hydrogen-containing compounds then produce w ...
Answers
... convert g KClO3 mol KClO3 mol K g K. (The formula KClO3 shows the ratio of mol K to mol KClO3. This ratio is needed for the middle conversion factor). An alternative way to work this is to calculate the percent of K in KClO3 and figure out what number represents that percentage of 4.215 g. Ato ...
... convert g KClO3 mol KClO3 mol K g K. (The formula KClO3 shows the ratio of mol K to mol KClO3. This ratio is needed for the middle conversion factor). An alternative way to work this is to calculate the percent of K in KClO3 and figure out what number represents that percentage of 4.215 g. Ato ...
Nitrogen and Oxygen Family
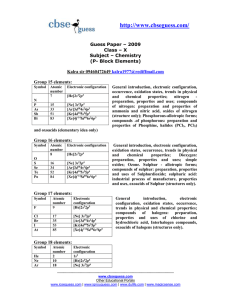
... Nitrogen differs from the rest of the members of this group due to its smaller size , high electronegativity, high ionisation enthalpy and non–availability of d orbitals. Nitrogen has unique ability to form p–p multiple bonds with itself and with other elements having small size and high electrone ...
... Nitrogen differs from the rest of the members of this group due to its smaller size , high electronegativity, high ionisation enthalpy and non–availability of d orbitals. Nitrogen has unique ability to form p–p multiple bonds with itself and with other elements having small size and high electrone ...
9278654 PS/Chemistry Ja03 - Dolgeville Central School
... question on your separate answer sheet. Write your answers to the Part B–2 and Part C questions in your answer booklet. All work should be written in pen, except for graphs and drawings, which should be done in pencil. You may use scrap paper to work out the answers to the questions, but be sure to ...
... question on your separate answer sheet. Write your answers to the Part B–2 and Part C questions in your answer booklet. All work should be written in pen, except for graphs and drawings, which should be done in pencil. You may use scrap paper to work out the answers to the questions, but be sure to ...
quantitative chemistry
... different components of a mixture often have different physical properties (such as melting point and density) and chemical properties (such as flammability and acidity). The properties of the mixture are similar to those of the components (e.g. a match burns in both air and pure oxygen), though the ...
... different components of a mixture often have different physical properties (such as melting point and density) and chemical properties (such as flammability and acidity). The properties of the mixture are similar to those of the components (e.g. a match burns in both air and pure oxygen), though the ...
Chapter 2 - hrsbstaff.ednet.ns.ca
... The isotopes, though, are not present in equal amounts. Carbon-12 comprises 98.9% of all carbon, while carbon-13 accounts for 1.1%. Carbon-14 is present in a very small amount— about 1 × 10−10 %. It makes sense that the average mass of all the isotopes of carbon is 12.01 u—very close to 12—since car ...
... The isotopes, though, are not present in equal amounts. Carbon-12 comprises 98.9% of all carbon, while carbon-13 accounts for 1.1%. Carbon-14 is present in a very small amount— about 1 × 10−10 %. It makes sense that the average mass of all the isotopes of carbon is 12.01 u—very close to 12—since car ...
STUDY MATERIAL 2016-17 CHEMISTRY CLASS XII
... of temperature and pressure. For example solid state, liquid, gases , plasma and BEC etc. Introduction: 1. The state of matter whose m.p. is above room temperature is found in solid state. Their constituent particles are held strongly. 2. Types of solid(Based on arrangement of particles) : 1 : Cryst ...
... of temperature and pressure. For example solid state, liquid, gases , plasma and BEC etc. Introduction: 1. The state of matter whose m.p. is above room temperature is found in solid state. Their constituent particles are held strongly. 2. Types of solid(Based on arrangement of particles) : 1 : Cryst ...
Specification – AS/A Level Chemistry A
... chemistry at Level 3 in the National Qualifications Framework (NQF). The AS specification has been written to provide progression from GCSE Science and GCSE Additional Science, or from GCSE Chemistry; achievement at a minimum of grade C in these qualifications should be seen as the normal requisite ...
... chemistry at Level 3 in the National Qualifications Framework (NQF). The AS specification has been written to provide progression from GCSE Science and GCSE Additional Science, or from GCSE Chemistry; achievement at a minimum of grade C in these qualifications should be seen as the normal requisite ...
Chapter 3 Stoichiometry: Calculations with Chemical
... • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Stoichiometry ...
... • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Stoichiometry ...
Science 9 Year End Review The following information includes all
... A _______________ _______________ (two words) is a cell that cannot be recharged, while _______________ _______________ (two words) is a cell that can be recharged. Topic 6: Generators and Motors A device that converts mechanical energy into electrical energy is known as a _______________. Wha ...
... A _______________ _______________ (two words) is a cell that cannot be recharged, while _______________ _______________ (two words) is a cell that can be recharged. Topic 6: Generators and Motors A device that converts mechanical energy into electrical energy is known as a _______________. Wha ...
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... • One mole of molecules or formula units contains Avogadro s number times the number of atoms or ions of each element in the compound Stoichiometry ...
... • One mole of molecules or formula units contains Avogadro s number times the number of atoms or ions of each element in the compound Stoichiometry ...
Calculations with Chemical Formulas and Equations
... • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Stoichiometry ...
... • One mole of molecules or formula units contains Avogadro’s number times the number of atoms or ions of each element in the compound Stoichiometry ...
GCSE Chemistry Specification Specification for exams from 2014 2014
... A hypothesis is a proposal intended to explain certain facts or observations. A prediction is a statement about the way something will happen in the future. Observations can lead to the start of an investigation, experiment or survey. Existing models can be used creatively to suggest explanations fo ...
... A hypothesis is a proposal intended to explain certain facts or observations. A prediction is a statement about the way something will happen in the future. Observations can lead to the start of an investigation, experiment or survey. Existing models can be used creatively to suggest explanations fo ...
Chemistry MCQS 12 class
... 2. so far 110 elements have been discovered. Out of these __________ elements are naturally ...
... 2. so far 110 elements have been discovered. Out of these __________ elements are naturally ...
OCR AS Level Chemistry B (Salters) H033
... Chemistry B (Salters) was first examined in 1992 as a new concept project examination. In contrast to the traditional ‘topic-based’ approach, Chemistry B (Salters) is ‘context-led’. Chemical concepts are introduced within a relevant context, the course being written as a series of teaching modules b ...
... Chemistry B (Salters) was first examined in 1992 as a new concept project examination. In contrast to the traditional ‘topic-based’ approach, Chemistry B (Salters) is ‘context-led’. Chemical concepts are introduced within a relevant context, the course being written as a series of teaching modules b ...
M - coercingmolecules
... Cu2S) by a multistep process. After an initial grinding, the first step is to “roast” the ore (heat it strongly with O2) to form Cu2O and SO2 2Cu2S(s) + 3O2(g) ...
... Cu2S) by a multistep process. After an initial grinding, the first step is to “roast” the ore (heat it strongly with O2) to form Cu2O and SO2 2Cu2S(s) + 3O2(g) ...
enjoy chemistry
... (i)The elements of group 18 are known as noble gases. The elements present in Group 18 have their valence shell orbitals completely filled and, therefore, react with a few elements only under certain conditions. Therefore, they are now known as noble gases. (ii)Noble gases are mostly chemically iner ...
... (i)The elements of group 18 are known as noble gases. The elements present in Group 18 have their valence shell orbitals completely filled and, therefore, react with a few elements only under certain conditions. Therefore, they are now known as noble gases. (ii)Noble gases are mostly chemically iner ...
48th CHEMISTRY OLYMPIAD CHEMISTRY
... 4. Chemical element X acts as a very strong oxidizing agent. Most of the reactions, in which participates, proceed with explosion or with the formation of a flame. At contact with many metals and nonmetals the reaction takes place already at ambient temperature. Even steam and glass wool (combustibl ...
... 4. Chemical element X acts as a very strong oxidizing agent. Most of the reactions, in which participates, proceed with explosion or with the formation of a flame. At contact with many metals and nonmetals the reaction takes place already at ambient temperature. Even steam and glass wool (combustibl ...
Study Guide for Content Mastery - Student Edition
... is worrisome because without ozone all organisms on Earth are subject to harm from too much radiation. In your textbook, read about chlorofluorocarbons. ...
... is worrisome because without ozone all organisms on Earth are subject to harm from too much radiation. In your textbook, read about chlorofluorocarbons. ...
Chapter 3 - Educator
... We read the + sign as “reacts with” and the arrow as “produces.” The chemical formulas to the left of the arrow represent the starting substances, called reactants. The chemical formulas to the right of the arrow represent substances produced in the reaction, called products. The numbers in front of ...
... We read the + sign as “reacts with” and the arrow as “produces.” The chemical formulas to the left of the arrow represent the starting substances, called reactants. The chemical formulas to the right of the arrow represent substances produced in the reaction, called products. The numbers in front of ...
Basic chemistry help is available here for high school or college
... classroom students. All hard copy materials distributed under this exception must have on every page distributed reference to http://www.chemtutor.com as source. Under the same exception granted to classroom teachers, full recognition of Chemtutor must be given when all or any part is included in an ...
... classroom students. All hard copy materials distributed under this exception must have on every page distributed reference to http://www.chemtutor.com as source. Under the same exception granted to classroom teachers, full recognition of Chemtutor must be given when all or any part is included in an ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten ...
... Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten ...
... Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten ...
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten ...
... Chemistry, The Central Science, 11th edition Theodore L. Brown, H. Eugene LeMay, Jr., and Bruce E. Bursten ...