Chemistry - Wheeling Jesuit University
... CHEMISTRY 43 Requirements for Bachelor of Arts Degree in Chemistry Upon completion of the BA degree program, students will be able to demonstrate the ability to: 1. Understand the principles and concepts fundamental to science, with an emphasis in chemistry. 2. Possess laboratory experiences approp ...
... CHEMISTRY 43 Requirements for Bachelor of Arts Degree in Chemistry Upon completion of the BA degree program, students will be able to demonstrate the ability to: 1. Understand the principles and concepts fundamental to science, with an emphasis in chemistry. 2. Possess laboratory experiences approp ...
Organic Chemistry Organic Chemistry
... In the early 18th century, the term “organic” had similar origins in chemistry. At that time, most chemists believed that compounds produced by living systems could not be made by any laboratory procedure. Scientists coined the chemical term “organic” to distinguish between compounds obtained from l ...
... In the early 18th century, the term “organic” had similar origins in chemistry. At that time, most chemists believed that compounds produced by living systems could not be made by any laboratory procedure. Scientists coined the chemical term “organic” to distinguish between compounds obtained from l ...
Peter Ertl - American Chemical Society
... substituents with possibly random distribution were not considered in the analysis. Examples of substituents with high drug-likeness are shown in Figure 8. These substituents (or their derivatives in the proper reactive state) may be used as reagents in drug optimization processes or as building blo ...
... substituents with possibly random distribution were not considered in the analysis. Examples of substituents with high drug-likeness are shown in Figure 8. These substituents (or their derivatives in the proper reactive state) may be used as reagents in drug optimization processes or as building blo ...
Mole Concept - Shailendra Kumar Chemistry
... (c) 200 ml of 3.0 M NaCl is added to 300 ml of 4.0 M NaCl. (d) 200 ml of 2.0 M BaCl2 is added to 400 ml of 3.0 M BaCl2 and 400 ml of water. (e) 300 ml of 3.0 M NaCl is added to 200 ml of 4.0 M BaCl2. (f) 400 ml of 2.0 M HCl is added to 150 ml of 4.0 M NaOH. (g) 100 ml of 2.0 M HCl and 200 ml of 1.5 ...
... (c) 200 ml of 3.0 M NaCl is added to 300 ml of 4.0 M NaCl. (d) 200 ml of 2.0 M BaCl2 is added to 400 ml of 3.0 M BaCl2 and 400 ml of water. (e) 300 ml of 3.0 M NaCl is added to 200 ml of 4.0 M BaCl2. (f) 400 ml of 2.0 M HCl is added to 150 ml of 4.0 M NaOH. (g) 100 ml of 2.0 M HCl and 200 ml of 1.5 ...
Introductory Chemistry, 2nd Edition Nivaldo Tro
... number of molecules as well as the number of moles of each substance ...
... number of molecules as well as the number of moles of each substance ...
5. Coenzyme HAD+ is derived
... training. Chemistry is a basic science and a powerful tool for studying and learning processes in living systems. Therefore, medical students must thoroughly understand the basic ideas, laws and methods of this science. Program expected to consider the foundations of the most important topics of the ...
... training. Chemistry is a basic science and a powerful tool for studying and learning processes in living systems. Therefore, medical students must thoroughly understand the basic ideas, laws and methods of this science. Program expected to consider the foundations of the most important topics of the ...
Unit 3 Answer Key
... 3. You would have two times the Avogadro constant of hydrogen atoms. Rounded off, the number would be 2(6.02 × 1023) = 1.20 × 1024 hydrogen atoms. 4. You would not be able to see one person, but a mole of people is so many that they would be visible, as a group, from space. In fact, a mole of pe ...
... 3. You would have two times the Avogadro constant of hydrogen atoms. Rounded off, the number would be 2(6.02 × 1023) = 1.20 × 1024 hydrogen atoms. 4. You would not be able to see one person, but a mole of people is so many that they would be visible, as a group, from space. In fact, a mole of pe ...
Question Bank - Edudel.nic.in
... In a fcc arrangement of A and B atoms. A are present at the corners of the unit cell and B are present at the face centres. If one atom of A is missing from its position at the corners, what is the formula of the compound? [Ans. : A7B24] ...
... In a fcc arrangement of A and B atoms. A are present at the corners of the unit cell and B are present at the face centres. If one atom of A is missing from its position at the corners, what is the formula of the compound? [Ans. : A7B24] ...
Chemistry - BYU
... This course involves a study of structure, physical and chemical behavior, and bond theory as applied to inorganic chemistry. Using concepts, models, and experimental data, a variety of inorganic systems will be analyzed in a systematic and detailed fashion. Underlying trends in both the elements an ...
... This course involves a study of structure, physical and chemical behavior, and bond theory as applied to inorganic chemistry. Using concepts, models, and experimental data, a variety of inorganic systems will be analyzed in a systematic and detailed fashion. Underlying trends in both the elements an ...
Chapter 3 - Educator
... French nobleman and scientist Antoine Lavoisier (Figure 3.1 «) discovered this important chemical law in the late 1700s. In a chemistry text published in 1789, Lavoisier stated the law in this eloquent way: “We may lay it down as an incontestable axiom that, in all the operations of art and nature, ...
... French nobleman and scientist Antoine Lavoisier (Figure 3.1 «) discovered this important chemical law in the late 1700s. In a chemistry text published in 1789, Lavoisier stated the law in this eloquent way: “We may lay it down as an incontestable axiom that, in all the operations of art and nature, ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... • Compounds containing C, H and O are routinely analyzed through combustion in a chamber like this. – C is determined from the mass of CO2 produced. – H is determined from the mass of H2O produced. – O is determined by difference after the C and H have been ...
... • Compounds containing C, H and O are routinely analyzed through combustion in a chamber like this. – C is determined from the mass of CO2 produced. – H is determined from the mass of H2O produced. – O is determined by difference after the C and H have been ...
CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... TOPIC 1. REACTION RATES - COLLISION THEORY ...
... TOPIC 1. REACTION RATES - COLLISION THEORY ...
quantitative_chemistry
... This is the same huge number as the number of amu’s in 1 g of a substance! By choosing to weigh out a mass in grams that has the same numerical value as the formula mass in amu’s (180.2 grams and 180.2 amu, in the case of aspirin) we are able to ensure that the sample contains just as many molecules ...
... This is the same huge number as the number of amu’s in 1 g of a substance! By choosing to weigh out a mass in grams that has the same numerical value as the formula mass in amu’s (180.2 grams and 180.2 amu, in the case of aspirin) we are able to ensure that the sample contains just as many molecules ...
Chapter
... with Chemical Formula • compounds are generally represented with a chemical formula • the amount of information about the structure of the compound varies with the type of formula – all formula and models convey a limited amount of information – none are perfect representations ...
... with Chemical Formula • compounds are generally represented with a chemical formula • the amount of information about the structure of the compound varies with the type of formula – all formula and models convey a limited amount of information – none are perfect representations ...
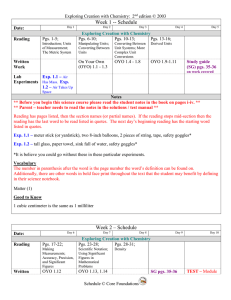
Week 1 -- Schedule
... Exp. 1.1 – meter stick (or yardstick), two 8-inch balloons, 2 pieces of string, tape, safety goggles* Exp. 1.2 – tall glass, paper towel, sink full of water, safety goggles* *It is believe you could go without these in these particular experiments. Vocabulary The number in parenthesis after the word ...
... Exp. 1.1 – meter stick (or yardstick), two 8-inch balloons, 2 pieces of string, tape, safety goggles* Exp. 1.2 – tall glass, paper towel, sink full of water, safety goggles* *It is believe you could go without these in these particular experiments. Vocabulary The number in parenthesis after the word ...
General chemistry laboratory activities, Lorentz
... accompanied by their necks, at the ends of which are ground glass joints to quickly and tightly connect to the rest of the apparatus (such as a reflux condenser or dropping funnel). The reaction flask is usually made of thick glass and they can tolerate large pressure differences, with the result th ...
... accompanied by their necks, at the ends of which are ground glass joints to quickly and tightly connect to the rest of the apparatus (such as a reflux condenser or dropping funnel). The reaction flask is usually made of thick glass and they can tolerate large pressure differences, with the result th ...
Calculations and the Chemical Equation
... for defining a "collection" of atoms is the mole, Avogadro's number of particles. Calculations based on the chemical equation relate the number of atoms, moles and their corresponding mass. Conversion factors are used to relate the information provided in the problem to the information requested by ...
... for defining a "collection" of atoms is the mole, Avogadro's number of particles. Calculations based on the chemical equation relate the number of atoms, moles and their corresponding mass. Conversion factors are used to relate the information provided in the problem to the information requested by ...
Chapter 8
... Calculations Using Mass of Reactants and Products In the laboratory we work with masses of reactants and products. However, as we have seen, chemical equations give relationships among moles of reactants and products. We therefore need a method for going back and forth between moles of substance an ...
... Calculations Using Mass of Reactants and Products In the laboratory we work with masses of reactants and products. However, as we have seen, chemical equations give relationships among moles of reactants and products. We therefore need a method for going back and forth between moles of substance an ...
Mass Relationships in Chemical Reactions
... When the element % composition is known: 1. Assume 100g sample and change element percents to grams 2. Convert each to moles by dividing by molar mass of each atom ...
... When the element % composition is known: 1. Assume 100g sample and change element percents to grams 2. Convert each to moles by dividing by molar mass of each atom ...
chapter 3 Questions
... This reaction generates a large amount of heat and many gaseous products. It is the sudden formation of these gases, together with their rapid expansion, that produces the explosion. Calculate the percent yield in this reaction if the amount of O2 generated from 2.00 102 g of nitroglycerin is foun ...
... This reaction generates a large amount of heat and many gaseous products. It is the sudden formation of these gases, together with their rapid expansion, that produces the explosion. Calculate the percent yield in this reaction if the amount of O2 generated from 2.00 102 g of nitroglycerin is foun ...
Click Chemistry in Peptide-Based Drug Design
... Bock et al. reported the synthesis of triazole-containing analogues (5–7, Figure 2) of the naturally occurring tyrosinase inhibitor cyclo-[Pro-Val-Pro-Tyr] (4, Figure 2) and showed that the analogues retained enzyme inhibitory activity, demonstrating the effectiveness of a 1,4-connected 1,2,3-triazo ...
... Bock et al. reported the synthesis of triazole-containing analogues (5–7, Figure 2) of the naturally occurring tyrosinase inhibitor cyclo-[Pro-Val-Pro-Tyr] (4, Figure 2) and showed that the analogues retained enzyme inhibitory activity, demonstrating the effectiveness of a 1,4-connected 1,2,3-triazo ...
Chemical Equilibrium
... concentrations of the reactants and products. • In an equilibrium expression, do not include pure solids (s) or liquids (l) • This is because their concentration is their density, which does not change at any given ...
... concentrations of the reactants and products. • In an equilibrium expression, do not include pure solids (s) or liquids (l) • This is because their concentration is their density, which does not change at any given ...
Communicating Chemistry Landscape Study
... Science Foundation (NSF) to develop a framework for effective chemistry communication, outreach, and education in informal settings, with the ultimate goal of increasing the effectiveness of such efforts in engaging the public with chemistry. BCST and BOSE are assembling a committee of experts to ex ...
... Science Foundation (NSF) to develop a framework for effective chemistry communication, outreach, and education in informal settings, with the ultimate goal of increasing the effectiveness of such efforts in engaging the public with chemistry. BCST and BOSE are assembling a committee of experts to ex ...
Spring 2016 CHEM Handbook - Chemistry | Virginia Tech
... Chemistry is central to the sciences, the understanding of the physical world, and the study of biological systems. Chemistry is the science of transformations and energetics of materials at the molecular level. Chemistry has applications from the nanoscale to the macroscopic. Chemists use their tra ...
... Chemistry is central to the sciences, the understanding of the physical world, and the study of biological systems. Chemistry is the science of transformations and energetics of materials at the molecular level. Chemistry has applications from the nanoscale to the macroscopic. Chemists use their tra ...
History of chemistry

The history of chemistry represents a time span from ancient history to the present. By 1000 BC, civilizations used technologies that would eventually form the basis to the various branches of chemistry. Examples include extracting metals from ores, making pottery and glazes, fermenting beer and wine, extracting chemicals from plants for medicine and perfume, rendering fat into soap, making glass, and making alloys like bronze.The protoscience of chemistry, alchemy, was unsuccessful in explaining the nature of matter and its transformations. However, by performing experiments and recording the results, alchemists set the stage for modern chemistry. The distinction began to emerge when a clear differentiation was made between chemistry and alchemy by Robert Boyle in his work The Sceptical Chymist (1661). While both alchemy and chemistry are concerned with matter and its transformations, chemists are seen as applying scientific method to their work.Chemistry is considered to have become an established science with the work of Antoine Lavoisier, who developed a law of conservation of mass that demanded careful measurement and quantitative observations of chemical phenomena. The history of chemistry is intertwined with the history of thermodynamics, especially through the work of Willard Gibbs.