Booklet Chapter 3
... Water of hydration The associated water in ionic hydrates. Electronegativity A measure of the electron-attracting ability of an atom in a chemical bond. Molecular compound A compound composed of molecules. In such compounds, all of the bonds between atoms are covalent bonds. Ionic compound A compoun ...
... Water of hydration The associated water in ionic hydrates. Electronegativity A measure of the electron-attracting ability of an atom in a chemical bond. Molecular compound A compound composed of molecules. In such compounds, all of the bonds between atoms are covalent bonds. Ionic compound A compoun ...
minerals & magma
... ** If atoms loose or gain electrons they have an electrical charge (-/+) and are called ions; also, a group of atoms (molecule) can behave as an ion if they have too many or too few electrons. ...
... ** If atoms loose or gain electrons they have an electrical charge (-/+) and are called ions; also, a group of atoms (molecule) can behave as an ion if they have too many or too few electrons. ...
atoms
... Unfortunately, there is no alternative: The names and charges of the common polyatomic ions must be memorized. (a) Lithium nitrate Lithium (group 1A) forms only the Li+ ion and does not need a Roman numeral. (b) Potassium hydrogen sulfate (potassium bisulfate) Potassium (group 1A) forms only the K+ ...
... Unfortunately, there is no alternative: The names and charges of the common polyatomic ions must be memorized. (a) Lithium nitrate Lithium (group 1A) forms only the Li+ ion and does not need a Roman numeral. (b) Potassium hydrogen sulfate (potassium bisulfate) Potassium (group 1A) forms only the K+ ...
chapter 1 - College Test bank - get test bank and solution manual
... 7. This answer can vary between students, but the combustion of gasoline to propel vehicles is an example of a useful chemical reaction. This reaction is a chemical change because the liquid gasoline is converted to heat and gases. Another constructive example is the burning of coal to heat water in ...
... 7. This answer can vary between students, but the combustion of gasoline to propel vehicles is an example of a useful chemical reaction. This reaction is a chemical change because the liquid gasoline is converted to heat and gases. Another constructive example is the burning of coal to heat water in ...
In the Classroom
... internuclear separation decreases. As will be shown later, the immediate cause of the molecular ground state is a sharp increase in electron kinetic energy. • The amount of electron density transferred to the bonding region is greatly overstated, sometimes implying that a pair of electrons is shared ...
... internuclear separation decreases. As will be shown later, the immediate cause of the molecular ground state is a sharp increase in electron kinetic energy. • The amount of electron density transferred to the bonding region is greatly overstated, sometimes implying that a pair of electrons is shared ...
end of year review
... _____ 5. Which is an example of a non-polar molecule that contains polar covalent bonds? A. CCl4 ...
... _____ 5. Which is an example of a non-polar molecule that contains polar covalent bonds? A. CCl4 ...
Spring Benchmark Exam
... have stronger forces of attraction between them. take the shape of the container they are in. ...
... have stronger forces of attraction between them. take the shape of the container they are in. ...
Bonding Experiments for Cryogenic Detectors
... [1] A. Dari et al. Breaking strength tests on silicon and sapphire bonding for gravitational wave detectors. Classical and Quantum Gravity, 27(4): 045010, 2010 [2] T. Suzuki et al. Application of sapphire bonding for suspension of cryogenic mirrors. Journal of Physics: Conference series, 32, pp 309, ...
... [1] A. Dari et al. Breaking strength tests on silicon and sapphire bonding for gravitational wave detectors. Classical and Quantum Gravity, 27(4): 045010, 2010 [2] T. Suzuki et al. Application of sapphire bonding for suspension of cryogenic mirrors. Journal of Physics: Conference series, 32, pp 309, ...
1) abcde 2) abcde 3) abcde 4) abcde 5) abcde 6) abcde 7) abcde 8
... 38) Explain how the ionization energy of fluorine as compared to oxygen is related to their relative atomic sizes. [3 marks] 39) Bonds may be covalent, polar covalent or ionic. Discuss the relationship between the electronegativities of the elements and the progression in bond character for F2 , CF4 ...
... 38) Explain how the ionization energy of fluorine as compared to oxygen is related to their relative atomic sizes. [3 marks] 39) Bonds may be covalent, polar covalent or ionic. Discuss the relationship between the electronegativities of the elements and the progression in bond character for F2 , CF4 ...
Chemistry specialism additional subject knowledge audit Ratings S
... mass. The use of mass spectrometry Calculations: reacting masses, concentrations, gases, percentage yields and atom economies Acid–base titrations ...
... mass. The use of mass spectrometry Calculations: reacting masses, concentrations, gases, percentage yields and atom economies Acid–base titrations ...
Chemistry Review Module Chapter 1
... Why? Because the least precise measurement had 3 significant digits, so our answer should not have more than 3 significant digits! The technique for addition and subtraction is slightly different (see p.396 ) but the concept is the same. You cannot make your result better than your measurements! ...
... Why? Because the least precise measurement had 3 significant digits, so our answer should not have more than 3 significant digits! The technique for addition and subtraction is slightly different (see p.396 ) but the concept is the same. You cannot make your result better than your measurements! ...
File
... A crystalline solid with a high melting point which conducts electricity only when molten or dissolved in water is: a. a molecular compound c. a metal b. an ionic compound d. a network covalent solid ...
... A crystalline solid with a high melting point which conducts electricity only when molten or dissolved in water is: a. a molecular compound c. a metal b. an ionic compound d. a network covalent solid ...
2s - Chemistry
... orbitals • when the combining atomic orbitals are identical and equal energy, the weight of each atomic orbital in the molecular orbital are equal • when the combining atomic orbitals are different kinds and energies, the atomic orbital closest in energy to the molecular orbital contributes more to ...
... orbitals • when the combining atomic orbitals are identical and equal energy, the weight of each atomic orbital in the molecular orbital are equal • when the combining atomic orbitals are different kinds and energies, the atomic orbital closest in energy to the molecular orbital contributes more to ...
Mn2 1 Many-particle Systems, 2 Multi
... Example: The ground state electronic configuration for H is 1s1; the first excited state (ignoring small magnetic effects) is 2s1. The ground state for He is 1s2; the first excited state is 1s12s1. The ground state for Li is 1s22s1; the first excited state is 1s22p1, not 1s12s2. The reason for the ...
... Example: The ground state electronic configuration for H is 1s1; the first excited state (ignoring small magnetic effects) is 2s1. The ground state for He is 1s2; the first excited state is 1s12s1. The ground state for Li is 1s22s1; the first excited state is 1s22p1, not 1s12s2. The reason for the ...
Chemical Reactions Chemical Arithmetic
... (ClO4-), or sulfate (SO42-) anions. • Exceptions: Ag+, Hg22+, & Pb2+ with halides and Ba2+, Hg22+ and Pb2+ with sulfates ...
... (ClO4-), or sulfate (SO42-) anions. • Exceptions: Ag+, Hg22+, & Pb2+ with halides and Ba2+, Hg22+ and Pb2+ with sulfates ...
first chapter - damtp - University of Cambridge
... some of the energy, W , is absorbed by the other particles in the metal.) However, a free particle can scatter the photon, so changing its energy and therefore its frequency; at the same time the particle recoils. The kinematics of the process can be worked out using (1.2), (1.4) and the relativisti ...
... some of the energy, W , is absorbed by the other particles in the metal.) However, a free particle can scatter the photon, so changing its energy and therefore its frequency; at the same time the particle recoils. The kinematics of the process can be worked out using (1.2), (1.4) and the relativisti ...
The Periodic Table - Mrs Molchany`s Webpage
... Harder than alkali metals More dense than alkali metals Higher melting points than alkali metals ...
... Harder than alkali metals More dense than alkali metals Higher melting points than alkali metals ...
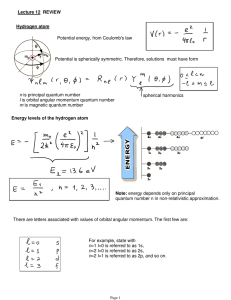
Lecture 12: Review.
... Example: hydrogen ground state J=1/2, I=1/2 (proton) F=0,1, so two levels (see picture on previous page) ...
... Example: hydrogen ground state J=1/2, I=1/2 (proton) F=0,1, so two levels (see picture on previous page) ...
- TestbankU
... 5) Why is it so difficult to remove the lid from a vacuum-sealed jar? A) The air pressure outside the jar pushes downward on the lid more strongly than the air pressure inside pushes upward on the lid. B) The vacuum inside the jar pushes outward on the lid, holding it firmly to the jar. C) The vacuu ...
... 5) Why is it so difficult to remove the lid from a vacuum-sealed jar? A) The air pressure outside the jar pushes downward on the lid more strongly than the air pressure inside pushes upward on the lid. B) The vacuum inside the jar pushes outward on the lid, holding it firmly to the jar. C) The vacuu ...
Syllabus for Chemical Sciences Inorganic 1. Atomic structure and
... 1. Atomic structure and chemical periodicity X‐ray spectra and atomic number, hydrogen spectra and Bohr’s theory, Sommerfeld’s theory and new energy states (derivation excluded), quantum rules and quantum numbers, Pauli exclusion principle (antisymmetry principle), Hund’s rule, Aufbau principle ...
... 1. Atomic structure and chemical periodicity X‐ray spectra and atomic number, hydrogen spectra and Bohr’s theory, Sommerfeld’s theory and new energy states (derivation excluded), quantum rules and quantum numbers, Pauli exclusion principle (antisymmetry principle), Hund’s rule, Aufbau principle ...
Atomic and Molecular Physics for Physicists Ben-Gurion University of the Negev
... J in the lowest energy state of the EXCITED atom (this will be the one interacting with the light as the light is red-detuned) ...
... J in the lowest energy state of the EXCITED atom (this will be the one interacting with the light as the light is red-detuned) ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.