Chem 1a Midterm Review
... 2. arrange the atoms in a structure and distribute the electrons so that each atom has 8 electrons around it (exceptions, H has 2, B can have 6 and third row and lower atoms can have more than 8). 3. Try to maximize the number of bonds that each atom has. 4. Calculated the partial charge on each ato ...
... 2. arrange the atoms in a structure and distribute the electrons so that each atom has 8 electrons around it (exceptions, H has 2, B can have 6 and third row and lower atoms can have more than 8). 3. Try to maximize the number of bonds that each atom has. 4. Calculated the partial charge on each ato ...
A1983PW59500001
... man in Paris. It was he who dug out my paper and suggested to Tejiro Yonezawa and me 1 2 that we apply my method to amino acids. . Only then did the ‘Del Re method’ start its career. A curious one indeed, as I could see when reviewing the method in 1980.~In spite of its simplicity, and in spite of b ...
... man in Paris. It was he who dug out my paper and suggested to Tejiro Yonezawa and me 1 2 that we apply my method to amino acids. . Only then did the ‘Del Re method’ start its career. A curious one indeed, as I could see when reviewing the method in 1980.~In spite of its simplicity, and in spite of b ...
Dr. Ali Abadi Lecture 1 Materials Properties CH. 1: Structure of
... electron → high electronegativity. Metals are electropositive – they can give up their few valence electrons to become positively charged ions. The electron volt (eV) – energy unit convenient for description of atomic bonding Electron volt - the energy lost / gained by an electron when it is taken t ...
... electron → high electronegativity. Metals are electropositive – they can give up their few valence electrons to become positively charged ions. The electron volt (eV) – energy unit convenient for description of atomic bonding Electron volt - the energy lost / gained by an electron when it is taken t ...
C1 Revision Fundamental ideas adapted CS
... Elements are shown in the P...................... T................... Metals are found on the l................. and c..................., non-metals are found on the r................ of the P...................... T.................... Elements in the same g.................... have similar p.... ...
... Elements are shown in the P...................... T................... Metals are found on the l................. and c..................., non-metals are found on the r................ of the P...................... T.................... Elements in the same g.................... have similar p.... ...
Atomic combinations: Electronegativity and ionic
... electronegativity is large, one atom will attract the shared electron pair much more strongly than the other, causing electrons to be transferred from one atom to the other. ...
... electronegativity is large, one atom will attract the shared electron pair much more strongly than the other, causing electrons to be transferred from one atom to the other. ...
Midterm Review 1
... 4. Which of the following are isotopes of the same element? 9Y 9Y 10Y 5. Describe Rutherford's experiment: ___________________________________________________________________ ___________________________________________________________________________________________ _________________________________ ...
... 4. Which of the following are isotopes of the same element? 9Y 9Y 10Y 5. Describe Rutherford's experiment: ___________________________________________________________________ ___________________________________________________________________________________________ _________________________________ ...
Webquest Review - Harrison High School
... polar and will have dipole-dipole interactions between molecules. That means CH3Cl will be harder to boil than CO2. H2O has H bonded to O and it’s polar. When you have species with H bonded to F, O, or N, hydrogen “bond”ing sets up between the molecules. Hydrogen bonding is the strongest intermolecu ...
... polar and will have dipole-dipole interactions between molecules. That means CH3Cl will be harder to boil than CO2. H2O has H bonded to O and it’s polar. When you have species with H bonded to F, O, or N, hydrogen “bond”ing sets up between the molecules. Hydrogen bonding is the strongest intermolecu ...
Covalent bonding
... Chemical bond formed by sharing a pair of eEach atom gains an octet of e- by sharing electron pairs, e.g. ...
... Chemical bond formed by sharing a pair of eEach atom gains an octet of e- by sharing electron pairs, e.g. ...
Intermolecular Forces Practice Problems
... What holds two fluorine molecules together in a sample of liquid fluorine? ...
... What holds two fluorine molecules together in a sample of liquid fluorine? ...
Chapter1011
... • Use valence bond theory to describe the bonding in the following (use clear 3-D pictures showing orbital overlap, etc) H2O NH3 CH4 PF3 --simple s bonds and lone pairs H2CNH --double bond like H2CCH2 ethene and H2CO formaldehyde) HCN --triple bond like HCCH ethyne and N2 nitrogen) ...
... • Use valence bond theory to describe the bonding in the following (use clear 3-D pictures showing orbital overlap, etc) H2O NH3 CH4 PF3 --simple s bonds and lone pairs H2CNH --double bond like H2CCH2 ethene and H2CO formaldehyde) HCN --triple bond like HCCH ethyne and N2 nitrogen) ...
Bonding
... electrons). KNO3 does not conduct because it is ionically bonded and has immobile ions (or immobile electrons). (b) SbCl3 has a measurable dipole moment because it has a lone pair of electrons which causes a dipole - or - its dipoles do not cancel - or - it has a trigonal pyramidal structure - or - ...
... electrons). KNO3 does not conduct because it is ionically bonded and has immobile ions (or immobile electrons). (b) SbCl3 has a measurable dipole moment because it has a lone pair of electrons which causes a dipole - or - its dipoles do not cancel - or - it has a trigonal pyramidal structure - or - ...
Chemistry 1 Revision: Metals and their uses
... Elements are shown in the P...................... T................... Metals are found on the l................. and c..................., non-metals are found on the r................ of the P...................... T.................... Elements in the same g.................... have similar p.... ...
... Elements are shown in the P...................... T................... Metals are found on the l................. and c..................., non-metals are found on the r................ of the P...................... T.................... Elements in the same g.................... have similar p.... ...
10th Grade Chemistry X (TJ) GRADE(S)/LEVELS SUBJECT Power
... LT 1 Predict the properties of elements in the periodic table, based on their position in a group and period. LT 2 Predict periodic table trends (e.g., electronegativity, reactivity, electron affinity, atomic radius, ionic radius). ...
... LT 1 Predict the properties of elements in the periodic table, based on their position in a group and period. LT 2 Predict periodic table trends (e.g., electronegativity, reactivity, electron affinity, atomic radius, ionic radius). ...
Review of Definitions
... 1. Intramolecular Forces. Forces between atoms that exist within individual molecules. 2. Intermolecular Forces. Forces that exist between molecules. 3. Electronegativity. The ability of an atom to attract to itself an electron pair shared with ...
... 1. Intramolecular Forces. Forces between atoms that exist within individual molecules. 2. Intermolecular Forces. Forces that exist between molecules. 3. Electronegativity. The ability of an atom to attract to itself an electron pair shared with ...
All of these can affect the rate at which a
... C chemical formula. D covalent bond 60. In many compounds, atoms of main-group elements form bonds so that the number of electrons in the outermost energy levels of each atom is A 10 B 8. C 6. D 2 61. What principle states that atoms tend to form compounds so that each atom can have eight electrons ...
... C chemical formula. D covalent bond 60. In many compounds, atoms of main-group elements form bonds so that the number of electrons in the outermost energy levels of each atom is A 10 B 8. C 6. D 2 61. What principle states that atoms tend to form compounds so that each atom can have eight electrons ...
Chapter 2
... electronegativities is also __________. • Because carbon and hydrogen do not differ greatly in electronegativities, the bonds of CH4 are nonpolar. ...
... electronegativities is also __________. • Because carbon and hydrogen do not differ greatly in electronegativities, the bonds of CH4 are nonpolar. ...
Writing formulas and naming ionic bonds
... What type of nuclear reaction produces electricity? Fission When a chemical reaction occurs, the mass of the reactants ___ the mass of the products. Equals If the mass of the reactants is 10 g, then the mass of the products is ___ g. ...
... What type of nuclear reaction produces electricity? Fission When a chemical reaction occurs, the mass of the reactants ___ the mass of the products. Equals If the mass of the reactants is 10 g, then the mass of the products is ___ g. ...
Glossary
... Dynamics − the branch of mechanics dealing with the motion of physical systems. Equilibrium − stable, at minimum energy with no apparent motion. Equipartition theorem − consequence of the kinetic molecular theory that molecules have average kinetic energy proportional to the number of different type ...
... Dynamics − the branch of mechanics dealing with the motion of physical systems. Equilibrium − stable, at minimum energy with no apparent motion. Equipartition theorem − consequence of the kinetic molecular theory that molecules have average kinetic energy proportional to the number of different type ...
Chapter 4 Notes - Atomic Theory
... Remember: positive charges must = negative charges Ex.1: What is the formula for magnesium phosphide? Magnesium is Mg2+ Phosphorous is P3– Lowest common multiple of 2 and 3 is 6 3 Mg2+ ions & 2 P3– ions (6 +ve’s & 6 –ve’s) ...
... Remember: positive charges must = negative charges Ex.1: What is the formula for magnesium phosphide? Magnesium is Mg2+ Phosphorous is P3– Lowest common multiple of 2 and 3 is 6 3 Mg2+ ions & 2 P3– ions (6 +ve’s & 6 –ve’s) ...
Chemical bond
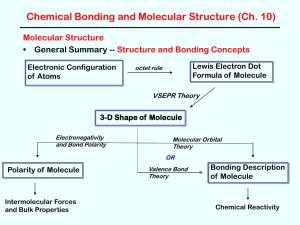
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.