Honors Chemistry Name_________________________________
... neutral atom in long form and short form. Write the electron configuration of an ion in long and short form. Relate the position of an atom on the periodic table and its predicted electron configuration. Identify the number of valence electrons in a given atom or ion. Explain the similaritie ...
... neutral atom in long form and short form. Write the electron configuration of an ion in long and short form. Relate the position of an atom on the periodic table and its predicted electron configuration. Identify the number of valence electrons in a given atom or ion. Explain the similaritie ...
Aps midREVIEW
... C. noble gas D. halogen 3. Which substance can be decomposed by chemical change? A. beryllium B. boron C. methanol D. magnesium 4. Which element is an active nonmetal? A. neon B. oxygen C. zinc D. chromium 5. To which group do the alkaline earth metals belong? A. 1 B. 2 C. 11 D. 1 ...
... C. noble gas D. halogen 3. Which substance can be decomposed by chemical change? A. beryllium B. boron C. methanol D. magnesium 4. Which element is an active nonmetal? A. neon B. oxygen C. zinc D. chromium 5. To which group do the alkaline earth metals belong? A. 1 B. 2 C. 11 D. 1 ...
Covalent bond indices and ionicities from similarity measures
... expressed as a percentage iBC rBH ¼ 85:8 which is the value given in the table. The other quantities are obtained straight from the formulas indicated above. As can be seen in that Table, the greatest quantitative difference between both metric definition results appears in the ionicity contributions ...
... expressed as a percentage iBC rBH ¼ 85:8 which is the value given in the table. The other quantities are obtained straight from the formulas indicated above. As can be seen in that Table, the greatest quantitative difference between both metric definition results appears in the ionicity contributions ...
Unit 4 review sheet
... Blast from the Past III – Quantum Numbers and Electron Configuration Practice! 1. When an electron in a hydrogen atom moves from a higher to a lower energy state, the energy difference is emitted as a quantum of ________. 2. Define the four quantum numbers (n, l, ml, ms) explain what information is ...
... Blast from the Past III – Quantum Numbers and Electron Configuration Practice! 1. When an electron in a hydrogen atom moves from a higher to a lower energy state, the energy difference is emitted as a quantum of ________. 2. Define the four quantum numbers (n, l, ml, ms) explain what information is ...
Notes-15 - KSU Physics
... The example in He shows that we can think that for each atom, there are electron orbitals, designated by n . Depending on the number of electrons available, one can put each electron in one of the orbitals. Each one of these orbitals for a fixed n is called a subshell, and each fixed n is called ...
... The example in He shows that we can think that for each atom, there are electron orbitals, designated by n . Depending on the number of electrons available, one can put each electron in one of the orbitals. Each one of these orbitals for a fixed n is called a subshell, and each fixed n is called ...
Review - Final Exam
... pure substances? Explain. How can the other term apply to substances and mixtures? Use examples to explain why. 7. What is the difference between: an element and a compound, an element and an atom, a compound and a molecule, & an element and an ion? Is it possible to have a molecule of an element? E ...
... pure substances? Explain. How can the other term apply to substances and mixtures? Use examples to explain why. 7. What is the difference between: an element and a compound, an element and an atom, a compound and a molecule, & an element and an ion? Is it possible to have a molecule of an element? E ...
ki̇mya
... • the electrons in an atom move at a certain distance from nucleus and their motions are stable . Each stationery state has a definite energy. • Electrons move in each stationary energy state in a circular orbital. These circular orbitals are called energy levels or shells. The possible states for t ...
... • the electrons in an atom move at a certain distance from nucleus and their motions are stable . Each stationery state has a definite energy. • Electrons move in each stationary energy state in a circular orbital. These circular orbitals are called energy levels or shells. The possible states for t ...
Dipole Moment & Polarity
... (d) that separates the centers of positive and negative charge. m = qd A unit of dipole moment is the debye (D). ...
... (d) that separates the centers of positive and negative charge. m = qd A unit of dipole moment is the debye (D). ...
Quantum Numbers and Electron Configurations Worksheet
... Use a phrase to describe why the 2s orbital is more stable (lower energy) versus 2p. When you superimpose the total radial probability of 2s and 2p onto the plot of 1s, you notice that the 2s has a small peak that is inside the 1s shield, which causes them to have more exposure to the full nuclear c ...
... Use a phrase to describe why the 2s orbital is more stable (lower energy) versus 2p. When you superimpose the total radial probability of 2s and 2p onto the plot of 1s, you notice that the 2s has a small peak that is inside the 1s shield, which causes them to have more exposure to the full nuclear c ...
Chemistry Notes
... Acids are compounds which break into Hydrogen (H+) ions and another compound when placed in an aqueous (water) solution. Bases are compounds which break up into Hydroxide (OH-) ions and another compound when placed in an aqueous solution. ...
... Acids are compounds which break into Hydrogen (H+) ions and another compound when placed in an aqueous (water) solution. Bases are compounds which break up into Hydroxide (OH-) ions and another compound when placed in an aqueous solution. ...
Application of Quantum Theory 1- Particle in 1
... position where probability of finding particle = 0 (No. of nodes = n-1) f. The probability of finding the particle between two points x 1 and x2 are different when n have small values, while as n increases (n >100) these 2 probabilities become the same. Generally the probability density become unifo ...
... position where probability of finding particle = 0 (No. of nodes = n-1) f. The probability of finding the particle between two points x 1 and x2 are different when n have small values, while as n increases (n >100) these 2 probabilities become the same. Generally the probability density become unifo ...
Word - chemmybear.com
... d. Molecules move with respect to one another but are held together in a condensed state. e. Molecules close together but having sufficiently high kinetic energies to overcome the intermolecular forces. f. Valence electrons delocalized over huge arrays of atoms. g. Totally random molecular order wit ...
... d. Molecules move with respect to one another but are held together in a condensed state. e. Molecules close together but having sufficiently high kinetic energies to overcome the intermolecular forces. f. Valence electrons delocalized over huge arrays of atoms. g. Totally random molecular order wit ...
Chapter 2: Atomic Structure and Inter-atomic Bonding
... expansion and relatively small dimensional alterations for changes in temperature. ...
... expansion and relatively small dimensional alterations for changes in temperature. ...
Ionic Bonding - Effingham County Schools
... Two models of the crystal structure of sodium chloride are shown. (a)To illustrate the ions’ actual arrangement, the sodium and chloride ions are shown with their electron clouds just touching. (b) In an expanded view, the distances between ions have been exaggerated in order to clarify the positio ...
... Two models of the crystal structure of sodium chloride are shown. (a)To illustrate the ions’ actual arrangement, the sodium and chloride ions are shown with their electron clouds just touching. (b) In an expanded view, the distances between ions have been exaggerated in order to clarify the positio ...
Chemistry at Karlsruhe 1860
... • Like the Berzelians they dealt only with empirical data and equivalent weights • Organic Compounds generally are made up of carbon, hydrogen and oxygen • Organic compounds when combusted in air form water, and carbon dioxide • Liebig determined the simplest formulas (empirical formulas) of any com ...
... • Like the Berzelians they dealt only with empirical data and equivalent weights • Organic Compounds generally are made up of carbon, hydrogen and oxygen • Organic compounds when combusted in air form water, and carbon dioxide • Liebig determined the simplest formulas (empirical formulas) of any com ...
PHY 104: Modern Physics - Physlab
... others soft, why do metals, for example, conduct electricity and heat easily, while glass doesn’t. Quantum physics also forms the basis of our understanding of the chemical world, materials science, as well as electronic devices permeating the modern digital age. The course is aimed at introducing t ...
... others soft, why do metals, for example, conduct electricity and heat easily, while glass doesn’t. Quantum physics also forms the basis of our understanding of the chemical world, materials science, as well as electronic devices permeating the modern digital age. The course is aimed at introducing t ...
Section 4.2 The Quantum Model of the Atom
... Thomson’s experiments demonstrated that electrons act like particles that have mass. In 1924, Louis de Broglie pointed out that the behavior of electrons according to Bohr’s model was similar to the behavior of waves. De Broglie suggested that electrons could be considered waves confined to the spac ...
... Thomson’s experiments demonstrated that electrons act like particles that have mass. In 1924, Louis de Broglie pointed out that the behavior of electrons according to Bohr’s model was similar to the behavior of waves. De Broglie suggested that electrons could be considered waves confined to the spac ...
Ms - cloudfront.net
... 18. Describe how a cation and an anion is formed. 19. What do metals typically do when they become ions? What about nonmetals? 20. What type of elements bond together in ionic bonds? covalent bonds? metallic bonds? 21. How do electrons in ionic bonding interact? Covalent bonding? 22. How does the re ...
... 18. Describe how a cation and an anion is formed. 19. What do metals typically do when they become ions? What about nonmetals? 20. What type of elements bond together in ionic bonds? covalent bonds? metallic bonds? 21. How do electrons in ionic bonding interact? Covalent bonding? 22. How does the re ...
Glossary - WordPress.com
... In the light of experiments, the scientists try to explain observations and facts. This tentative explanation is called hypothesis. It is quite possible that after sometime, on the basis of new experiments this hypothesis may be rejected. Hydrogen Bonding When a hydrogen atom is attached to any one ...
... In the light of experiments, the scientists try to explain observations and facts. This tentative explanation is called hypothesis. It is quite possible that after sometime, on the basis of new experiments this hypothesis may be rejected. Hydrogen Bonding When a hydrogen atom is attached to any one ...
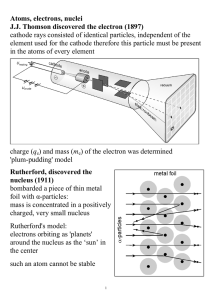
Atoms, electrons, nuclei J.J. Thomson discovered the electron (1897
... through a thin metal foil: interference interference phenomena have been shown with various other particles: duality is a general characteristic of matter Bohr's model (incorrect, but useful) electrons in an atom can only occupy certain distinct orbits around the nucleus: no radiation atoms radiate ...
... through a thin metal foil: interference interference phenomena have been shown with various other particles: duality is a general characteristic of matter Bohr's model (incorrect, but useful) electrons in an atom can only occupy certain distinct orbits around the nucleus: no radiation atoms radiate ...
2nd Semester Exam 1 Review Key
... 4) Benzene (C6H6) combusts with oxygen gas to yield carbon dioxide and water. 2C6H6 + 15O2 → 6H2O + 12CO2 combustion ...
... 4) Benzene (C6H6) combusts with oxygen gas to yield carbon dioxide and water. 2C6H6 + 15O2 → 6H2O + 12CO2 combustion ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.