The Historical X-ray Transient KY TrA in quiescence
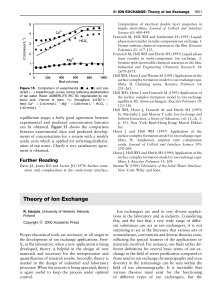
... centre. For comparison we have also marked the star labeled as ‘S’ in that paper. In this figure, we also show a zoom of the central region of 30×30 arcsec where an elongation of the source profile along the NW direction is clearly visible. We measure an elongation coefficient (the IRAF ‘ellipsoidal ...
... centre. For comparison we have also marked the star labeled as ‘S’ in that paper. In this figure, we also show a zoom of the central region of 30×30 arcsec where an elongation of the source profile along the NW direction is clearly visible. We measure an elongation coefficient (the IRAF ‘ellipsoidal ...
The destruction and survival of polycyclic aromatic hydrocarbons in
... In Spitzer observations of Tauri stars and their disks, features of polycyclic aromatic hydrocarbons (PAHs) are detected in less than 10% of the objects, although the stellar photosphere is sufficiently hot to excite PAHs. To explain the deficiency, we discuss PAH destruction by photons, assuming that ...
... In Spitzer observations of Tauri stars and their disks, features of polycyclic aromatic hydrocarbons (PAHs) are detected in less than 10% of the objects, although the stellar photosphere is sufficiently hot to excite PAHs. To explain the deficiency, we discuss PAH destruction by photons, assuming that ...
Targets
... Production of RIB's via the fission process induced - either by fast neutrons from a C converter in a UCx target goal >1013 fissions/s - or by direct bombardment of fissile material Production of heavy ion beams for fusion-evaporation physics C D ...
... Production of RIB's via the fission process induced - either by fast neutrons from a C converter in a UCx target goal >1013 fissions/s - or by direct bombardment of fissile material Production of heavy ion beams for fusion-evaporation physics C D ...
Chapter 8 Concepts of Chemical Bonding
... • G. N. Lewis developed a method to denote potential bonding electrons by using one dot for every valence electron around the element symbol. • When forming compounds, atoms tend to gain, lose, or share electrons until they are surrounded by eight valence electrons - electrons involved in bonding ( ...
... • G. N. Lewis developed a method to denote potential bonding electrons by using one dot for every valence electron around the element symbol. • When forming compounds, atoms tend to gain, lose, or share electrons until they are surrounded by eight valence electrons - electrons involved in bonding ( ...
Jahn−Teller Distortion in the Phosphorescent Excited State of Three
... Luminescent transition metal complexes have been receiving increasing attention due to their role in a variety of optoelectronic applications. For example, molecular light-emitting devices (LEDs) have been increasingly reported1 to exhibit superior efficiencies when phosphorescence is enhanced by th ...
... Luminescent transition metal complexes have been receiving increasing attention due to their role in a variety of optoelectronic applications. For example, molecular light-emitting devices (LEDs) have been increasingly reported1 to exhibit superior efficiencies when phosphorescence is enhanced by th ...
From: BXSVR0::OWOCKI 12-JAN-2001 10:16:41.12 To: DCOHEN
... the original referee was the question of the line optical depth for strong permitted lines. I have done a quickie calculation. One can construct a kind of upper limit to the line optical depth for any given ion species using Sobolev theory. It helps to think about the integrated line optical depth T ...
... the original referee was the question of the line optical depth for strong permitted lines. I have done a quickie calculation. One can construct a kind of upper limit to the line optical depth for any given ion species using Sobolev theory. It helps to think about the integrated line optical depth T ...
Chapter 10
... Bonding in CH4 How do we account for 4 C—H sigma bonds 109o apart? Need to use 4 atomic orbitals — s, px, py, and pz — to form 4 new hybrid orbitals pointing in the correct direction. Dr. S. M. Condren ...
... Bonding in CH4 How do we account for 4 C—H sigma bonds 109o apart? Need to use 4 atomic orbitals — s, px, py, and pz — to form 4 new hybrid orbitals pointing in the correct direction. Dr. S. M. Condren ...
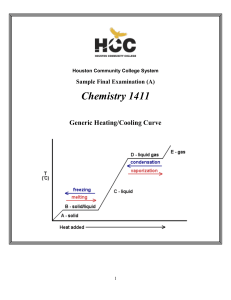
CHEM-1411 Final Practice Exam
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
Correlation between Crystal Structure and Mobility in Organic Field
... Abstract: Recently, it was reported that crystals of the organic material dithiophene-tetrathiafulvalene (DTTTF) have a high field-effect charge carrier mobility of 1.4 cm2/(V‚s). These crystals were formed by a simple drop-casting method, making this material interesting to investigate for possible ...
... Abstract: Recently, it was reported that crystals of the organic material dithiophene-tetrathiafulvalene (DTTTF) have a high field-effect charge carrier mobility of 1.4 cm2/(V‚s). These crystals were formed by a simple drop-casting method, making this material interesting to investigate for possible ...
Turbulent convection in stellar interiors
... • Minimum Residual Approximate Implicit scheme (c.f. Botchev et al. 1997) • Perform an explicit step with a large (unstable!) time step (15-20x Δtstab) • Stabilise the scheme by performing a few GMRES iterations on the implicit residual • Pro: Jacobian-free GMRES (i.e. tractable in 3D!), contra: con ...
... • Minimum Residual Approximate Implicit scheme (c.f. Botchev et al. 1997) • Perform an explicit step with a large (unstable!) time step (15-20x Δtstab) • Stabilise the scheme by performing a few GMRES iterations on the implicit residual • Pro: Jacobian-free GMRES (i.e. tractable in 3D!), contra: con ...
R E S U M E - UCF College of Sciences
... electrons within the atoms and molecules through theoretical analysis of the interaction of photons, electrons and positrons with individual atoms and molecules in the gas phase. Over the past twenty years some of the most productive activities in atomic physics research have been in the area of pho ...
... electrons within the atoms and molecules through theoretical analysis of the interaction of photons, electrons and positrons with individual atoms and molecules in the gas phase. Over the past twenty years some of the most productive activities in atomic physics research have been in the area of pho ...
0004-637X 778 2 119
... and with similar basic disk parameters, motivates us to further test the model by constraints provided by the sources with extreme properties. Here, we investigate the long-term evolution of the second “low-B magnetar,” Swift J1822.3–1606. We try to determine the evolutionary epoch of the source and ...
... and with similar basic disk parameters, motivates us to further test the model by constraints provided by the sources with extreme properties. Here, we investigate the long-term evolution of the second “low-B magnetar,” Swift J1822.3–1606. We try to determine the evolutionary epoch of the source and ...
Student Solutions Manual Errata
... spheres in the diagram) as being separate from, but strongly attracted to, one another. Covalent bonding occurs when two atoms are mutually attracted to a pair (or pairs) of electrons. Because the atoms share the electrons, we can think of the atoms (or the spheres in the diagram) as being joined to ...
... spheres in the diagram) as being separate from, but strongly attracted to, one another. Covalent bonding occurs when two atoms are mutually attracted to a pair (or pairs) of electrons. Because the atoms share the electrons, we can think of the atoms (or the spheres in the diagram) as being joined to ...
chapter twenty-one transition metals and coordination chemistry
... Chromium ([Ar]:4s03d5) and copper [Ar]:4s13d10) have electron configurations which are different from that predicted from the periodic table. Other exceptions to the predicted filling order are transition metal ions. These all lose the s electrons before they lose the d electrons. In neutral atoms, ...
... Chromium ([Ar]:4s03d5) and copper [Ar]:4s13d10) have electron configurations which are different from that predicted from the periodic table. Other exceptions to the predicted filling order are transition metal ions. These all lose the s electrons before they lose the d electrons. In neutral atoms, ...
- Te Kura
... under the most powerful microscope. How do we know what its structure is? Are all atoms the same? Can one atom be changed into another kind of atom? People have pondered these questions for the last few thousand years, from the alchemists who have tried to turn lead into gold to the physicists tryin ...
... under the most powerful microscope. How do we know what its structure is? Are all atoms the same? Can one atom be changed into another kind of atom? People have pondered these questions for the last few thousand years, from the alchemists who have tried to turn lead into gold to the physicists tryin ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.