Full Text - Verlag der Zeitschrift für Naturforschung
... PH3 , H2 O, H2 S) and BH3 is formed through the participation of lone pairs of N, P, O and S atoms in X groups into the unfilled boron orbital. Therefore, the charge density around donor atom in X and consequently its χ decreases. The χ values of N, O and S atoms in the complexes are much smaller th ...
... PH3 , H2 O, H2 S) and BH3 is formed through the participation of lone pairs of N, P, O and S atoms in X groups into the unfilled boron orbital. Therefore, the charge density around donor atom in X and consequently its χ decreases. The χ values of N, O and S atoms in the complexes are much smaller th ...
Organic Chemistry II: Here We Go Again!
... is a measure of the ability of a compound to rotate light. The specific rotation comes from the observed rotation (α) divided by the product of the concentration of the solution and the length of the container. Other than optical activity, the physical properties of enantiomers are the same. A racem ...
... is a measure of the ability of a compound to rotate light. The specific rotation comes from the observed rotation (α) divided by the product of the concentration of the solution and the length of the container. Other than optical activity, the physical properties of enantiomers are the same. A racem ...
Lecture - Ch 12
... Common Functional Groups • Halides – Elements comprising two common isotopes possess a distinctive appearance as a mass spectra ...
... Common Functional Groups • Halides – Elements comprising two common isotopes possess a distinctive appearance as a mass spectra ...
CHEM 210 Nomenclature Lecture
... 3. Name and number the substituents. • Name the substituents as alkyl groups. • Every carbon belongs to either the longest chain or a substituent, not both. • Each substituent needs its own number • If two or more identical substituents are bonded to the longest chain, use prefixes to indicate how m ...
... 3. Name and number the substituents. • Name the substituents as alkyl groups. • Every carbon belongs to either the longest chain or a substituent, not both. • Each substituent needs its own number • If two or more identical substituents are bonded to the longest chain, use prefixes to indicate how m ...
CH 2
... 4. The location and name of each substituent are followed by the root alkane name. The substituents are listed in alphabetical order (irrespective of any prefix), and the prefixes di-, tri-, etc. are used to indicate multiple identical substituents. H3C1 ...
... 4. The location and name of each substituent are followed by the root alkane name. The substituents are listed in alphabetical order (irrespective of any prefix), and the prefixes di-, tri-, etc. are used to indicate multiple identical substituents. H3C1 ...
poly- and heterofunctional compounds
... Cyclic hemiacetals. If an aldehyde or a ketone contains a hydroxyl group at an appropriate distance (at the C-4 or C-5 atom for aldehydes), the carbonyl and hydroxyl groups may react with each other. The molecule takes a conformation in which both functional groups are favourably located. The result ...
... Cyclic hemiacetals. If an aldehyde or a ketone contains a hydroxyl group at an appropriate distance (at the C-4 or C-5 atom for aldehydes), the carbonyl and hydroxyl groups may react with each other. The molecule takes a conformation in which both functional groups are favourably located. The result ...
HIGHLIGHTS OF NUCLEOPHILIC SUBSTITUTION REACTIONS
... hindered primary substrate can be forced into an Sn1 mechanism if sufficient energy and time are allowed, for example boiling the substrate in a nucleophilic solvent such as ethanol. The nucleophile cannot do a backside attack, and the substrate cannot form stable cations. In this case the substrate ...
... hindered primary substrate can be forced into an Sn1 mechanism if sufficient energy and time are allowed, for example boiling the substrate in a nucleophilic solvent such as ethanol. The nucleophile cannot do a backside attack, and the substrate cannot form stable cations. In this case the substrate ...
title of abstract
... A possible interaction between Am3+ and the iminodiacetic moiety is schematically given in Fig. 1 and could be described as a cation exchange reaction between the protons and the americium cations. Because proton concentration is a key factor governing both, the proton dissociation of the iminodiace ...
... A possible interaction between Am3+ and the iminodiacetic moiety is schematically given in Fig. 1 and could be described as a cation exchange reaction between the protons and the americium cations. Because proton concentration is a key factor governing both, the proton dissociation of the iminodiace ...
United States Patent Dolphin et al.
... desired effect even more pronounced. There has only been one general method known to convert meso-tetraphenyl 45 porphyrins into the corresponding chlorins, namely the diimide reduction introduced by Whitlock et aI., "Diimide Reduction of Porphyrins", J. Am. Chern. Soc., 91, 7485-89 (1969). However, ...
... desired effect even more pronounced. There has only been one general method known to convert meso-tetraphenyl 45 porphyrins into the corresponding chlorins, namely the diimide reduction introduced by Whitlock et aI., "Diimide Reduction of Porphyrins", J. Am. Chern. Soc., 91, 7485-89 (1969). However, ...
Alcohols
... In phenols the -OH group is directly attached to a carbon that is part of an aromatic ring. Alcohols and phenols are similar in some ways, but there are enough differences so that they are considered different functional groups. One major difference is that phenols are typically about a million ...
... In phenols the -OH group is directly attached to a carbon that is part of an aromatic ring. Alcohols and phenols are similar in some ways, but there are enough differences so that they are considered different functional groups. One major difference is that phenols are typically about a million ...
Document
... Organic compounds that have the same functional group often have similar physical properties, such as boiling points, melting points, and solubilities. Physical properties are largely determined by intermolecular forces, the forces of attraction and repulsion between particles. Three types of interm ...
... Organic compounds that have the same functional group often have similar physical properties, such as boiling points, melting points, and solubilities. Physical properties are largely determined by intermolecular forces, the forces of attraction and repulsion between particles. Three types of interm ...
Electophilic Aromatic Substituion
... under conditions that reduce alkene double bonds Can selectively reduce an alkene double bond in the presence of an aromatic ring Reduction of an aromatic ring requires more powerful reducing conditions (high pressure or rhodium ...
... under conditions that reduce alkene double bonds Can selectively reduce an alkene double bond in the presence of an aromatic ring Reduction of an aromatic ring requires more powerful reducing conditions (high pressure or rhodium ...
Chapter 16 - Chemistry of Benzene
... under conditions that reduce alkene double bonds Can selectively reduce an alkene double bond in the presence of an aromatic ring Reduction of an aromatic ring requires more powerful reducing conditions (high pressure or rhodium ...
... under conditions that reduce alkene double bonds Can selectively reduce an alkene double bond in the presence of an aromatic ring Reduction of an aromatic ring requires more powerful reducing conditions (high pressure or rhodium ...
2 The Nature of Matter
... elements of matter: earth, air, fire, and water. These elements were not equivalent to matter in modern chemistry. For the alchemists, earth, air, fire, and water represented four fundamental properties of matter. Alchemists believed that these properties existed independent of matter and could be a ...
... elements of matter: earth, air, fire, and water. These elements were not equivalent to matter in modern chemistry. For the alchemists, earth, air, fire, and water represented four fundamental properties of matter. Alchemists believed that these properties existed independent of matter and could be a ...
Wed March 3 lecture
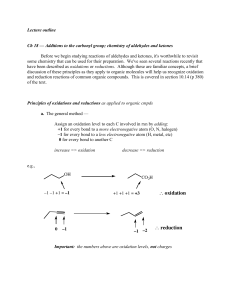
... Ch 18 — Additions to the carbonyl group; chemistry of aldehydes and ketones Before we begin studying reactions of aldehydes and ketones, it's worthwhile to revisit some chemistry that can be used for their preparation. We've seen several reactions recently that have been described as oxidations or r ...
... Ch 18 — Additions to the carbonyl group; chemistry of aldehydes and ketones Before we begin studying reactions of aldehydes and ketones, it's worthwhile to revisit some chemistry that can be used for their preparation. We've seen several reactions recently that have been described as oxidations or r ...
Chapter 18 Organic Chemistry - American Public University System
... The rules to systematically name alkanes 4. Write the name of the compound in the format: (subst. number)–(subst. name) (base name) If there are two or more substituents, give each one a number and list them alphabetically with hyphens between words and numbers. 5. If a compound has two or more ide ...
... The rules to systematically name alkanes 4. Write the name of the compound in the format: (subst. number)–(subst. name) (base name) If there are two or more substituents, give each one a number and list them alphabetically with hyphens between words and numbers. 5. If a compound has two or more ide ...
Synthesis and Characterisation of N
... compounds containing low-valent main group elements. Recently, more work was put forth in the synthesis and study of molecules containing phosphorous in its +1 oxidation state (PI). One way of stabilizing the PI center is by using N-heterocyclic carbenes (NHCs). Our group previously reported several ...
... compounds containing low-valent main group elements. Recently, more work was put forth in the synthesis and study of molecules containing phosphorous in its +1 oxidation state (PI). One way of stabilizing the PI center is by using N-heterocyclic carbenes (NHCs). Our group previously reported several ...
SBI4U: Biochemistry Functional Groups Activity Molecular Shape
... bonding to another atom. This functional accepts protons (H+ ions) in solution, which decreases the[H+ ion] and increases the pH. ...
... bonding to another atom. This functional accepts protons (H+ ions) in solution, which decreases the[H+ ion] and increases the pH. ...
ChemistryPPT
... Describe a chemical reaction that you have observed in everyday life. Underline: 3 pieces of quantitative info Underline: 5 pieces of qualitative info How did the chemical reaction start? What did it look like? What was the end result? How did you know it was a chemical ...
... Describe a chemical reaction that you have observed in everyday life. Underline: 3 pieces of quantitative info Underline: 5 pieces of qualitative info How did the chemical reaction start? What did it look like? What was the end result? How did you know it was a chemical ...
4.8 Preparation of Alkyl Halides from Alcohols and Hydrogen
... electrons in this σ bond can be shared by positively charged carbon because the s orbital can overlap with the empty 2p orbital of positively charged carbon ...
... electrons in this σ bond can be shared by positively charged carbon because the s orbital can overlap with the empty 2p orbital of positively charged carbon ...
Chemdraw B&W - Pennsylvania State University
... • Alkylation occurs when the nucleophilic enolate ion reacts with the electrophilic alkyl halide or tosylate and displaces the leaving group ...
... • Alkylation occurs when the nucleophilic enolate ion reacts with the electrophilic alkyl halide or tosylate and displaces the leaving group ...
Aromatic Substitution Reactions
... ophile, in a fashion very similar to the addition reactions described in Chapter 11, which begin by reaction of an electrophile with the pi electrons of an alkene. This results in the formation of a carbocation called an arenium ion. Removal of a proton from the arenium ion by some weak base that is ...
... ophile, in a fashion very similar to the addition reactions described in Chapter 11, which begin by reaction of an electrophile with the pi electrons of an alkene. This results in the formation of a carbocation called an arenium ion. Removal of a proton from the arenium ion by some weak base that is ...
Homoaromaticity

Homoaromaticity in organic chemistry refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thought to be a requirement for aromaticity, considerable thermodynamic stability and many of the spectroscopic, magnetic, and chemical properties associated with aromatic compounds are still observed for such compounds. This formal discontinuity is apparently bridged by p-orbital overlap, maintaining a contiguous cycle of π electrons that is responsible for this preserved chemical stability.The concept of homoaromaticity was pioneered by Saul Winstein in 1959, prompted by his studies of the “tris-homocyclopropenyl” cation. Since the publication of Winstein's paper, much research has been devoted to understanding and classifying these molecules, which represent an additional “class” of aromatic molecules included under the continuously broadening definition of aromaticity. To date, homoaromatic compounds are known to exist as cationic and anionic species, and some studies support the existence of neutral homoaromatic molecules, though these are less common. The 'homotropylium' cation (C8H9+) is perhaps the best studied example of a homoaromatic compound.