Chemical Reactions Chemistry - is the study of matter, its properties
... exhibit behaviors similar to both metals and non-metals. However, all members of this Family are ready to donate three electrons found in their valence shell. The non-metals in the Groups usually form compounds by sharing electrons. Group IV A *******check metals and covalent****** This group which ...
... exhibit behaviors similar to both metals and non-metals. However, all members of this Family are ready to donate three electrons found in their valence shell. The non-metals in the Groups usually form compounds by sharing electrons. Group IV A *******check metals and covalent****** This group which ...
Specification
... These are written in parentheses printed in italic type, immediately after the formula or substance and on the same line as chemical formula symbols. ...
... These are written in parentheses printed in italic type, immediately after the formula or substance and on the same line as chemical formula symbols. ...
Lecture notes in Solid State 3 Eytan Grosfeld
... (b) Surrounding the nucleus are Za electrons of total charge −eZa , composed of Z weakly bound valence electrons, and Za −Z tightly bound electrons, the core electrons. In a metal, the core electrons remain bound to the nucleus and form the (immobile, positively charged) metallic ion, while the (mob ...
... (b) Surrounding the nucleus are Za electrons of total charge −eZa , composed of Z weakly bound valence electrons, and Za −Z tightly bound electrons, the core electrons. In a metal, the core electrons remain bound to the nucleus and form the (immobile, positively charged) metallic ion, while the (mob ...
Lecture 7
... Going down the group the cations formed get bigger. This means that the positive charge is spread over a larger volume and so is less concentrated. A small ion with its concentrated charge has a high charge density. This high charge density gives the ion the ability to distort or polarize nearby ani ...
... Going down the group the cations formed get bigger. This means that the positive charge is spread over a larger volume and so is less concentrated. A small ion with its concentrated charge has a high charge density. This high charge density gives the ion the ability to distort or polarize nearby ani ...
if on the Internet, press on your browser to
... As you use chemistry to move a metal toward the ORMUS/BEC state, the chemical reactions necessary to do this moving become weaker-and-weaker since fewer and fewer of the valence electrons are available to participate in the chemical reactions. Eventually there are no electron handles that can be use ...
... As you use chemistry to move a metal toward the ORMUS/BEC state, the chemical reactions necessary to do this moving become weaker-and-weaker since fewer and fewer of the valence electrons are available to participate in the chemical reactions. Eventually there are no electron handles that can be use ...
Chemistry I Exam
... A student performed a laboratory procedure to determine the relative reactivity of elements bromine, chlorine, and iodine. To do this, she prepared a water solution of each element, as well as a solution of sodium salt (bromide, chloride, iodide) of each of these elements. In separate test tubes, sh ...
... A student performed a laboratory procedure to determine the relative reactivity of elements bromine, chlorine, and iodine. To do this, she prepared a water solution of each element, as well as a solution of sodium salt (bromide, chloride, iodide) of each of these elements. In separate test tubes, sh ...
Physical Science Semester 2 Final Exam 2013 –STUDY GUIDE
... 17. Increasing the speed of an object ____ its potential energy. 18. The SI (metric) unit for energy is the ____. 19. You can calculate kinetic energy by using the equation ____. 20. You can calculate gravitational potential energy by using the equation ____. 21. According to the law of conservatio ...
... 17. Increasing the speed of an object ____ its potential energy. 18. The SI (metric) unit for energy is the ____. 19. You can calculate kinetic energy by using the equation ____. 20. You can calculate gravitational potential energy by using the equation ____. 21. According to the law of conservatio ...
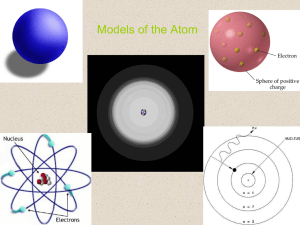
The Atom and Its Properties
... When n = 1, then l = 0 Therefore, in n = 1, there is just 1 type of sublevel and that sublevel has a single orbital This sublevel is labeled s (“ess”) Each level has 1 orbital labeled s, and it is SPHERICAL in shape. ...
... When n = 1, then l = 0 Therefore, in n = 1, there is just 1 type of sublevel and that sublevel has a single orbital This sublevel is labeled s (“ess”) Each level has 1 orbital labeled s, and it is SPHERICAL in shape. ...
Electron spin and the periodic table
... corresponding to spin up and spin down. Angular momentum, including the internal angular momentum, is always quantized in quantum mechanics. (Actually, the original experiment used silver atoms, but their moment comes from the spin of an unpaired electron, so it is basically the same ...
... corresponding to spin up and spin down. Angular momentum, including the internal angular momentum, is always quantized in quantum mechanics. (Actually, the original experiment used silver atoms, but their moment comes from the spin of an unpaired electron, so it is basically the same ...
E - Purdue Physics
... Polarization of Insulators Insulator: Electrons are bound to the atoms or molecules. Electrons can shift slightly (<1 Å), but remain bound to the molecule. Individual atoms or molecules can be polarized by external electric field. There are a lot of molecules – the net effect produced by the induce ...
... Polarization of Insulators Insulator: Electrons are bound to the atoms or molecules. Electrons can shift slightly (<1 Å), but remain bound to the molecule. Individual atoms or molecules can be polarized by external electric field. There are a lot of molecules – the net effect produced by the induce ...
Chapter 5: Electrons in Atoms
... 1. Determine the energy levels, sublevels and orbitals of electrons in an atom. 2. Determine how to write electron configuration and orbital notation for atoms and ions. ...
... 1. Determine the energy levels, sublevels and orbitals of electrons in an atom. 2. Determine how to write electron configuration and orbital notation for atoms and ions. ...
Chapter
... 1. An electron configuration describes the arrangement of electrons in the subshells of an atom. 2. The chemical properties of elements are related to these configurations. 3. The four quantum numbers n, l, m, and ms enable us to label completely an electron in any orbital in any atom. ...
... 1. An electron configuration describes the arrangement of electrons in the subshells of an atom. 2. The chemical properties of elements are related to these configurations. 3. The four quantum numbers n, l, m, and ms enable us to label completely an electron in any orbital in any atom. ...
There can be only one
... developed a spin-force-modified kinetic theory for a magnetized plasma with immobile ions. A new high-frequency ordinary mode, which is polarized parallel to the external magnetic field direction, appears. Furthermore, the model also identifies new types of wave–particle interactions involving the e ...
... developed a spin-force-modified kinetic theory for a magnetized plasma with immobile ions. A new high-frequency ordinary mode, which is polarized parallel to the external magnetic field direction, appears. Furthermore, the model also identifies new types of wave–particle interactions involving the e ...
Introduction to spectroscopy
... • The electronic interactions between atoms in molecules or solids provide a binding force and a restoring force often compared to springs. Therefore each system (molecule, solid) displays characteristic vibrations (normal modes) associated with bond stretching and bond bending motions (just like a ...
... • The electronic interactions between atoms in molecules or solids provide a binding force and a restoring force often compared to springs. Therefore each system (molecule, solid) displays characteristic vibrations (normal modes) associated with bond stretching and bond bending motions (just like a ...
Diodes and Transistors HOW Theq Work
... surrounded by a swarm of negatively charged electrons. The speeding electrons are held in orbits around the nucleus by electrostatic attraction. The nucleus receives its positive charge from positively charged protons. Each chemical element (hydrogen, oxygen, and silicon, for example) has a differen ...
... surrounded by a swarm of negatively charged electrons. The speeding electrons are held in orbits around the nucleus by electrostatic attraction. The nucleus receives its positive charge from positively charged protons. Each chemical element (hydrogen, oxygen, and silicon, for example) has a differen ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.