V. Chemical reactions
... iii.Catalysts- affect reactions without being part of the reaction iv. Concentration- how much of a material is in a given area ...
... iii.Catalysts- affect reactions without being part of the reaction iv. Concentration- how much of a material is in a given area ...
CHEM_S1CourseReview_2011
... How do intermolecular forces differ from intramolecular forces? How is a positive and negative ion formed? Why does an element gain or lose electrons to become and ion? How do ionic compounds form? How does covalent bonding satisfy the octet rule? How is the bonding in Covalent (molecula ...
... How do intermolecular forces differ from intramolecular forces? How is a positive and negative ion formed? Why does an element gain or lose electrons to become and ion? How do ionic compounds form? How does covalent bonding satisfy the octet rule? How is the bonding in Covalent (molecula ...
Worksheet 3A on Molecules
... electronegativity between O and C; O3 is polar because it has 3 RHED and one lone pair on the central atom. This lone pair is an area where negative charge is concentrated, so this results in the molecule having an overall dipole moment. In the other species, I2 and CS2 are both linear and in the ca ...
... electronegativity between O and C; O3 is polar because it has 3 RHED and one lone pair on the central atom. This lone pair is an area where negative charge is concentrated, so this results in the molecule having an overall dipole moment. In the other species, I2 and CS2 are both linear and in the ca ...
Document
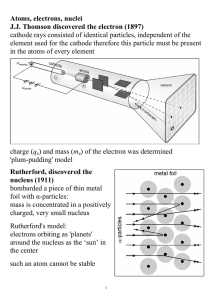
... vi. As a result, the atom’s positive and negative charges are balanced, and its charge is zero, or neutral. f. 3.6 Introduction to the Modern Concept of Atomic Structure i. An atom’s electrons move around the nucleus at a distance of about 63,000 times the diameter of the nucleus. ii. Thus, most of ...
... vi. As a result, the atom’s positive and negative charges are balanced, and its charge is zero, or neutral. f. 3.6 Introduction to the Modern Concept of Atomic Structure i. An atom’s electrons move around the nucleus at a distance of about 63,000 times the diameter of the nucleus. ii. Thus, most of ...
Counting atoms
... accurate determination of NA needs to be realized. By fixing NA, the atomic model of matter becomes embedded in the International System of Units (SI) and the mole redefined in terms of the number of entities it represents. Furthermore, as the ratio between the 28Si mass and the Planck constant can ...
... accurate determination of NA needs to be realized. By fixing NA, the atomic model of matter becomes embedded in the International System of Units (SI) and the mole redefined in terms of the number of entities it represents. Furthermore, as the ratio between the 28Si mass and the Planck constant can ...
objectives chm 1025 - Miami Dade College
... b. Demonstrating an ability to understand electronic transitions by working problems involving the Rydberg equation for hydrogen-like species. [OPTIONAL] c. Comparing and contrasting the particle and wave description of light. d. Relating important advances made in atomic theory to electronic emissi ...
... b. Demonstrating an ability to understand electronic transitions by working problems involving the Rydberg equation for hydrogen-like species. [OPTIONAL] c. Comparing and contrasting the particle and wave description of light. d. Relating important advances made in atomic theory to electronic emissi ...
Exam2
... Ethylene glycol is the primary component in antifreeze. How many grams ofethylene glycol, C2H4(OH)2 are in 394 mL of a 9.087 M solution of antifreeze? (molar mass C2H4(OH)2 = 62 g/mol) a. 222 g b. 43.7 g c. 3.58 g d. 2.69 g e. 0.157 g Calculate the number of oxygen atoms in 45.0 g of Co2(SO4)3 (mola ...
... Ethylene glycol is the primary component in antifreeze. How many grams ofethylene glycol, C2H4(OH)2 are in 394 mL of a 9.087 M solution of antifreeze? (molar mass C2H4(OH)2 = 62 g/mol) a. 222 g b. 43.7 g c. 3.58 g d. 2.69 g e. 0.157 g Calculate the number of oxygen atoms in 45.0 g of Co2(SO4)3 (mola ...
Answer Key
... E) 42 g 9. The mass of 1.63 1021 silicon atoms is A) 1.04 104 g. B) 28.08 g. C) 2.71 10–23 g. D) 7.60 10–2 g. E) 4.58 1022 g. ...
... E) 42 g 9. The mass of 1.63 1021 silicon atoms is A) 1.04 104 g. B) 28.08 g. C) 2.71 10–23 g. D) 7.60 10–2 g. E) 4.58 1022 g. ...
Final Exam review semester 1
... 1. Ninety-nine percent of all the matter that can be observed in the universe exists as 2. Hydrochloric acid, HCl, is added to solid NaOH. After the reaction is complete, NaCl dissolved in water remains. What are the products of this chemical reaction? ____ ...
... 1. Ninety-nine percent of all the matter that can be observed in the universe exists as 2. Hydrochloric acid, HCl, is added to solid NaOH. After the reaction is complete, NaCl dissolved in water remains. What are the products of this chemical reaction? ____ ...
8.P.1.1 Warm-Up Questions for Website
... B.It can be formed through a physical reaction. C.It can be changed into simpler substances through a physical change. D.It is a pure substance containing elements that are chemically combined. ...
... B.It can be formed through a physical reaction. C.It can be changed into simpler substances through a physical change. D.It is a pure substance containing elements that are chemically combined. ...
ATOMIC STRUCTURE NOTES n hcZ E ℜ
... can penetrate, while a 2p electron is not as effective at this because it has a nodal plane through the nucleus. Therefore, it is more shielded from the nucleus by the electrons of the core. We can conclude that a 2s electron has lower energy (more bound) than a 2p, so for Lithium the ground state c ...
... can penetrate, while a 2p electron is not as effective at this because it has a nodal plane through the nucleus. Therefore, it is more shielded from the nucleus by the electrons of the core. We can conclude that a 2s electron has lower energy (more bound) than a 2p, so for Lithium the ground state c ...
File
... Formation of a permanent dipole – (polar covalent) bond A polar covalent bond forms when the elements in the bond have different electronegativities . (Of around 0.3 to 1.7) When a bond is a polar covalent bond it has an unequal distribution of electrons in the bond and produces a charge separation, ...
... Formation of a permanent dipole – (polar covalent) bond A polar covalent bond forms when the elements in the bond have different electronegativities . (Of around 0.3 to 1.7) When a bond is a polar covalent bond it has an unequal distribution of electrons in the bond and produces a charge separation, ...
15.2 Electrons and Chemical Bonds
... numbers combines with other atoms to make a compound. Therefore, we say that sodium has an oxidation number of 1+. An oxidation number indicates the charge on the remaining atom (ion) when electrons are lost, gained, or shared in chemical bonds. Table 15.1 shows the oxidation numbers for some elemen ...
... numbers combines with other atoms to make a compound. Therefore, we say that sodium has an oxidation number of 1+. An oxidation number indicates the charge on the remaining atom (ion) when electrons are lost, gained, or shared in chemical bonds. Table 15.1 shows the oxidation numbers for some elemen ...
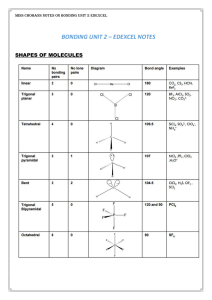
Structural Organic Chemistry The Shapes of Molecules Functional
... bond lengths with some degree of reality. In the ball-and-stick type, the sticks usually come in various lengths to simulate different kinds of bonds; C-H bonds typically are scaled to represent 1.1 A, C-C bonds to be 1.54 A, and C=C and C = C to be correspondingly shorter. In some model sets the bo ...
... bond lengths with some degree of reality. In the ball-and-stick type, the sticks usually come in various lengths to simulate different kinds of bonds; C-H bonds typically are scaled to represent 1.1 A, C-C bonds to be 1.54 A, and C=C and C = C to be correspondingly shorter. In some model sets the bo ...
Honors Chemistry
... Students should be able to identify these elements simply based on total number of electrons Significance of electron configurations Valence shell electrons - outermost electrons involved with bonding for n = 5, pattern is very complicated - no atom has more than 8 valence electrons Noble gases - 8 ...
... Students should be able to identify these elements simply based on total number of electrons Significance of electron configurations Valence shell electrons - outermost electrons involved with bonding for n = 5, pattern is very complicated - no atom has more than 8 valence electrons Noble gases - 8 ...
Chemical Equations and Reactions
... The negative ion of one compound replaces the negative ion of the other compound to form 2 new compounds. Usually forms a precipitate, water or a gas. ...
... The negative ion of one compound replaces the negative ion of the other compound to form 2 new compounds. Usually forms a precipitate, water or a gas. ...
CHEM 121 Chp 2 Spaulding
... elements gives it a versatility not seen with any other element in the periodic table Elemental forms of carbon include the following carbon-only structures: ...
... elements gives it a versatility not seen with any other element in the periodic table Elemental forms of carbon include the following carbon-only structures: ...
CHEM1405 2012-J-2 June 2012 • What is the ground state electron
... • Glycine, NH2CH2COOH, the simplest of all naturally occurring amino acids, has a melting point of 292 °C. The pKa of the acid group is 2.35 and the pKa associated with the amino group is 9.78. Draw a Lewis structure that indicates the charges on the molecule at the physiological pH of 7.4. ...
... • Glycine, NH2CH2COOH, the simplest of all naturally occurring amino acids, has a melting point of 292 °C. The pKa of the acid group is 2.35 and the pKa associated with the amino group is 9.78. Draw a Lewis structure that indicates the charges on the molecule at the physiological pH of 7.4. ...
Atomic
... atomic theory that he created using the laws of matter and previously known atomic theory 1. All matter is composed of atoms 2. All atoms of a given element are identical in size, mass, and other properties 3. Atoms can not be divided, created or destroyed 4. Atoms of different elements combine in s ...
... atomic theory that he created using the laws of matter and previously known atomic theory 1. All matter is composed of atoms 2. All atoms of a given element are identical in size, mass, and other properties 3. Atoms can not be divided, created or destroyed 4. Atoms of different elements combine in s ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.