Introduction to Bioinorganic Chemistry
... => Acces to the spin state of a paramagnetic metal ion. Once the magnetic properties of the metal centre are known, the measurement of the chemical shifts and relaxation times allows insights into the structural parameters of the active site, in particular the distance between the paramagnetic centr ...
... => Acces to the spin state of a paramagnetic metal ion. Once the magnetic properties of the metal centre are known, the measurement of the chemical shifts and relaxation times allows insights into the structural parameters of the active site, in particular the distance between the paramagnetic centr ...
New Palladium Polymerisation Catalyst Systems
... cation may undergo chain migration before being trapped by ethylene, resulting in chain branching. The extent of the chain migration, and thus the extent of the branching, can be controlled via the parameters of the physical process. The steric hindrance caused by the bulky diimine ligands leads to ...
... cation may undergo chain migration before being trapped by ethylene, resulting in chain branching. The extent of the chain migration, and thus the extent of the branching, can be controlled via the parameters of the physical process. The steric hindrance caused by the bulky diimine ligands leads to ...
Complexation and Protein Binding
... containing cupric ions should result in a decrease in pH. Titration curves can be obtained by adding a strong base to a solution of glycine and to another solution containing glycine and a copper salt and plotting the pH against the equivalents of base added. The results of such a potentiometric tit ...
... containing cupric ions should result in a decrease in pH. Titration curves can be obtained by adding a strong base to a solution of glycine and to another solution containing glycine and a copper salt and plotting the pH against the equivalents of base added. The results of such a potentiometric tit ...
Chapter 24
... molecule of the type H2A with four electrons is predicted to be linear. NH2 has one electron fewer than H2O, and using the same reasoning as for water, is bent. Chapter 24: Molecular Structure and Energy Levels for Polyatomic Molecules Physical Chemistry 2nd Edition © 2010 Pearson Education South As ...
... molecule of the type H2A with four electrons is predicted to be linear. NH2 has one electron fewer than H2O, and using the same reasoning as for water, is bent. Chapter 24: Molecular Structure and Energy Levels for Polyatomic Molecules Physical Chemistry 2nd Edition © 2010 Pearson Education South As ...
Electronic Structure and the Periodic Table
... describe their electron configurations. For any element, you know the number of electrons in the neutral atom equals the atomic number. For an ion, the number of electrons equals the number of protons minus the charge. Start filling orbitals, from lowest to highest energy. If two or more orbitals ex ...
... describe their electron configurations. For any element, you know the number of electrons in the neutral atom equals the atomic number. For an ion, the number of electrons equals the number of protons minus the charge. Start filling orbitals, from lowest to highest energy. If two or more orbitals ex ...
Electronic structure of tris(8-hydroxyquinoline) states
... peak appears at lower binding energy in the N(1s) spectrum, indicating that charge has been transferred from the metal atoms to the nitrogen atoms. In addition, the original N(1s) peak gradually disappears during the course of metal deposition. The next-to-the-lowest curves correspond to an approxim ...
... peak appears at lower binding energy in the N(1s) spectrum, indicating that charge has been transferred from the metal atoms to the nitrogen atoms. In addition, the original N(1s) peak gradually disappears during the course of metal deposition. The next-to-the-lowest curves correspond to an approxim ...
Applications of UV
... Many transition metals have colored solutions and are also colored in the solid state. The transition metals have some of their d orbitals empty where a d-d transition can occur. The d-d transitions require excitation energy in the UV-Vis region. The direct interaction of the d electrons with ligand ...
... Many transition metals have colored solutions and are also colored in the solid state. The transition metals have some of their d orbitals empty where a d-d transition can occur. The d-d transitions require excitation energy in the UV-Vis region. The direct interaction of the d electrons with ligand ...
Answers - U of L Class Index
... It has three-center two-electron bonds (i.e. bonds in which the pair of electrons are shared between three atoms instead of the usual two). This is shown more clearly in the structure on the left. ...
... It has three-center two-electron bonds (i.e. bonds in which the pair of electrons are shared between three atoms instead of the usual two). This is shown more clearly in the structure on the left. ...
Document
... • Structural isomers are molecules that have the same number and type of atoms, but they are attached in a different order. • Stereoisomers are molecules that have the same number and type of atoms, and that are attached in the same order, but the atoms or groups of atoms point in a different spatia ...
... • Structural isomers are molecules that have the same number and type of atoms, but they are attached in a different order. • Stereoisomers are molecules that have the same number and type of atoms, and that are attached in the same order, but the atoms or groups of atoms point in a different spatia ...
Cu II complex - IONiC / VIPEr
... “relatively stable” (meaning relative to its CuII complex under the controlled experimental conditions). 3. Suggest why they are soluble in H2O. 4. It turns out for the reaction: CuI + n L CuILn Kf(CuIMe6Trien) < Kf(CuIBCA2) < Kf(CuIBCS2) Suggest why it is useful to have 3 ligands with 3 differing ...
... “relatively stable” (meaning relative to its CuII complex under the controlled experimental conditions). 3. Suggest why they are soluble in H2O. 4. It turns out for the reaction: CuI + n L CuILn Kf(CuIMe6Trien) < Kf(CuIBCA2) < Kf(CuIBCS2) Suggest why it is useful to have 3 ligands with 3 differing ...
2P32 - Pilkington Group
... For [M(H20)6]n+ in order for the metal ion to accommodate six ligands it must have at least a 52 pm radius. Six waters fit exactly around a metal ion with 52 pm radius (not crowded). At 92 pm’s the six waters have moved far enough apart that more waters can fit. If the metal ion is smaller than 52pm ...
... For [M(H20)6]n+ in order for the metal ion to accommodate six ligands it must have at least a 52 pm radius. Six waters fit exactly around a metal ion with 52 pm radius (not crowded). At 92 pm’s the six waters have moved far enough apart that more waters can fit. If the metal ion is smaller than 52pm ...
spin and electron density redistribution upon binding of non
... biomimetic systems). Practical quantum chemistry, although rooted in firstprinciples quantum mechanics, suffers from the separation of nuclear and electronic motions within Born-Oppenheimer (B.-O.) approximation. Therefore, robust quantum-chemical methods (like the density functional theory, DFT) offer ...
... biomimetic systems). Practical quantum chemistry, although rooted in firstprinciples quantum mechanics, suffers from the separation of nuclear and electronic motions within Born-Oppenheimer (B.-O.) approximation. Therefore, robust quantum-chemical methods (like the density functional theory, DFT) offer ...
Chapter 24. Organic Chemistry
... pairs form bonds with hydrogen atoms and other two are lone pairs. To minimize electrostatic repulsion between the electron pairs, all four pairs form tetrahedral arrangement. The repulsion interaction between pairs involved in bonds is weaker, and the repulsion interaction between lone pairs is str ...
... pairs form bonds with hydrogen atoms and other two are lone pairs. To minimize electrostatic repulsion between the electron pairs, all four pairs form tetrahedral arrangement. The repulsion interaction between pairs involved in bonds is weaker, and the repulsion interaction between lone pairs is str ...
Chapter 21 Transition Metals
... which show chemical similarities only within the group, the transition metals show great similarities within a group as well as within a given period. Most transition metals have high melting points and high densities. They are hard and strong; most have more than one oxidation states in their commo ...
... which show chemical similarities only within the group, the transition metals show great similarities within a group as well as within a given period. Most transition metals have high melting points and high densities. They are hard and strong; most have more than one oxidation states in their commo ...
lecture_notes_5
... c. Situations in which there is a “special metal”, i.e., the metal is a transition metal or a group 14 metal. i. The bad news about these metals is that it is very difficult for a 1st year chemistry student to figure out how a transition metal forms an ion. ii. For instance, iron (Fe) has the electr ...
... c. Situations in which there is a “special metal”, i.e., the metal is a transition metal or a group 14 metal. i. The bad news about these metals is that it is very difficult for a 1st year chemistry student to figure out how a transition metal forms an ion. ii. For instance, iron (Fe) has the electr ...
How molecules become magnetic - The following sites may be
... In everyday life, the word “magnetism” most often refers to solid materials, metals, alloys, oxides. As for molecules, they are considered to be isolated, non-magnetic objects. Nevertheless, to be or not to be (magnetic) is not the appropriate question: everything is magnetic; the true problem lies ...
... In everyday life, the word “magnetism” most often refers to solid materials, metals, alloys, oxides. As for molecules, they are considered to be isolated, non-magnetic objects. Nevertheless, to be or not to be (magnetic) is not the appropriate question: everything is magnetic; the true problem lies ...
5.4 Transition Metals notes - A
... When metal ions are in solution, they are usually represented as the simple ion, such as Fe2+(aq), Co2+(aq), Cr3+(aq) or Fe2+(aq). This is, however, a simplified representation as all d-block cations and many other cations with high polarising power exist as the hexaaqua complex, eg [Fe(H2O)6]2+. Eg ...
... When metal ions are in solution, they are usually represented as the simple ion, such as Fe2+(aq), Co2+(aq), Cr3+(aq) or Fe2+(aq). This is, however, a simplified representation as all d-block cations and many other cations with high polarising power exist as the hexaaqua complex, eg [Fe(H2O)6]2+. Eg ...
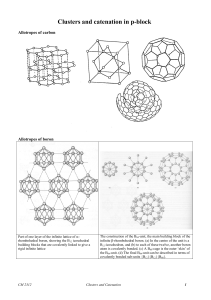
Clusters
... electrons are said to be isoelectronic. However, this term is often used to refer to species that simply possess the same number of valence electrons. Example: BN is isoelectronic with CC The structure of layered, hexagonal boron Nitride, note that the atoms are in register ...
... electrons are said to be isoelectronic. However, this term is often used to refer to species that simply possess the same number of valence electrons. Example: BN is isoelectronic with CC The structure of layered, hexagonal boron Nitride, note that the atoms are in register ...
Test Objectives: Unit 1 – Measurement
... o know that when a bond is broken, energy is absorbed; when a bond is made, energy is released o define the term covalent bond and list the properties of covalent substances o know how to draw Lewis Dot structures for covalent compounds o distinguish between non-polar & polar covalent bonds o be abl ...
... o know that when a bond is broken, energy is absorbed; when a bond is made, energy is released o define the term covalent bond and list the properties of covalent substances o know how to draw Lewis Dot structures for covalent compounds o distinguish between non-polar & polar covalent bonds o be abl ...
Jahn-Teller assisted Na diffusion for high performance Na ion batteries
... Fe4+O6 octahedron. The new mechanism could potentially be applied to any type of sodium intercalation compound whenever the Jahn−Teller active transition metal is facesharing with the activated site for sodium diffusion, including channeled31 and zigzag layered structures.32 A Jahn−Teller (JT) distor ...
... Fe4+O6 octahedron. The new mechanism could potentially be applied to any type of sodium intercalation compound whenever the Jahn−Teller active transition metal is facesharing with the activated site for sodium diffusion, including channeled31 and zigzag layered structures.32 A Jahn−Teller (JT) distor ...
i PREFACE The ability of Schiff bases to form complexes with metal
... The ability of Schiff bases to form complexes with metal ions has been well established. This is due to the presence of lone pair of electrons on the nitrogen atom and of the general electron donating character of the double bond. They also form five or, six membered stable chelates with metals, if ...
... The ability of Schiff bases to form complexes with metal ions has been well established. This is due to the presence of lone pair of electrons on the nitrogen atom and of the general electron donating character of the double bond. They also form five or, six membered stable chelates with metals, if ...
Studying of transition metal complexes containing
... calculations were performed by using a Personal Computer with a Core i7 processor (CPU 2.20GHz) and (RAM 8GB), with a Windows7 operating system, and these computational works used five software, and figures are sketched by ChemBio office v.13.0 (2012)(32). While molecular operating environment MOE v ...
... calculations were performed by using a Personal Computer with a Core i7 processor (CPU 2.20GHz) and (RAM 8GB), with a Windows7 operating system, and these computational works used five software, and figures are sketched by ChemBio office v.13.0 (2012)(32). While molecular operating environment MOE v ...
Jahn–Teller effect
-3D-balls.png?width=300)
The Jahn–Teller effect, sometimes also known as Jahn–Teller distortion, describes the geometrical distortion of molecules and ions that is associated with certain electron configurations. This electronic effect is named after Hermann Arthur Jahn and Edward Teller, who proved, using group theory, that orbital nonlinear spatially degenerate molecules cannot be stable. The Jahn–Teller theorem essentially states that any nonlinear molecule with a spatially degenerate electronic ground state will undergo a geometrical distortion that removes that degeneracy, because the distortion lowers the overall energy of the species. For a description of another type of geometrical distortion that occurs in crystals with substitutional impurities see article off-center ions.