Document
... theoretical yield: the maximum amount of product that can be formed – calculated by stoichiometry (using LR only) 1 mol Al 3 mol Cu 0.030 g Al x x = 0.0017 mol Cu 26.98 g Al 2 mol Al • This is different from the actual yield, the amount one actually produces and measures (or experimental) ...
... theoretical yield: the maximum amount of product that can be formed – calculated by stoichiometry (using LR only) 1 mol Al 3 mol Cu 0.030 g Al x x = 0.0017 mol Cu 26.98 g Al 2 mol Al • This is different from the actual yield, the amount one actually produces and measures (or experimental) ...
PART 6-ICHO-26-30
... Calculate the concentration of nitrogen in the sample, in percent by mass. ...
... Calculate the concentration of nitrogen in the sample, in percent by mass. ...
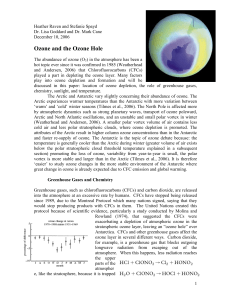
Ozone Writeup - International Research Institute for Climate and
... vortex is more stable and larger than in the Arctic (Tilmes et al., 2006). It is therefore ‘easier’ to study ozone changes in the more stable environment of the Antarctic where great change in ozone is already expected due to CFC emission and global warming. Greenhouse Gases and Chemistry Greenhouse ...
... vortex is more stable and larger than in the Arctic (Tilmes et al., 2006). It is therefore ‘easier’ to study ozone changes in the more stable environment of the Antarctic where great change in ozone is already expected due to CFC emission and global warming. Greenhouse Gases and Chemistry Greenhouse ...
Teaching with CAChe - Photochemical Dynamics Group
... something new, or at least gained unexpected insight into the system. The fun began when we found unpredicted results; the results often pointed out our own misconceptions about the underlying chemistry. Presently, we site license CAChe software. The site license has made CAChe accessible to faculty ...
... something new, or at least gained unexpected insight into the system. The fun began when we found unpredicted results; the results often pointed out our own misconceptions about the underlying chemistry. Presently, we site license CAChe software. The site license has made CAChe accessible to faculty ...
Solutions Manual
... atoms per molecule are used for food wrap films; 60 000 atoms per molecule make milk containers; 80 000 atoms per molecule make bleach containers and 800 000 atoms per molecule can be used in artificial ice rinks. As chain length increases, density, hardness and melting point increase. Branching is ...
... atoms per molecule are used for food wrap films; 60 000 atoms per molecule make milk containers; 80 000 atoms per molecule make bleach containers and 800 000 atoms per molecule can be used in artificial ice rinks. As chain length increases, density, hardness and melting point increase. Branching is ...
Science 9 Year End Review The following information includes all
... _________________ will speed up a reaction. Decreasing these will slow it down. Rusting is one type of _________________. Define CORROSION and provide an example (including an equation) Coating a corrosive metal (a metal that can corrode) with zinc is referred to as _________________. Define ...
... _________________ will speed up a reaction. Decreasing these will slow it down. Rusting is one type of _________________. Define CORROSION and provide an example (including an equation) Coating a corrosive metal (a metal that can corrode) with zinc is referred to as _________________. Define ...
Chem Agenda+ETDsHWK to End of Year 102714 Update
... 3. Atoms of a given element are different from those of another element (different elements have different atoms) 4. atoms of one element can combine with other elements to form compounds. 5. Atoms are indivisible in chemical processes. In Chem Rxn atoms simply rearrange but matter is conserved. ...
... 3. Atoms of a given element are different from those of another element (different elements have different atoms) 4. atoms of one element can combine with other elements to form compounds. 5. Atoms are indivisible in chemical processes. In Chem Rxn atoms simply rearrange but matter is conserved. ...
Chemistry and Biochemistry
... Graduate students must maintain a GPA of 3.0/4.0. Only courses applicable to the graduate program, excluding those for making up deficiencies or satisfying proficiencies, are counted in the GPA. If the cumulative GPA drops below 3.0 for one semester, the student will be placed on academic probation. ...
... Graduate students must maintain a GPA of 3.0/4.0. Only courses applicable to the graduate program, excluding those for making up deficiencies or satisfying proficiencies, are counted in the GPA. If the cumulative GPA drops below 3.0 for one semester, the student will be placed on academic probation. ...
EVS - RSC - Developments in Microwave Chemistry
... such as ashing, digestion, extraction, fat analysis and protein hydrolysis. As microwave chemical synthesis has advanced, its applications have been extended to include the synthesis of fine chemicals, organometallic, coordination, intercalation compounds, and nanoparticles. Microwave technology als ...
... such as ashing, digestion, extraction, fat analysis and protein hydrolysis. As microwave chemical synthesis has advanced, its applications have been extended to include the synthesis of fine chemicals, organometallic, coordination, intercalation compounds, and nanoparticles. Microwave technology als ...
Document
... compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the temperature even insoluble compounds dissolve, just not enough to be ...
... compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the temperature even insoluble compounds dissolve, just not enough to be ...
TRO Chapter 4
... compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the temperature even insoluble compounds dissolve, just not enough to be ...
... compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the temperature even insoluble compounds dissolve, just not enough to be ...
Chapter 4 Chemical Quantities and Aqueous Reactions
... compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the temperature even insoluble compounds dissolve, just not enough to be ...
... compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the temperature even insoluble compounds dissolve, just not enough to be ...
Calculations with Chemical Formulas and Equations
... real-world scale The number of moles correspond to the number of molecules. 1 mole of any substance has the same number of molecules. Stoichiometry ...
... real-world scale The number of moles correspond to the number of molecules. 1 mole of any substance has the same number of molecules. Stoichiometry ...
Chapter 3 Stoichiometry: Calculations with Chemical
... real-world scale The number of moles correspond to the number of molecules. 1 mole of any substance has the same number of molecules. Stoichiometry ...
... real-world scale The number of moles correspond to the number of molecules. 1 mole of any substance has the same number of molecules. Stoichiometry ...
Chapter 3 Stoichiometry: Calculations with Chemical Formulas and
... real-world scale The number of moles correspond to the number of molecules. 1 mole of any substance has the same number of molecules. Stoichiometry ...
... real-world scale The number of moles correspond to the number of molecules. 1 mole of any substance has the same number of molecules. Stoichiometry ...
Photo-oxidation of pinonaldehyde at low NOx
... mixing) in the chamber and the onset of photo-oxidation. Extrapolation of this signal after the photo-oxidation process starts defines the seed mass concentration as a function of time. At t = 0 (onset of photo-oxidation) and later, the difference between the total aerosol mass concentration in the ...
... mixing) in the chamber and the onset of photo-oxidation. Extrapolation of this signal after the photo-oxidation process starts defines the seed mass concentration as a function of time. At t = 0 (onset of photo-oxidation) and later, the difference between the total aerosol mass concentration in the ...
History of chemistry

The history of chemistry represents a time span from ancient history to the present. By 1000 BC, civilizations used technologies that would eventually form the basis to the various branches of chemistry. Examples include extracting metals from ores, making pottery and glazes, fermenting beer and wine, extracting chemicals from plants for medicine and perfume, rendering fat into soap, making glass, and making alloys like bronze.The protoscience of chemistry, alchemy, was unsuccessful in explaining the nature of matter and its transformations. However, by performing experiments and recording the results, alchemists set the stage for modern chemistry. The distinction began to emerge when a clear differentiation was made between chemistry and alchemy by Robert Boyle in his work The Sceptical Chymist (1661). While both alchemy and chemistry are concerned with matter and its transformations, chemists are seen as applying scientific method to their work.Chemistry is considered to have become an established science with the work of Antoine Lavoisier, who developed a law of conservation of mass that demanded careful measurement and quantitative observations of chemical phenomena. The history of chemistry is intertwined with the history of thermodynamics, especially through the work of Willard Gibbs.