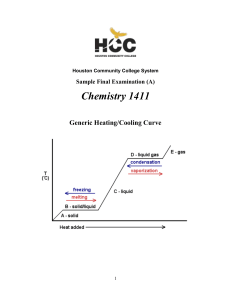
1411FINALSAMPLEs and Key
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
Propagation and Scattering of High-Intensity X
... highly charged ions. In heavy atoms such as xenon the absorption of 8 x-ray photons with 1.5 keV photon energy leads to charge states as high as Xe36+ [17]. The efficient photo ionization of matter leads to severe radiation damage. The positively charged ions repel each other and lead to a Coulomb e ...
... highly charged ions. In heavy atoms such as xenon the absorption of 8 x-ray photons with 1.5 keV photon energy leads to charge states as high as Xe36+ [17]. The efficient photo ionization of matter leads to severe radiation damage. The positively charged ions repel each other and lead to a Coulomb e ...
CHEMISTRY
... units. I use dimensional analysis to do metric-metric conversions, stressing that many different types of calculations can be done using dimensional analysis. I will also intentionally set up a problem incorrectly and show the students that the units do not cancel, demonstrating the importance of ch ...
... units. I use dimensional analysis to do metric-metric conversions, stressing that many different types of calculations can be done using dimensional analysis. I will also intentionally set up a problem incorrectly and show the students that the units do not cancel, demonstrating the importance of ch ...
Workshop on Semiconductor Materials and Detectors,
... that detection efficiency of CdTe, CdZnTe, TlBr semiconductor for gamma-ray and X-ray is high. In current-voltage characteristic and capacitance-voltage characteristic, CdTe and CdZnTe semiconductor have schottky junction and TlBr has ohmic contact. And when CdTe and CdZnTe compared, the leakage cur ...
... that detection efficiency of CdTe, CdZnTe, TlBr semiconductor for gamma-ray and X-ray is high. In current-voltage characteristic and capacitance-voltage characteristic, CdTe and CdZnTe semiconductor have schottky junction and TlBr has ohmic contact. And when CdTe and CdZnTe compared, the leakage cur ...
UNIT-VII (A) LASER Engineering Physics 1. Introduction:
... Working:When an electric discharge is posses the He-Ne gas mixture, helium atoms are excited to higher levels He2 and He3 through collisions with accelerated electrons. In this neon atom contain six energy levels Ne1 , Ne2 , Ne3 , Ne4 , Ne5 and Ne6 . Here it should be noted that Ne4 and He2 have sam ...
... Working:When an electric discharge is posses the He-Ne gas mixture, helium atoms are excited to higher levels He2 and He3 through collisions with accelerated electrons. In this neon atom contain six energy levels Ne1 , Ne2 , Ne3 , Ne4 , Ne5 and Ne6 . Here it should be noted that Ne4 and He2 have sam ...
Holt Modern Chemistry Workbook: intro - ch 5
... technology of fiber optic cables that carry television, telephone, and computer signals. ...
... technology of fiber optic cables that carry television, telephone, and computer signals. ...
Hunting for Orphaned Central Compact Objects among Radio Pulsars
... the small number of known pulsating CCOs and the possibility of small beaming fractions, the result is inconclusive and a more systematic study is needed. In addition to deeper radio observations, we can turn the search around to look for CCO-like X-ray emission from selected weak-field radio pulsar ...
... the small number of known pulsating CCOs and the possibility of small beaming fractions, the result is inconclusive and a more systematic study is needed. In addition to deeper radio observations, we can turn the search around to look for CCO-like X-ray emission from selected weak-field radio pulsar ...
Text - Enlighten: Publications
... Ar ¼ 2,6-Me2-C6H3; ArF ¼ 3,5-(CF3)2-C6H3) have been studied by Cu K-edge X-ray absorption spectroscopy, as well as single- and multi-reference computational methods (DFT, TD-DFT, CASSCF, MRCI, and OVB). The study was extended to a range of both known and theoretical compounds bearing 2p-element dono ...
... Ar ¼ 2,6-Me2-C6H3; ArF ¼ 3,5-(CF3)2-C6H3) have been studied by Cu K-edge X-ray absorption spectroscopy, as well as single- and multi-reference computational methods (DFT, TD-DFT, CASSCF, MRCI, and OVB). The study was extended to a range of both known and theoretical compounds bearing 2p-element dono ...
Chemistry - RESONANCE PCCP IDEAL for NTSE, IJSO, Olympiads
... (iv) To get the empirical formula, symbols of various elements present are written side by side with their respective whole number ratio as a subscript to the lower right hand corner of the symbol. (v) The molecular formula of a substance may be determined from the empirical formula if the molecula ...
... (iv) To get the empirical formula, symbols of various elements present are written side by side with their respective whole number ratio as a subscript to the lower right hand corner of the symbol. (v) The molecular formula of a substance may be determined from the empirical formula if the molecula ...
Molecular diffusion at surfaces
... of the square move 0.18 A away to a distance of 2.09 A and the other two Fe atoms at corners of the other side also relax 0.12 A away. The four Fe atom surrounding the two Fe atoms at the apexes move 0.18 A. The other four atoms surrounding the two Fe atoms close to C move 0. 13 A. Twelve more surro ...
... of the square move 0.18 A away to a distance of 2.09 A and the other two Fe atoms at corners of the other side also relax 0.12 A away. The four Fe atom surrounding the two Fe atoms at the apexes move 0.18 A. The other four atoms surrounding the two Fe atoms close to C move 0. 13 A. Twelve more surro ...
FERMI GBM detections of four AXPs at soft gamma-rays
... field. These starquakes can be observed as outbursts and timing glitches. The untwisting of the strong magnetic field leads to a current of charged particles along the magnetic field lines which heats the stellar surface. Historically there are two types of magnetar candidates, Soft Gamma Repeaters ...
... field. These starquakes can be observed as outbursts and timing glitches. The untwisting of the strong magnetic field leads to a current of charged particles along the magnetic field lines which heats the stellar surface. Historically there are two types of magnetar candidates, Soft Gamma Repeaters ...
Coordination Chemistry of cis,trans-1,3,5
... The coordination chemistry of the rigid, aliphatic triamino ligand cis,trans-1,3,5triaminocyclohexane (trans-tach) is investigated. With closed shell transition metals, transtach forms 1-D {Ag(OTf), ZnCl2} networks and a 3-D {AgNO3} coordination polymer with unprecedented topology. Coordination to t ...
... The coordination chemistry of the rigid, aliphatic triamino ligand cis,trans-1,3,5triaminocyclohexane (trans-tach) is investigated. With closed shell transition metals, transtach forms 1-D {Ag(OTf), ZnCl2} networks and a 3-D {AgNO3} coordination polymer with unprecedented topology. Coordination to t ...
Effect of N-donor ancillary ligands on structural and magnetic
... to metal joints and play a key role in the design of new functional coordination networks. They have presented highly versatile binding modes (Scheme 1) and also serve as good hydrogen-bonding participants to give rise to higher dimensional structures. On the other hand, oxalate bridges act as efficie ...
... to metal joints and play a key role in the design of new functional coordination networks. They have presented highly versatile binding modes (Scheme 1) and also serve as good hydrogen-bonding participants to give rise to higher dimensional structures. On the other hand, oxalate bridges act as efficie ...
Chem I Review Part 2
... 69. Which of these ionic solids would have the largest lattice energy? A. NaCl B. NaF C. CaBr2 D. CsI E. CaCl2 70. Which of these solids would have the highest melting point? A. NaF B. NaCl C. NaBr D. NaI 71. Which of these elements has the greatest electronegativity? A. Na B. As C. Ga D. Cs E. Sb ...
... 69. Which of these ionic solids would have the largest lattice energy? A. NaCl B. NaF C. CaBr2 D. CsI E. CaCl2 70. Which of these solids would have the highest melting point? A. NaF B. NaCl C. NaBr D. NaI 71. Which of these elements has the greatest electronegativity? A. Na B. As C. Ga D. Cs E. Sb ...
Covalent Bonding and Nomenclature
... The main idea behind VSEPR theory is that electron pairs (bonding and nonbonding) will ...
... The main idea behind VSEPR theory is that electron pairs (bonding and nonbonding) will ...
Ca(ii), Cd(ii), Cu(ii) and Pb(ii)
... the equilibrium states, carry out quantitative analysis of stability constants or evaluate the binding selectivities.24–39 This method is considered to be a ‘‘soft ionization’’ process (involving the transfer of solution ions into the gas phase), and therefore typically yields molecular ions with li ...
... the equilibrium states, carry out quantitative analysis of stability constants or evaluate the binding selectivities.24–39 This method is considered to be a ‘‘soft ionization’’ process (involving the transfer of solution ions into the gas phase), and therefore typically yields molecular ions with li ...
CHE 110 Dr. Nicholas Bizier Office DS 337b email
... composition: C, H, O, N. In one experiment, 2.175 g of lysine was combusted to produce 3.94 g of CO2 and 1.89 g H2O. In a separate experiment, 1.873 g of lysine was burned to produce 0.436 g of NH2. The molar mass of lysine is 150 g/mol. Determine the empirical and molecular formula of lysine. ...
... composition: C, H, O, N. In one experiment, 2.175 g of lysine was combusted to produce 3.94 g of CO2 and 1.89 g H2O. In a separate experiment, 1.873 g of lysine was burned to produce 0.436 g of NH2. The molar mass of lysine is 150 g/mol. Determine the empirical and molecular formula of lysine. ...
FREE Sample Here
... E) There are covalent bonds between the hydrogen atoms. Answer: A Topic: Concept 2.3 Skill: Knowledge/Comprehension 46) When two atoms are equally electronegative, they will interact to form A) equal numbers of isotopes. B) ions. C) polar covalent bonds. D) nonpolar covalent bonds. E) ionic bonds. A ...
... E) There are covalent bonds between the hydrogen atoms. Answer: A Topic: Concept 2.3 Skill: Knowledge/Comprehension 46) When two atoms are equally electronegative, they will interact to form A) equal numbers of isotopes. B) ions. C) polar covalent bonds. D) nonpolar covalent bonds. E) ionic bonds. A ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.